Behavior of Gases Kinetic Molecular Theory KMT l

Behavior of Gases

Kinetic Molecular Theory (KMT) l Theory related to motion of particles. l Particles of an ideal gas… l have elastic collisions. l are in constant, random, straight-line motion. l have an avg. KE directly related to Kelvin temperature. l have no volume. l don’t attract or repel each other.

Real Gases l But…. particles in a REAL gas… l Have their own volume l Although it’s really small relative to total volume of gas l Can attract each other l Weakly, usually Van Der Waals force only!!

When is a Gas Most Ideal? l Thus, gas behavior is most IDEAL… l When gas particles are tiny, have weak attractive forces, are far apart and moving fast. l l at low pressures at high temperatures when very small in size when nonpolar PLIGHT

Characteristics of Gases l Gases expand to fill any container uniformly. l Are in random motion, have no attraction l Gases have very low densities. l Particles have no volume = lots of empty space
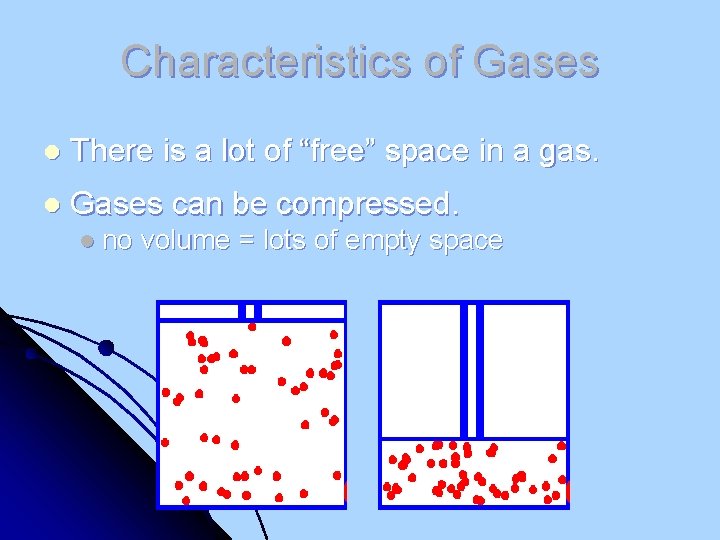
Characteristics of Gases l There is a lot of “free” space in a gas. l Gases can be compressed. l no volume = lots of empty space

Gas Pressure Gas pressure is a gauge of the number and force of collisions between gas particles and the walls of container The SI unit for pressure is the pascal (Pa), but other pressure terms include atmospheres (atms), millimeters of mercury (mm. Hg), and torr. Standard pressure in every unit for pressure: 1 Atm = 101. 325 k. Pa = 101, 325 Pa = 760 torr =760 mm. Hg = 14. 7 lb/in 2

Factors Affecting Gas Pressure l Number of Moles (Amount of gas) l As # of particles increase, the number of collisions with the container wall increases. l Draw an Example:

Factors Affecting Gas Pressure l Volume l Smaller the volume, the greater the pressure exerted on the container. l Draw an Example:

Factors Affecting Gas Pressure l Temperature l As temp. increases, KE increases, this increases frequency of collisions making pressure increase. l Draw an Example:

Measuring Gas Pressure l Barometer l measures atmospheric pressure l (developed by Torricelli in 1643) Note: The weight of the column of mercury is equal to the force of the atmospheric pressure. It the column was water the column would have to be 34 feet tall!! Mercury Barometer


Atmospheric Pressure "Atmospheric Pressure" is defined as the force per unit area exerted against a surface by the weight of the air above that surface. The pressure at point "X" increases as the weight of the air above it increases. The same can be said about decreasing pressure. In the mountains there is less air above you so less pressure. Since gases are compressible, this creates an environment in the mountains that has less air per unit volume. It will be harder to breathe in the mountains because there is actually less O 2 per volume compared to at sea level.

Hg rises in tube until force of Hg (down) balances the force of atmosphere (pushing up). (Just like a straw in soft drink) World’s Longest Vertical Straw: https: //youtu. be/HUm. Zrti. XDik Column height measures pressure of atmosphere 1 standard atmosphere (atm) = 760 mm Hg (or torr) So how dense is Mercury? Watch and find out. https: //www. youtube. com/watch? feature=player_emb edded&v=Rm 5 D 47 n. G 9 k 4

l Standard Pressure (Sea Level) 101. 3 k. Pa (kilopascal) 1 atm 760 mm Hg (also called torr) You may be asked to convert between units of pressure!
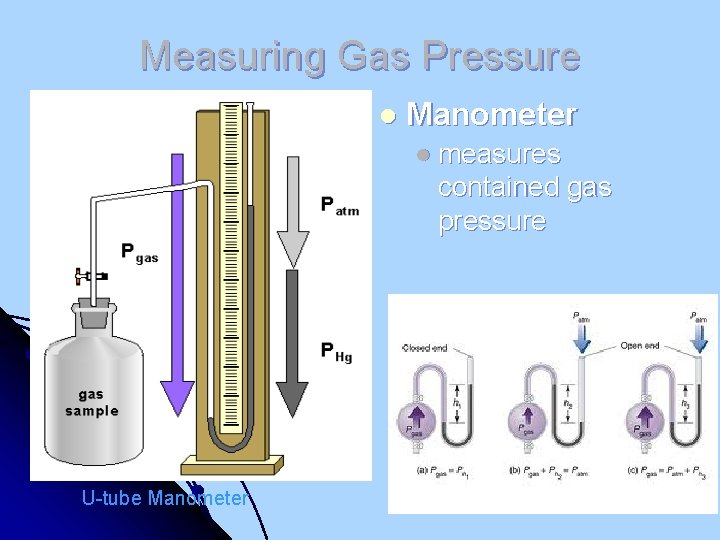
Measuring Gas Pressure l Manometer l measures contained gas pressure U-tube Manometer

http: //www. kentchemistry. com/links/Gas. Laws/man. Bar. htm

The Gas Laws Working with Formulas

Gas properties can be modeled using math. Model depends on: l V = volume of the gas (L) l T = temperature (K) l n = amount (moles) l P = pressure (atmospheres or k. Pa)

STP Standard Temperature & Pressure 0°C 273 K -OR- 1 atm 101. 325 k. Pa 760 mm. Hg
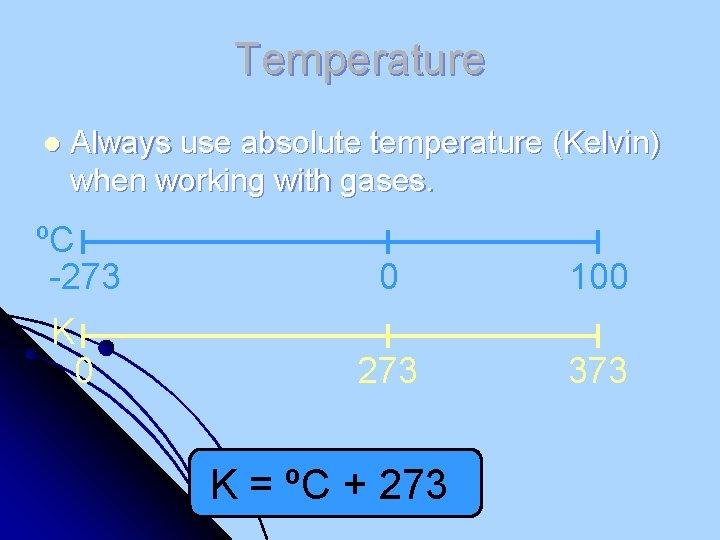
Temperature l Always use absolute temperature (Kelvin) when working with gases. ºC -273 0 100 K 0 273 373 K = ºC + 273

Boyle’s Law l Robert Boyle (1627 -1691). Son of Earl of Cork, Ireland. Pressure and volume of a gas are inversely related l at constant mass & temp PV = k P V Cartesian Diver: https: //www. youtube. com/watch? feature=player_embedded&v=3 yv. SQ u. TP 4 T 4
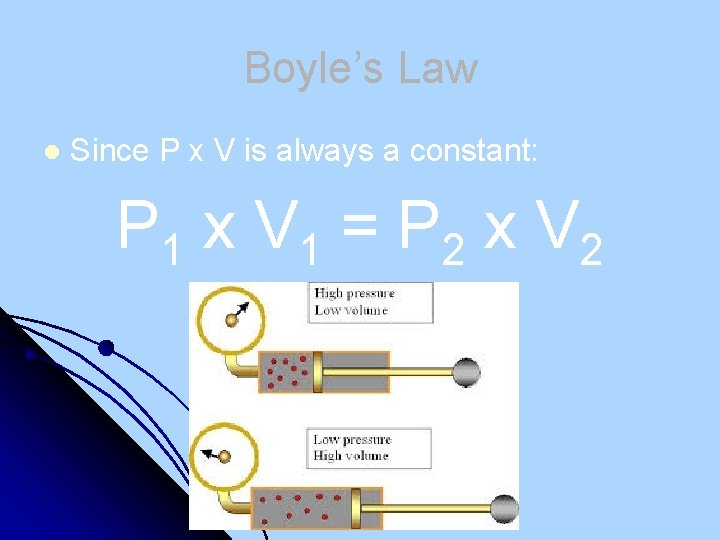
Boyle’s Law l Since P x V is always a constant: P 1 x V 1 = P 2 x V 2

Ex: l Consider a 3. 0 L sample of gaseous SO 2 at a pressure of 1. 0 atm. If the pressure is changed to 1. 5 atm at a constant temperature, what will be the new volume of the gas?
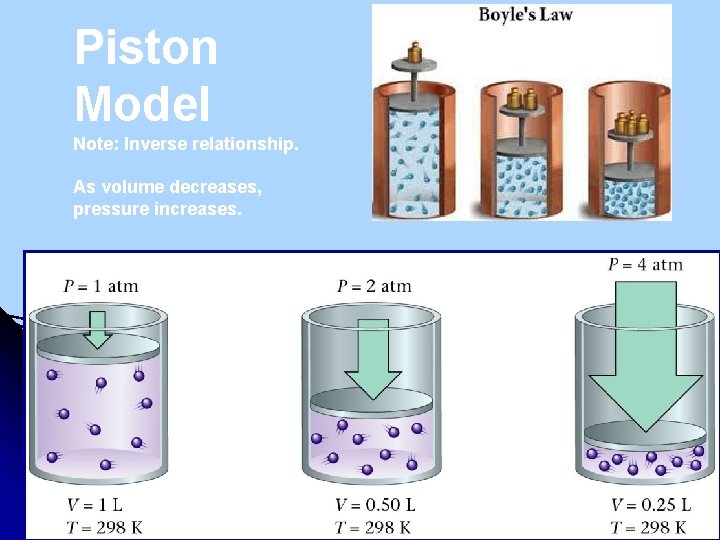
Piston Model Note: Inverse relationship. As volume decreases, pressure increases.
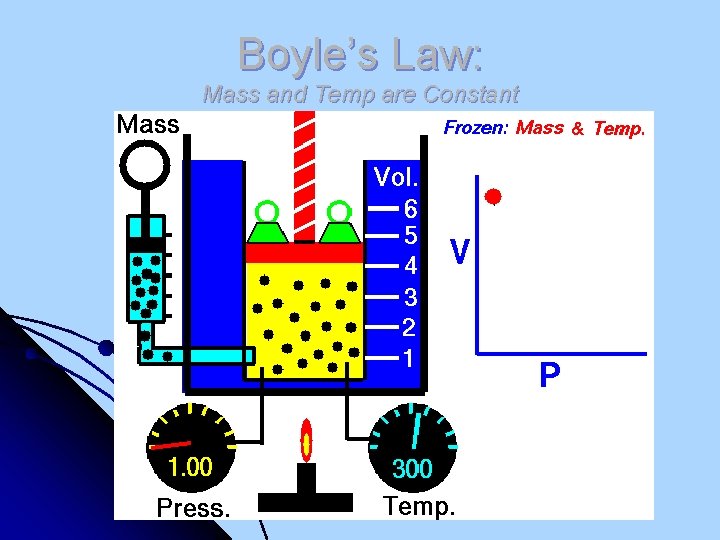
Boyle’s Law: Mass and Temp are Constant

Boyle’s Law Balloon: https: //youtu. be/b. W 0 j 936 U 4 EU Pierre and Marie: Peeps in a vacuum https: //www. youtube. com/watch? v=lf. NJJ Ed. Kg. LU Cartesian Diver https: //www. youtube. com/watch? v=s. NOX Fi. J 4 IDU Balloon in a Vacuum

Charles’ Law Jacques Charles (1746 -1823). Isolated boron and studied gases. Balloonist. l Volume and absolute temp. (K) of a gas are directly related l at constant mass & pressure V T

l Think of it this way. l As temperature of the gas increases, gas molecules begin to move around more quickly and hit the walls of container with more force, thus the volume will increase. l Keep in mind that you must use only the Kelvin temperature scale when working with temperature in all gas law formulas!

Charles’ Law l Since V/T is always a constant: V 1 = V 2 T 1 T 2 https: //www. youtube. com/watch? feature=player_embedded&v=XHi. YKf. Am. TMc Can Crush https: //www. youtube. com/watch? v=vcsx. B 5 d. KJMg

Ex: l A sample of gas at 15ºC and 1 atm has a volume of 2. 50 L. What volume will this gas occupy at 30ºC and 1 atm? l
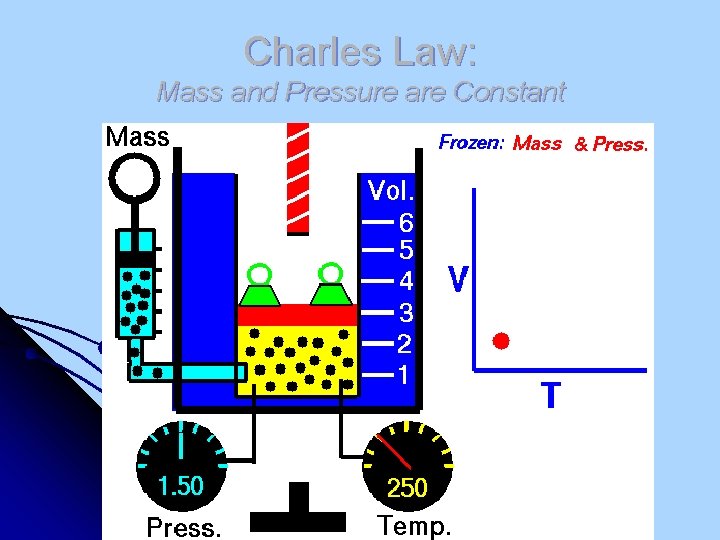
Charles Law: Mass and Pressure are Constant

Charles’ Law Pour Liquid Nitrogen on Balloon!! Bill Bye Vaccuum: https: //www. youtube. com/watch? v=lf. NJJEd. Kg. LU Can Crush: https: //www. youtube. com/watch? v=c 5_ho 2 sc 0 fc Big Can Crush https: //www. youtube. com/watch? feature=player_embedded&v=Uy-SN 5 j 1 ogk http: //group. chem. iastate. edu/Greenbowe/sections/projectfolder/flashfiles/gaslaw/charles_law. html http: //www. pinnaclescience. com/demo. htm

Gay-Lussac’s Law l Pressure and absolute temp. (K) of a gas are directly related l at constant mass & volume P T

Gay – Lussac’s Law l Since P/T is always a constant: P 1 = P 2 T 1 T 2

Ex: Find the final pressure of gas at 150. K, if the pressure of gas is 210. k. Pa at 120. K if the volume remains constant l

Review of 3 Gas Laws Summary of Changing Variables http: //www. mhhe. com/physsci/chemistry/essentialchemistry/flash/gasesv 6. swf

Combined Gas Law (on Ref Table) The good news is that you don’t have to remember all three gas laws! We can combine them into a single equation. If you should only need one We can combine them into a single equation. of the other gas laws, you can cover up the item that is constant and you will get that gas law! P 1 V 1 T 1 = P 2 V 2 T 2 P 1 V 1 T 2 = P 2 V 2 T 1
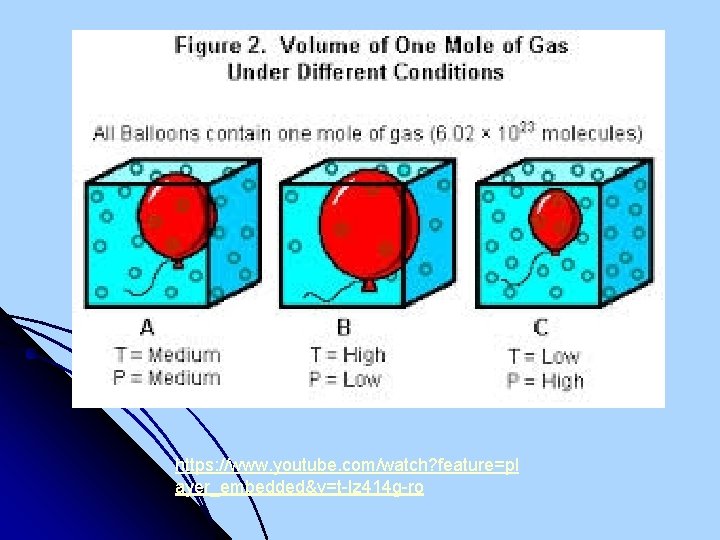
https: //www. youtube. com/watch? feature=pl ayer_embedded&v=t-Iz 414 g-ro

Example • A sample of He (g) has a volume of 0. 180 L, a pressure of 0. 800 atm and a temperature of 29°C. What is the new temperature (°C) of the gas at a volume of. 090 L and a pressure of 3. 20 atm? Set up Data Table P 1 = 0. 800 atm V 1 =. 180 L T 1 = 302 K P 2 = 3. 20 atm V 2=. 090 L T 2 = ? ?
- Slides: 40