BEII SEMESTER ADVANCED PHYSICS UNITI LASERS AND WAVE

BE-II SEMESTER ADVANCED PHYSICS UNIT-I LASERS AND WAVE OPTICS DEPARTMENT OF APPLIED PHYSICS

SYLLABUS ØIntroduction & Quantum Transitions: Absorption, Spontaneous Emission & Stimulated Emission, Metastable State, Population Inversion ØPumping Schemes & LASER Properties, Spatial and temporal coherence of a light wave ØPrinciple & working of He-Ne LASER

SYLLABUS ØRuby Laser ØSemiconductor laser & applications ØIntroduction, interference in thin film, Wedge shaped thin film ØNewton’s Ring And Antireflection Coatings ØAdvanced applications of interference in thin film

LEARNING OBJECTIVES Ø Understand basic laser physics. Ø Describe the concept of stimulated emission and what is an active medium. Ø Learn about the types of laser and their applications. Ø Know what conditions are necessary for interference to be observed. Ø Know how light changes phase when it is reflected from a surface. Ø Understand the necessary conditions for interference to occur in thin films.

LASER L: LIGHT A: AMPLIFICATION by S: STIMULATED E: EMISSION of R: RADIATION
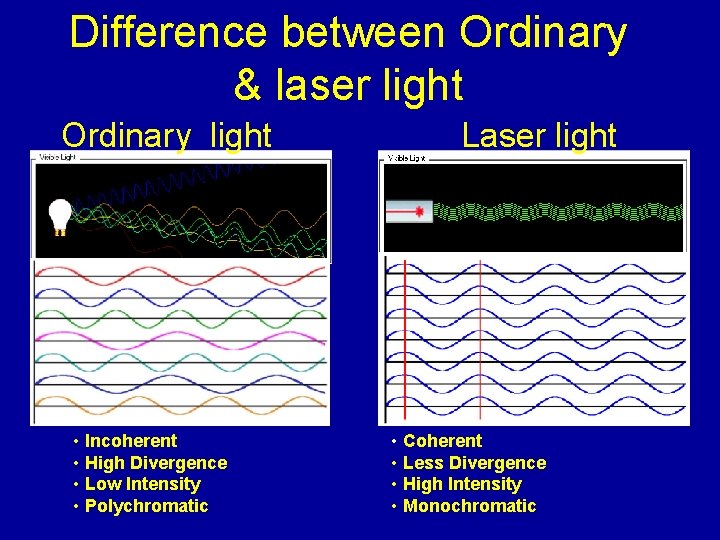
Difference between Ordinary & laser light Ordinary light • Incoherent • High Divergence • Low Intensity • Polychromatic Laser light • Coherent • Less Divergence • High Intensity • Monochromatic

Characteristics of Laser ØMonochromaticity ØCoherence v Temporal coherence v Spatial coherence ØIntensity ØUnidirectionaly ØDivergence

ØCoherence length (lcoh ) : The length of the wavetrain upto which it is perfectly sinusoidal. lcoh = c. tcoh = c Ø Coherence time(tcoh) : The time for which the wave train is perfectly sinusoidal. Ø Since = Δt = 1/ Δ טּ lcoh = c. ∆t = c / ∆ν, Ø We know , ν = c / λ ∴ ∆ν = - c. ∆ λ / λ 2 , ignoring minus sign lcoh = λ 2 / ∆ λ , Where Δ λ Bandwidth

Quantum processes in Laser: Three distinct processes can take place. i) Absorption ii) Spontaneous emission iii) Stimulated emission

Absorption and Emission Atom

1. Absorption Ø Energy of photon h =E 2 -E 1 ‘ absorb by atoms in the lower energy states and excites to higher energy states. ØEinstein Equation for absorption Nab = B 12 N 1ρ(υ)Δt Ø A + h A*
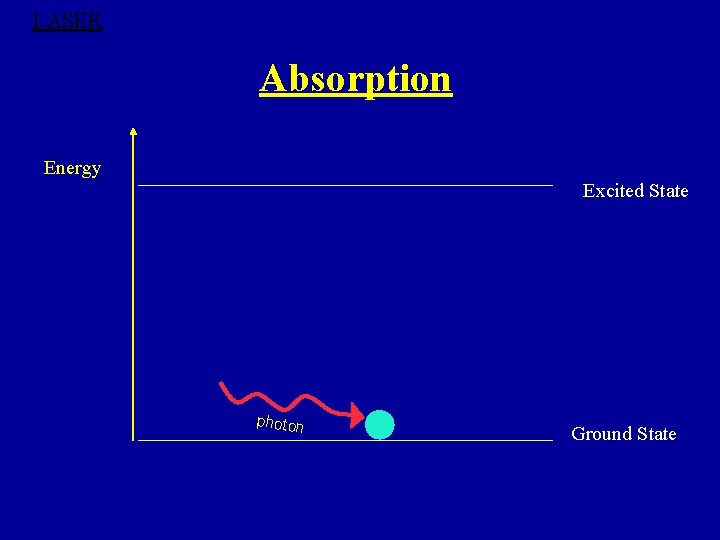
LASER Absorption Energy Excited State photon Ground State

1. Spontaneous Emission ØThe process of emission of photons by an excited atoms by its own , without the influence of external agent is called spontaneous emission. A* A + h Spontaneous Emission Nsp = A 21 N 2Δt , A* A + h (photon)

LASER Spontaneous Emission Energy Excited State photon Ground State

LASER Spontaneous Emission Energy photon photon Ground State
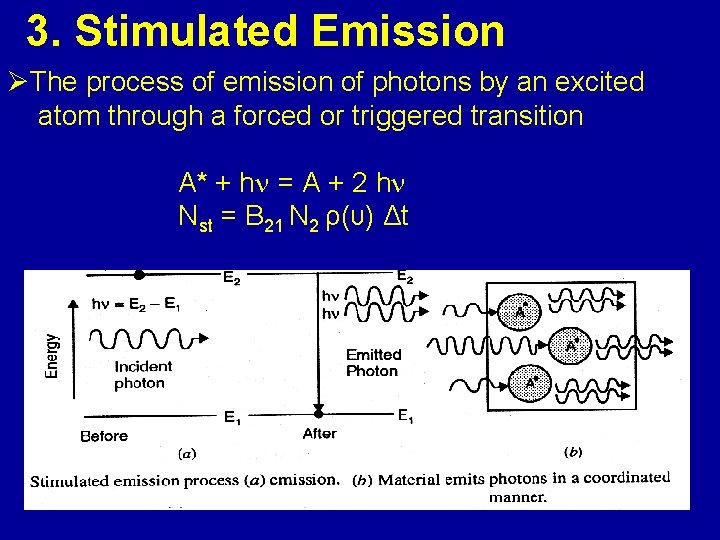
3. Stimulated Emission ØThe process of emission of photons by an excited atom through a forced or triggered transition A* + h = A + 2 h Nst = B 21 N 2 ρ(υ) Δt
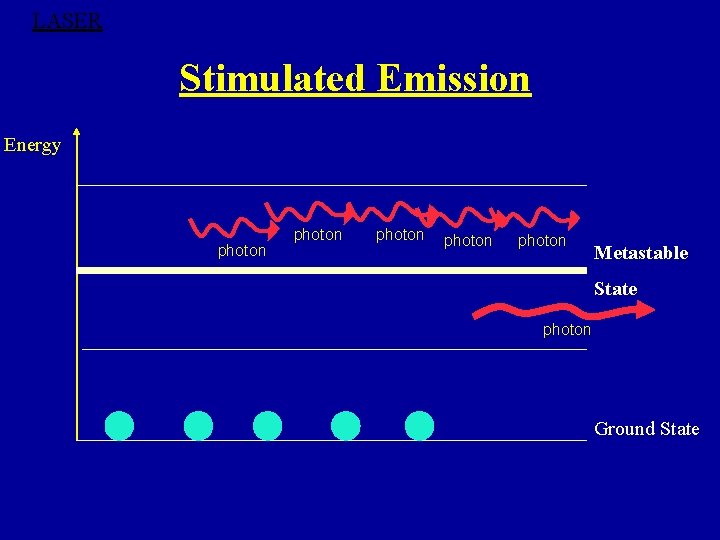
LASER Stimulated Emission Energy photon photon Metastable State photon Ground State
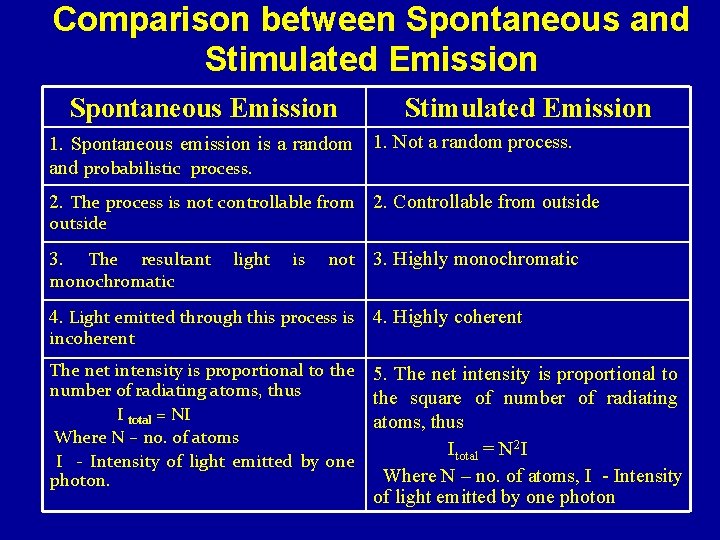
Comparison between Spontaneous and Stimulated Emission Spontaneous Emission Stimulated Emission 1. Spontaneous emission is a random 1. Not a random process. and probabilistic process. 2. The process is not controllable from 2. Controllable from outside 3. The resultant monochromatic light is not 3. Highly monochromatic 4. Light emitted through this process is 4. Highly coherent incoherent The net intensity is proportional to the number of radiating atoms, thus I total = NI Where N – no. of atoms I - Intensity of light emitted by one photon. 5. The net intensity is proportional to the square of number of radiating atoms, thus Itotal = N 2 I Where N – no. of atoms, I - Intensity of light emitted by one photon

Condition for Light Amplification 1) Condition for Stimulated emission to dominate Spontaneous Emission 2) Condition for stimulated emission to dominate absorption transitions This condition indicates that Stimulated transition will overwhelm the absorption process if N 2 is greater than N 1. The system must achieve the state of population inversion.
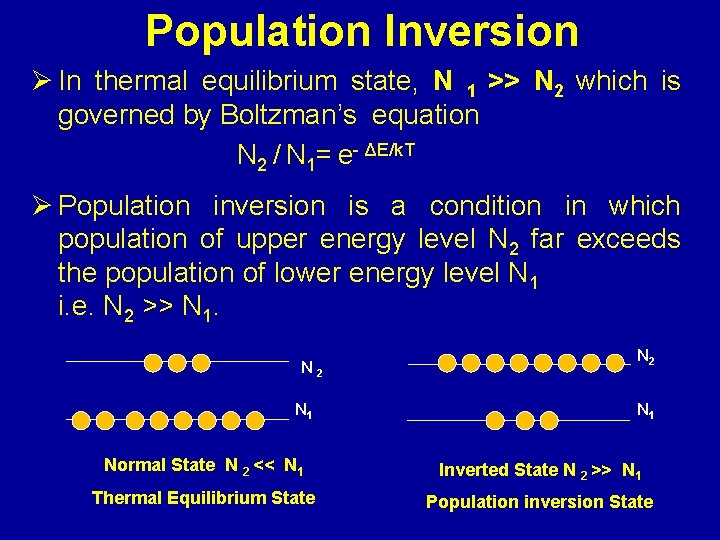
Population Inversion Ø In thermal equilibrium state, N 1 >> N 2 which is governed by Boltzman’s equation N 2 / N 1= e- ΔE/k. T Ø Population inversion is a condition in which population of upper energy level N 2 far exceeds the population of lower energy level N 1 i. e. N 2 >> N 1. N 2 N 1 Normal State N 2 << N 1 Inverted State N 2 >> N 1 Thermal Equilibrium State Population inversion State

Metastable State ØMetastable state can be defined as a state where excited atom can remain for longer time than the normal excited state. ØAtoms stay in metastable states for about 10 -6 to 10 -3 s. This is 103 to 106 times longer than the time of stay of atom at excited levels. ØIf the metastable states do not exist, there could be no population inversion, no stimulated emission and hence no laser operation.

Components of Laser The essential components of Laser are Ø An active medium Ø A pumping agent Ø Optical Resonator

An active medium ØA medium in which light gets amplified is called an active medium. ØThe medium may be solid , liquid or gas. ØActive centres are those atoms which are responsible for stimulated emission.

Pumping Ø The process of supplying energy to the medium with a view to transfer it into the state of population inversion is known as pumping. Ø Techniques to achieve the state of population inversion: § Optical pumping (used in Ruby Laser) § Electric discharge (used in He-Ne Laser) § Direct Conversion (used in Semiconductor Laser)

Fabry - Perot optical resonator (Resonant cavity) ØOptical resonator consist of two opposing plane parallel mirrors, with an active material placed in between them. ØOne of the mirror is semitransparent while the other is 100 % reflecting. ØThe mirrors are set normal to optic axis of the material. 100 % reflecting mirror Active medium Semitransparent mirror Optic axis
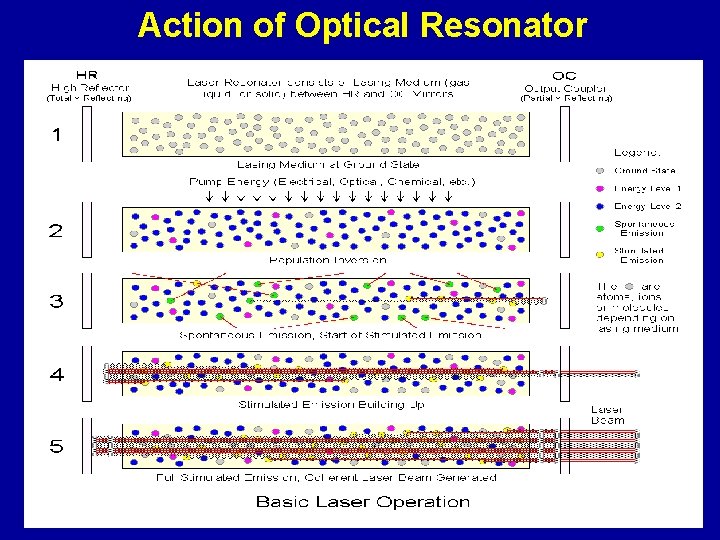
Action of Optical Resonator
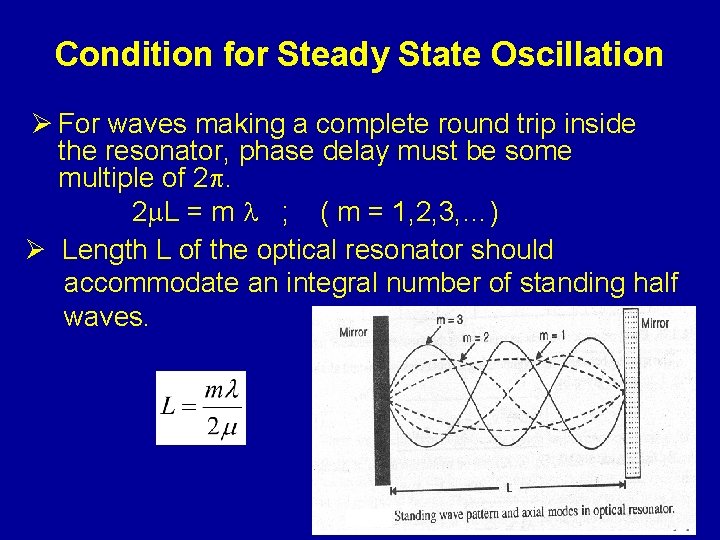
Condition for Steady State Oscillation Ø For waves making a complete round trip inside the resonator, phase delay must be some multiple of 2. 2 L = m ; ( m = 1, 2, 3, …) Ø Length L of the optical resonator should accommodate an integral number of standing half waves.

Pumping Schemes are Classified as: Ø Two-level Ø Three-level and Ø Four –level schemes. • Two-level scheme will not lead to laser action. • Three-level and four-level schemes are important and widely employed.

Two Level Pumping Scheme Energy ØPumping radiation excites the ground state atoms. ØInduces transitions from the upper level to the lower level. ØHence, population inversion cannot be attained in a two-level pumping scheme. E 2 E 1
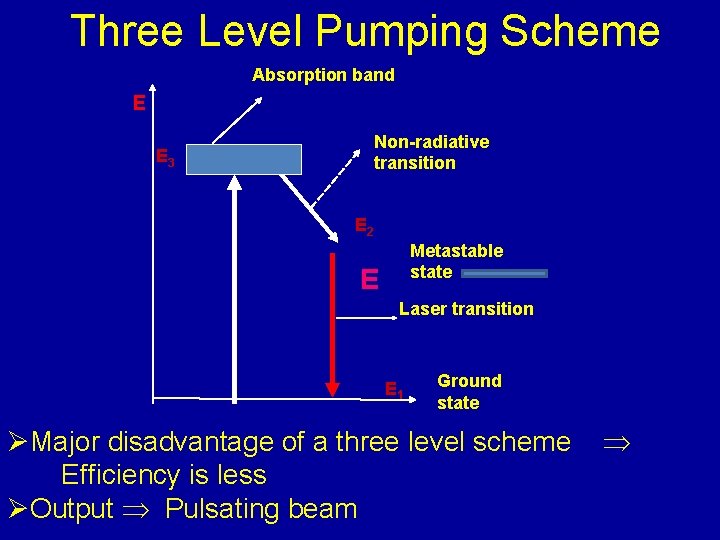
Three Level Pumping Scheme Absorption band E E 3 Non-radiative transition E 2 Metastable state E Laser transition E 1 Ground state ØMajor disadvantage of a three level scheme Efficiency is less ØOutput Pulsating beam
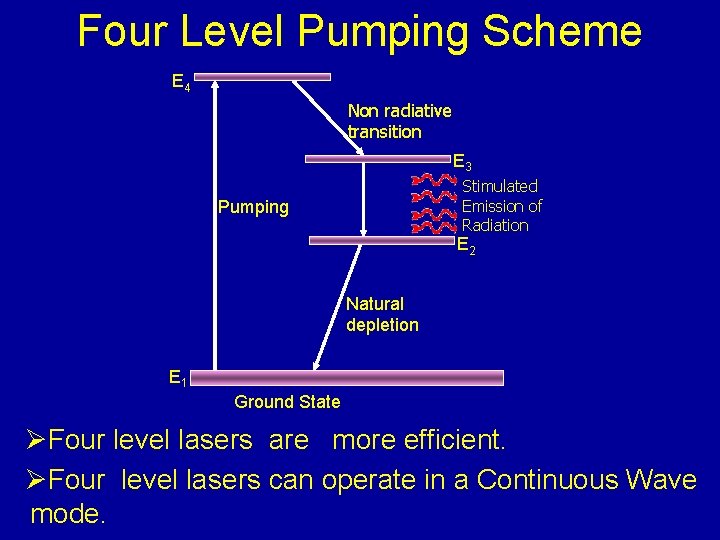
Four Level Pumping Scheme E 4 Non radiative transition E 3 Stimulated Emission of Radiation Pumping E 2 Natural depletion E 1 Ground State ØFour level lasers are more efficient. ØFour level lasers can operate in a Continuous Wave mode.

Types of LASER Ø Solid-state lasers – Ruby laser, “Nd: YAG“, Nd: Glass, lasers etc Ø Gas lasers - He-Ne, He-Cd, CO 2, N 2 lasers etc Ø Semiconductor lasers ( diode lasers)-These electronic devices are generally very small and use low power. Ga. As, Ga. As. P
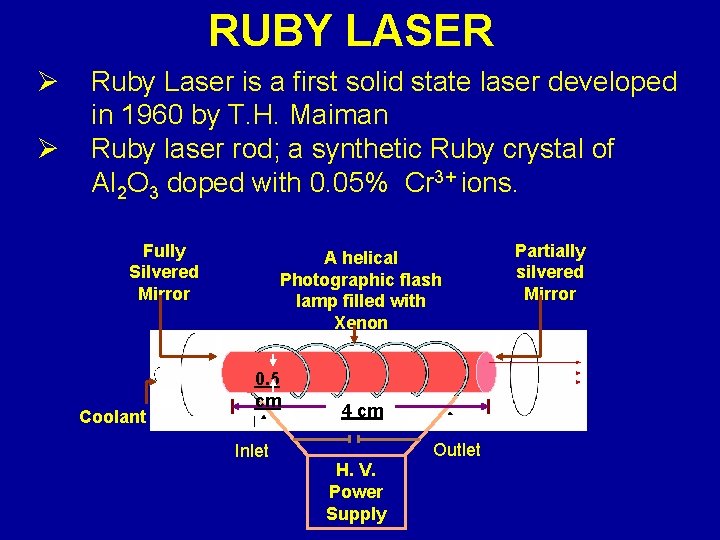
RUBY LASER Ø Ø Ruby Laser is a first solid state laser developed in 1960 by T. H. Maiman Ruby laser rod; a synthetic Ruby crystal of Al 2 O 3 doped with 0. 05% Cr 3+ ions. Fully Silvered Mirror Coolant A helical Photographic flash lamp filled with Xenon 0. 5 cm Inlet 4 cm Outlet H. V. Power Supply Partially silvered Mirror
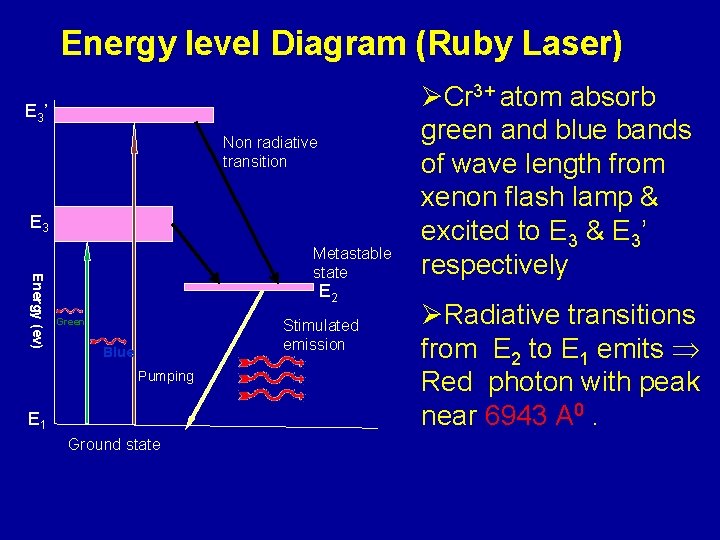
Energy level Diagram (Ruby Laser) E 3’ Non radiative transition E 3 Energy (ev) Metastable state E 2 Green Stimulated emission Blue Pumping E 1 Ground state ØCr 3+ atom absorb green and blue bands of wave length from xenon flash lamp & excited to E 3 & E 3’ respectively ØRadiative transitions from E 2 to E 1 emits Red photon with peak near 6943 A 0.

He-Ne Laser • First gas laser was developed in 1961 by Ali Javan and his coworkers
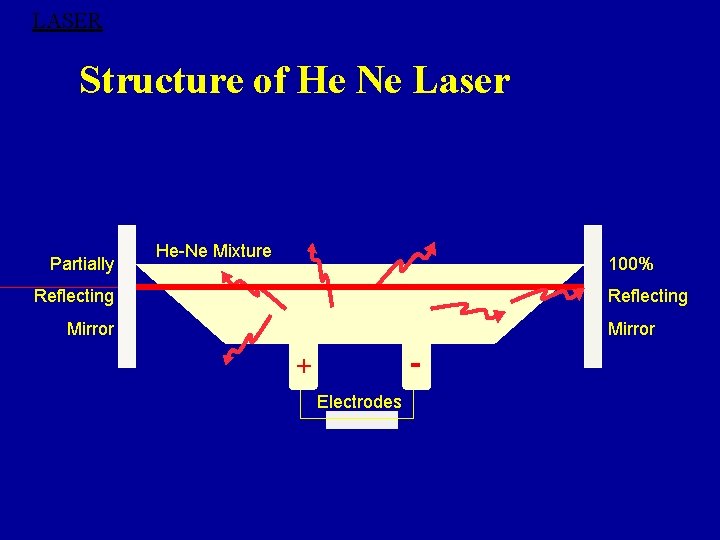
LASER Structure of He Ne Laser Partially He-Ne Mixture 100% Reflecting Mirror - + Electrodes

Construction: Ø Discharge tube of about 50 cm long, 1 cm in diameter, filled with a mixture of He & Ne gases in the ratio of 10: 1 which is active medium. Ø Ne-atoms are active centers- have energy levels suitable for laser transitions Ø He-atoms is efficient to excite the Ne-atoms. Ø Energy transfer between He and Ne-atom takes place through collision and the Ne atoms get excited.
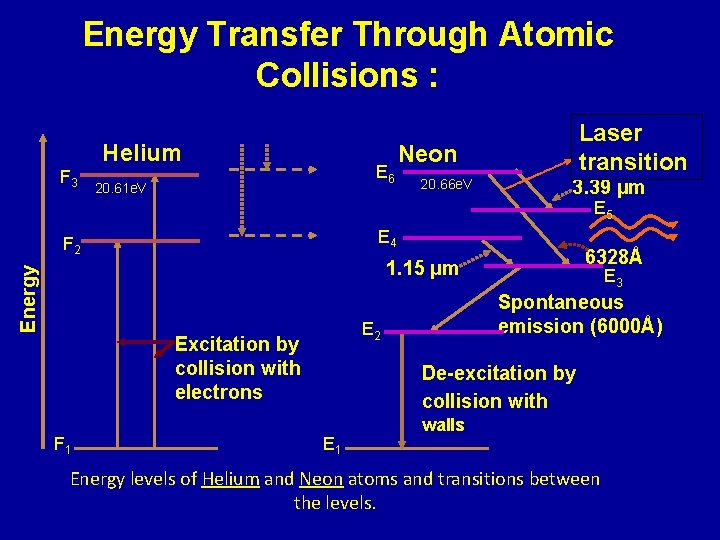
Energy Transfer Through Atomic Collisions : Helium F 3 E 6 20. 61 e. V Laser transition Neon 20. 66 e. V 3. 39 µm E 5 E 4 F 2 6328Å Energy 1. 15 µm F 1 Spontaneous emission (6000Å) E 2 Excitation by collision with electrons E 3 De-excitation by collision with E 1 walls Energy levels of Helium and Neon atoms and transitions between the levels.

Working: ØHe atoms are excited to levels F 2 & F 3 – metastable levels. ØE 4 & E 6 levels in Ne are metastable states accumulation of atoms takes place in level E 6 and E 4 ØPopulation inversion can be achieved between: E 6 a nd E 5, E 6 and E 3 levels E 4 and E 3 levels Ø E 6 E 3 transitions; laser beam of red colour at 632. 8 nm (6328 A) Ø E 4 E 3 transitions; laser beam at wavelength of 1150 nm(11500 A ) ØE 6 E 5 transitions; laser beam in IR region at 3390 nm(33900 A)
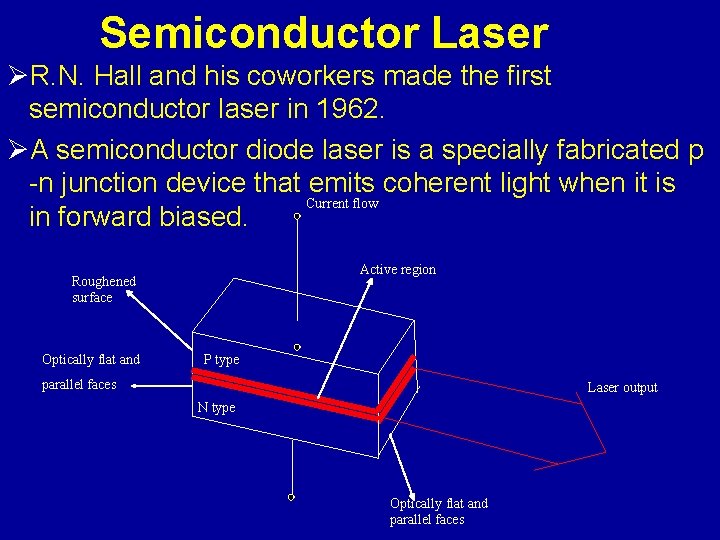
Semiconductor Laser ØR. N. Hall and his coworkers made the first semiconductor laser in 1962. ØA semiconductor diode laser is a specially fabricated p -n junction device that emits coherent light when it is Current flow in forward biased. Active region Roughened surface Optically flat and P type parallel faces Laser output N type Optically flat and parallel faces

Working: Ø Semiconductor laser is heavily doped PN junction diode Ø When diode is forward biased electron from C. B. recombine with holes in V. B. Ø During recombination it emits energy in the form of light and junction acts as laser which emites coherence beam of laser light Ø At low FB. Junction acts as a LED which emits incoherent light Ø A Ga. As. P laser emits light of wavelength 9000 Ao in IR region (red).

Energy band structure of a semiconductor diode ØWhen the junction is F. B, electrons and holes are injected across the junction to cause population inversion. Ø Population inversion is created in a very narrow zone called the active region

Applications of Laser Ø Industrial applications Ø Applications in the field of medical science Ø Astronomical and geophysical applications Ø Metrology applications Ø Applications in communication Ø Defence application Ø Environmental monitoring and Scientific Research

ENTERTAINMENT APPLICATION Laser show

DEFENCE APPLICATION Finger print detection Detection of submarine and mines Laser at war time

OPTICAL COMMUNICATION Frequency in the visible region ~ 1014 cycle/sec Frequency in the microwave region ~ 109 cycle/sec i. e. communication capacity: light wave 105 > microwave

SCIENCE AND TECHNOLOGY Laser fusion Laser Eraser Compact Disk (CD)

HOLOGRAPHY DISPLAY HOLOGRAM: EXHIBIT

SECURITY HOLOGRAM An Embedded Hologram™ cannot be removed, erased, duplicated or simulated by photocopying, photography or scanning.
MEDICAL APPLICATION Brain tumor surgery Eye surgery

WAVE OPTICS Interference: Ø Redistribution of light energy due to the superposition of light waves from two or more coherent sources is known as Interference § Constructive Interference § Destructive Interference

Interference in Thin Film Ø Thin Film: A film whose thickness is about the order of one wavelength of visible light i. e. 5500 Ǻ. Ø Range of thickness of thin film is 0. 5 μm to 10 μm Ø If the film of thickness 10μm to 50 μm, it is considered as a thick film.

Plane Parallel Film ØInterference due to reflected light Path difference ∆=2μt cos r 1) For Maxima ( Brightness) 2μt cos r = (2 m+1) λ / 2 2) For Minima ( Darkness) 2μt cos r = m λ

ØInterference due to transmitted light Path difference ∆=2μt cos r 1) For Maxima ( Brightness) 2μt cos r = m λ 2) For Minima ( Darkness) 2μt cos r = (2 m+1) λ / 2
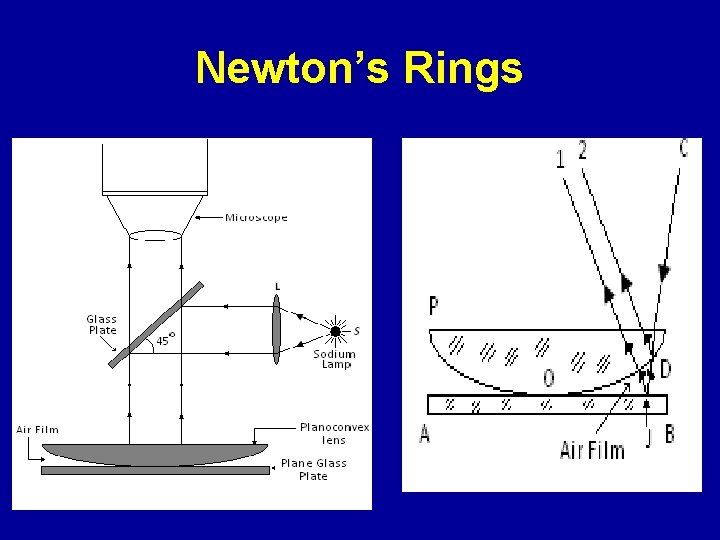
Newton’s Rings

Newton’s Rings For destructive interference For constructive interference R : Radius of Curvature of Plano Convex lens λ : Wavelength of light r : Radii of ring

Salient Features of Newton’s Ring ØCentral spot is dark ØRings are circular ØFringes are localized at the top surface of the film ØLens of large radius of curvature is used ØRings get closer away from the centre ØFor transmitted light central spot is bright ØIllumination with white light results in few coloured rings near the point of contact

Applications ØDetermination of wavelength λ =(D 2 m+p - D 2 m ) /4µR ØDetermination of refractive index of liquid R. I Of liquid = µ = (D 2 m+p - D 2 m )air / (D 2 m+p - D 2 m )liquid ØTesting of lens surface ØTesting of surface finish

Interference in Wedge Shaped Thin Film (Film of Varying Thickness) Microscope S Glass Plate 450 Sodium Lamp Microscope Slides Thin Sheet / Spacer Air Wedge Typical interference fringes pattern

Salient Features Ø Fringes are bright and dark for monochromatic light ØFringes are coloured for white light Ø Fringes are equidistant β=λ/2μθ ØFringe at the apex is dark (t=0) Δt= 2μt-λ/2 =λ/2 ØFringes are straight and parallel ØWedge angle, θ= N λ / 2(x 2 -x 1) ØSpacer thickness, t= LNλ /2(x 2 -x 1) = Lλ /2 β ØFringes are localised

Anti-Reflection Coating ØA thin transparent film coated on a surface in order to suppress the reflection from its surface is called as Anti –reflection film coating.

Conditions for antireflection coating 1)Amplitude condition : It requires two reflected waves 1 & 2 should have equal amplitudes. 2) Phase condition: The waves reflected from the top & bottom surface (ray 1&ray 2) of the thin film should be 180º out of phase with each other.

THANK YOU
- Slides: 63