BEHAVIOR OF GASES Chapter 12 Properties of Gases

BEHAVIOR OF GASES Chapter 12

Properties of Gases Gas properties can be modeled using math. Model depends on— • V = volume of the gas (L) • T = temperature (Kelvin) • P = pressure (atmospheres)

Volume of Gas • In a smaller container, molecules have less room to move. • Hit the sides of the container more often. • As volume decreases, pressure increases. (think of a syringe)
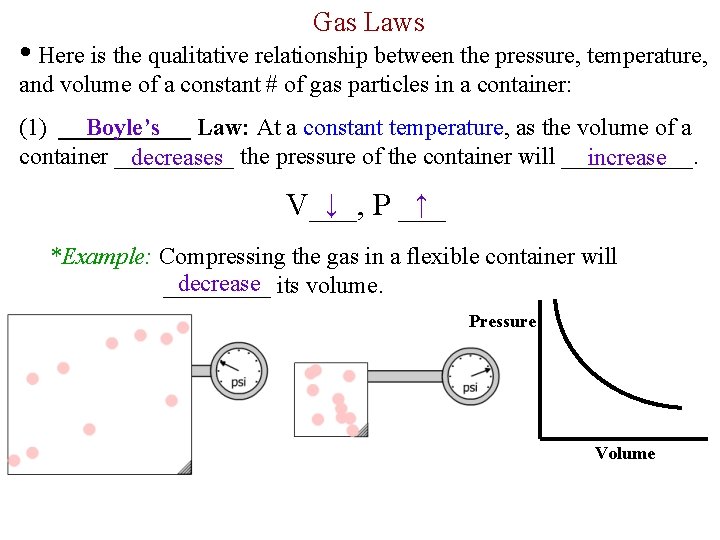
Gas Laws • Here is the qualitative relationship between the pressure, temperature, and volume of a constant # of gas particles in a container: Boyle’s (1) ______ Law: At a constant temperature, as the volume of a container _____ decreases the pressure of the container will ______. increase ↓ P ___ ↑ V___, *Example: Compressing the gas in a flexible container will decrease its volume. _____ Pressure Volume

Boyle’s Law If Temperature is constant, then P 1 V 1 = P 2 V 2 This means that as Pressure goes up as Volume goes down. Pressure and Volume are indirectly related How you doin’? <3 Robert Boyle (1627 -1691). Son of Early of Cork, Ireland.

Boyle’s Law A bicycle pump is a good example of Boyle’s law. As the volume of the air trapped in the pump is reduced, its pressure goes up, and air is forced into the tire.

Boyle’s Law P 1 V 1 = P 2 V 2 P 1 = initial pressure V 1 = initial volume P 2 = final pressure V 2 = final volume STP = standard temperature (273 Kelvin) and pressure (1 atmosphere) Pressure can be measured in atmospheres (atm), torrs (torr), millimeters mercury (mm. Hg) or kilopascals (kpa) Conversions: 1 atm = 760 torr = 760 mm. Hg = 101. 325 kpa

Boyle’s Law Practice Problems 1. If we have 4 L of methane gas at a pressure of 1. 50 x 103 mm. Hg. What will the pressure of the gas be if the volume is decreased to 2. 5 L? P 1 = 1. 50 x 103 mm. Hg Plug the #’s (1. 05 x 103) (4) = (P 2) (2. 5) V 1 = 4 liters into the P 2 = equation and ? solve for P 2. V 2 = 2. 5 liters (1. 50 x 103) (4) = P 2 2. 5 2400 mm. Hg = P 2

Boyle’s Law Practice Problems 2. You have a car with an internal volume of 12, 000 L. If you drive your car into the river and it implodes, what will be the volume of the gas when the pressure goes from 1. 0 atm to 1. 4 atm? P 1 = V 1 = P 2 = V 2 = 1 atm 12, 000 L 1. 4 atm ? Plug the #’s into the equation and solve for V 2. (1) (12, 000) = V 2 1. 4 8571. 43 L = V 2 (1) (12, 000) = (1. 4) (V 2)

Boyle’s Law Practice Problems v If P 1 and P 2 are given, they must both be in the same unit. If they are not, you must make a conversion. Usually the conversion is to atm’s since it is the measure of standard pressure. Make the following conversions using: 1 atm = 760 torr = 760 mm. Hg = 101. 325 kpa Practice Problems: (1) Convert 6. 5 atm to torr 760 torr x = 4940 torr 1 atm (2) Convert 98 torr to mm. Hg 760 mm. Hg 98 torr x = 760 torr (3) Convert 177 mm. Hg to atm 6. 5 atm 177 mm. Hg x 1 atm 760 mm. Hg = 98 mm. Hg . 23 atm

Gas Laws (continued) Charles’s Law: At a constant pressure, as the temperature of a (2) ______ container _____ increases the volume of the container will ______. increase ↑ V ___ ↑ T___, inflate *Examples: Heating a balloon will cause it to ______. Taking a balloon outside on a cold winter day will cause shrink it to _______. • If you could keep a gas from condensing, you could cool it off to absolute zero and the zero volume of the gas would be _____! Volume Temperature (K)

Charles’s Law Hey baby! You need a date? If Pressure is constant, then V 1 T 2 = V 2 T 1 This means as Volume goes up so does Temperature. This means Volume and Temperature are directly related. Jacques Charles (17461823). Isolated boron and studied gases. Balloonist.

Charles’s original balloon Modern long-distance balloon

Charles’s Law

Charles’s Law V 1 T 2 = V 2 T 1 V 1 = initial volume T 2 = final temperature V 2 = final volume T 1 = initial temperature STP = standard temperature (273 Kelvin) and pressure (1 atmosphere) Conversions: Kelvin = degrees Celsius + 273

Charles’s Law Practice Problems 1. If we have 2 L of methane gas at a temperature of 40 degrees Celsius, what will the volume of the gas be if we heat the gas to 80 degrees Celsius? V 1 = 2 liters Plug the #’s (2)(353) = (V ) (313) 2 T 2 = 80 C + 273 = 353 K into the V 2 = equation and ? solve for V 2. T 1 = 40 C + 273 = 313 K (2) (353) = V 2 313 2. 26 L = V 2

Charles’s Law Practice Problems 1. If you have 45 L of helium in a balloon at 25 C and you increase the temperature of the balloon to 55 C, what will the new volume of the balloon be? V 1 = T 2 = V 2 = T 1 = 45 liters 55 C + 273 = 328 K ? 25 C + 273 = 298 K Plug the #’s into the equation and solve for V 2. (45) (328) = V 2 298 49. 53 L = V 2 (45)(328) = (V 2) (298)

Gas Laws (continued) (3) Guy-Lussac’s ______ Law: At a constant volume, as the temperature of a container _____ increases the pressure of the container will ______. increase P 1 T 2 = P 2 T 1 ↑ P ___ ↑ T___, *Example: Heating a rigid container causes the gas inside faster which causes _____ more pressure. to move _____ Be careful! Too much heat will make it explode! Pressure Temperature (K)

The Combined Gas Law

Combining the gas laws • So far we have seen three gas laws: Bachelor #1 Bachelor #2 Check me out! Pick me baby! Robert Boyle P 1 V 1 = P 2 V 2 Jacques Charles V 1 T 2 = V 2 T 1 Bachelor #3 I’m the hottest pick me! Joseph Louis Gay-Lussac P 1 T 2 = P 2 T 1 These are all subsets of a more encompassing law: the combined gas law P 1 V 1 T 2 = P 2 V 2 T 1

The Combined Gas Law • This equation combines all of the previous three laws into one convenient form. Boyles Law: Pressure and volume Guy-Lussac’s Law: Pressure and temperature Combined Gas Law Charles’s Law: Volume and temperature • Using the Combined Gas Law requires you to have the P 1 x V 1 x T 2 = P 2 x V 2 x T 1 temperature in _______ Kelvin units. The pressure and volume units can be anything as long as (initial conditions) = (final conditions) the initial and final units are the _____. same ______

Standard Temperature and Pressure: (STP) • Often the volume of a gas is needed at “standard conditions. ” For 273 scientists, this means “STP”. Standard temperature is ______K, 1 and standard pressure is ______ atmosphere (atm) 1 atmosphere (atm) = 760 Torr = 760 mm. Hg = 101. 3 k. Pa = 14. 7 lbs/in 2 (psi) Practice Problems: 1) 80. 0 m. L of helium is in a balloon at 25˚C. What will the new volume of the balloon be if the temp. is raised to 100˚C? (Since pressure is not mentioned, it can be assumed that it was constant. You can throw it out of our equation. ) P 1 = ______ P 2 = ______ 80. 0 m. L V 2= ______ ? ? ? V 1= ______ 298 K TK 2= ______ 373 K TK 1= ______ Plug the #’s (80. 0) (373) (V 2) (298) = into the equation and V 2 = 100 m. L solve for V 2.

Practice Problems (continued): 2) A rigid steel container is filled with neon under a pressure of 760 mm Hg and a temperature of 325 K. If the temperature is reduced to standard temperature, what will the new pressure be? P 1 = 760 ______ mm P 2 = ______ ? ? ? V 1= ______ V 2= ______ 325 K TK 2= ______ 273 K TK 1= ______ Plug the #’s (760) (273) = (P 2) (325) into the equation and P 2 = 638 mm Hg solve for P 2. Volume is not mentioned, so assume it is constant. 3) A balloon at a pressure of 4. 5 atmospheres, 300 K, and a volume of 35. 0 liters is changed to STP conditions. What will the new volume of the balloon become? P 1 = ______ 4. 5 atm P 2 = ______ 1 atm 35. 0 L V 2= ______ ? ? ? V 1= ______ 300 K TK 2= ______ 273 K TK 1= ______ Plug the #’s (4. 5)(35. 0) (273) = (300) (1)(V 2) into the equation and solve for V 2 = 143 L


Dalton’s Law of Partial Pressure • • What happens to the pressure of a gas as we mix different gases in the container? total gas The ______ sum of each individual gas pressure equals the _______ pressure of the container. P(total)= P 1+P 2+P 3…


Dalton’s Law of Partial Pressures 1) A container has oxygen, nitrogen, and helium in it. The total pressure of the container is 2. 4 atmospheres. If all the partial pressures are equal to one another, what ate the partial pressures of each gas? P total = 2. 4 atm Total number of gases = 3 Pgas = 2. 4 atm ÷ 3 = 0. 8 atm 27

Dalton’s Law of Partial Pressures 2) Two flasks are connected with a rubber hose. The first flask contains N 2 at a pressure of 0. 75 atm. , and the second flask contains O 2 at a pressure of 1. 25 atm. When the two flasks are opened and mix, what will the pressure be in the resulting mixture? P 1 = 0. 75 atm P 2 = 1. 25 atm Ptotal = 0. 75 atm + 1. 25 atm = 2. 0 atm 28

Dalton’s Law of Partial Pressures 3) A mixture of neon and argon gases exerts a total pressure of 2. 39 atm. The partial pressure of the neon alone is 1. 84 atm. What is the partial pressure of the argon? Ptotal = 2. 39 atm P 1 = 1. 84 atm P 2 = 2. 39 atm – 1. 84 atm = 0. 55 atm 29

DALTON’S LAW OF PARTIAL PRESSURE: WATER DISPLACEMENT METHOD • SAMPLE PROBLEM In a laboratory, oxygen gas was collected by water displacement at an atmospheric pressure of 726 torr and a temperature of 22°C. Calculate the partial pressure of dry oxygen. Water vapor pressure is 2. 6 k. Pa (from table in reading packet). Convert 2. 6 k. Pa to torr to solve.

DALTON’S LAW OF PARTIAL PRESSURE: WATER DISPLACEMENT METHOD • SOLUTION to Sample Problem PTOTAL = 726 torr PWATER= 19. 5 torr (22 °C) POXYGEN = ? PTOTAL = POXYGEN + PWATER POXYGEN = PTOTAL – PWATER = 726 torr – 19. 5 torr POXYGEN = 706. 5 torr

Dalton’s Law of Partial Pressures 3) A mixture of helium, neon and argon gases exerts a total pressure of 2. 39 atm. What is the partial pressure of the argon if it makes up 40% of the mixutre? Ppartial pressure = Ptotal x (%of mixture) Ptotal = 2. 39 atm % = 0. 40 Ppartial pressure = 2. 39 atm x 0. 40 = 0. 956 atm 32

Avogadro’s Hypothesis • equal volumes of gases Avogadro’s hypothesis states that ____ (under the same temp. and pressure conditions) contain _______ equal number of particles. • If containers have the same ____, T ____, P and ___, V then they will # of particles regardless of the _____ have the same ____ of the size gas particle. • You might think that a small gas molecule would take up ______ less doesn’t space than a large gas molecule, but it ______ at the same temperature pressure _________ and _______!!

AVOGADRO’S LAW The mathematical form of Avogadro’s law is: V 1 = V 2 n 1 n 2 SAMPLE PROBLEM 1 A sample of gas with a volume of 9. 20 L is known to contain 1. 225 mol. If the amount of gas is increased to 2. 85 mol, what new volume will result if the pressure and temperature remain constant?

AVOGADRO’S LAW SOLUTION Given: V 1 = 9. 20 L V 2 = ? n 1 = 1. 225 mol n 2 = 2. 85 mol Solution: V 2 = n 2 V 1 = (2. 85 mol) (9. 20 L) = 21. 4 L n 1 (1. 225 mol)

• • The Ideal Gas Law An equation used to calculate the _____ amount of gas in a container (in units of _____. ) moles PV=n. RT Kelvin Liters The units for T= _____, V = _____, n = # of moles R = Ideal Gas Constant • The value of R changes depending on the unit of ______ pressure used in the equation: R = 62. 4 (mm Hg)(L)/(mole)(K) R = 8. 31 (k. Pa)(L)/(mole)(K) R = 0. 0821 (atm. )(L)/(mole)(K) R = 2. 45 (in. Hg)(L)/(mole)(K)

“Ideal” Gases • condense into a Real gases, (like nitrogen), will eventually ______ liquid when the temperature gets too ____ low or the pressure gets too _____. high • If you want a gas to act more ideally, keep the temperature _____ high and the pressure ______. That way, they will act more like an ideal low gas and never have a chance of ________. condensing • The best real gas that acts like an ideal gas is _____. It helium 4 doesn’t condense until the temperature gets to ______K. Real Gas

Ideal Gases vs. Real Gases


Ideal Gas Law SAMPLE PROBLEM 1 What pressure will be exerted by 0. 400 mol of gas in a 5. 00 L container at 17. 0°C? Given: n = 0. 400 mol V= 5. 00 L T= 17. 0 °C + 273 = 290 K Solution: L atm P = n. RT = (0. 400 mol) (0. 0821 mol K) (290 K) = 1. 9 atm V (5. 00 L)

Diffusion vs. Effusion • The spreading out of a gas from _______ high to _____ low concentrations is called diffusion. *Example: ______ Perfume in a room spreads out • tiny _______ hole A gas escaping through a ______ in a container is called effusion. As the size of a molecule _______, the effusion increases speed and diffusion rate _______. . . (inverse relationship. ) decrease Effusion
- Slides: 41