Behandeling van het recidief ovariumcarcinoom Ignace Vergote MD

Behandeling van het recidief ovariumcarcinoom Ignace Vergote MD Ph. D FACS FSPS Chairman, Leuven Cancer Institute and Dept Ob& Gyn , Leuven, European Union
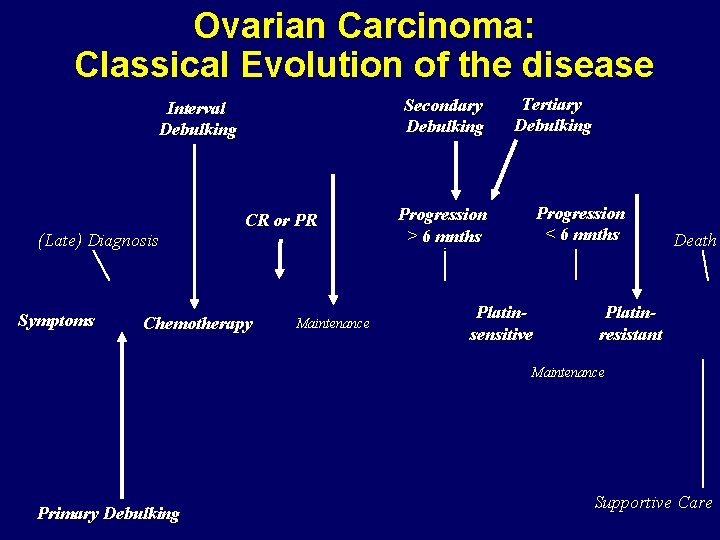
Ovarian Carcinoma: Classical Evolution of the disease Secondary Debulking Interval Debulking (Late) Diagnosis Symptoms CR or PR Chemotherapy Maintenance Tertiary Debulking Progression < 6 mnths Progression > 6 mnths Platinsensitive Death Platinresistant Maintenance Primary Debulking Supportive Care
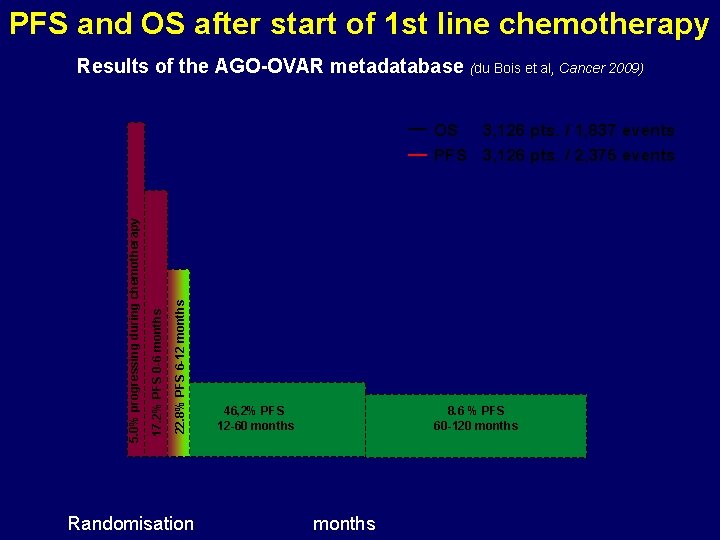
PFS and OS after start of 1 st line chemotherapy Results of the AGO-OVAR metadatabase (du Bois et al, Cancer 2009) 22. 8% PFS 6 -12 months 17. 2% PFS 0 -6 months 5. 0% progressing during chemotherapy OS 3, 126 pts. / 1, 837 events PFS 3, 126 pts. / 2, 375 events Randomisation 46, 2% PFS 12 -60 months 8. 6 % PFS 60 -120 months
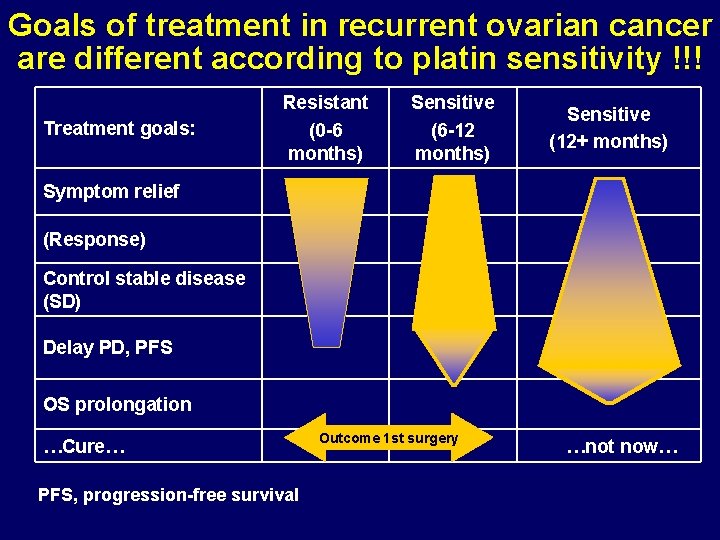
Goals of treatment in recurrent ovarian cancer are different according to platin sensitivity !!! Treatment goals: Resistant (0 -6 months) Sensitive (6 -12 months) Sensitive (12+ months) Symptom relief (Response) Control stable disease (SD) Delay PD, PFS OS prolongation …Cure… PFS, progression-free survival Outcome 1 st surgery …not now…
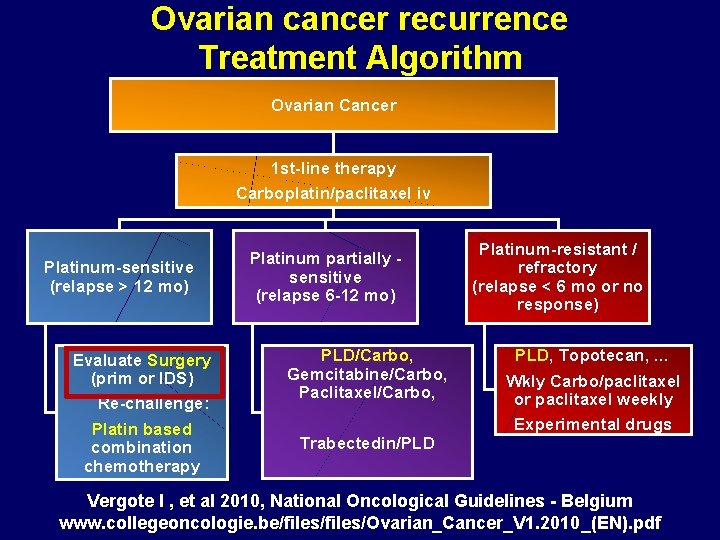
Ovarian cancer recurrence Treatment Algorithm Ovarian Cancer 1 st-line therapy Carboplatin/paclitaxel iv Platinum-sensitive (relapse > 12 mo) Evaluate Surgery (prim or IDS) Re-challenge: Platin based combination chemotherapy Platinum partially sensitive (relapse 6 -12 mo) PLD/Carbo, Gemcitabine/Carbo, Paclitaxel/Carbo, Trabectedin/PLD Platinum-resistant / refractory (relapse < 6 mo or no response) PLD, Topotecan, … Wkly Carbo/paclitaxel or paclitaxel weekly Experimental drugs Vergote I , et al 2010, National Oncological Guidelines - Belgium www. collegeoncologie. be/files/Ovarian_Cancer_V 1. 2010_(EN). pdf
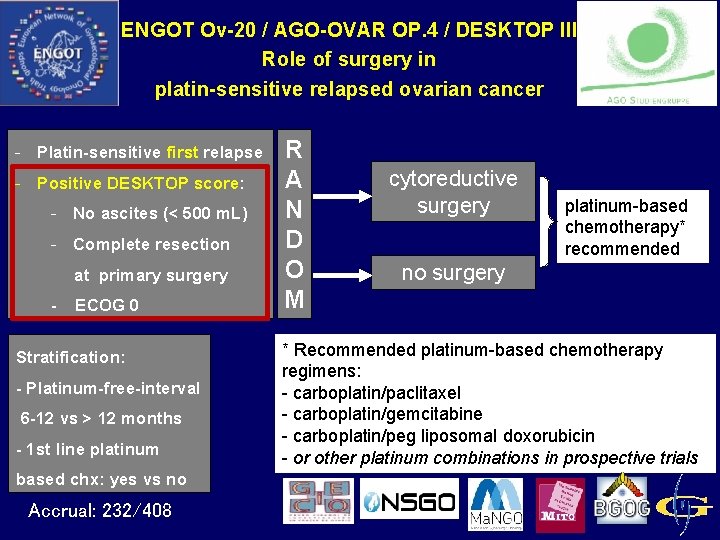
ENGOT Ov-20 / AGO-OVAR OP. 4 / DESKTOP III Role of surgery in platin-sensitive relapsed ovarian cancer - Platin-sensitive first relapse - Positive DESKTOP score: - No ascites (< 500 m. L) - Complete resection at primary surgery - ECOG 0 Stratification: - Platinum-free-interval 6 -12 vs > 12 months - 1 st line platinum based chx: yes vs no Accrual: 232/408 R A N D O M cytoreductive surgery platinum-based chemotherapy* recommended no surgery * Recommended platinum-based chemotherapy regimens: - carboplatin/paclitaxel - carboplatin/gemcitabine - carboplatin/peg liposomal doxorubicin - or other platinum combinations in prospective trials
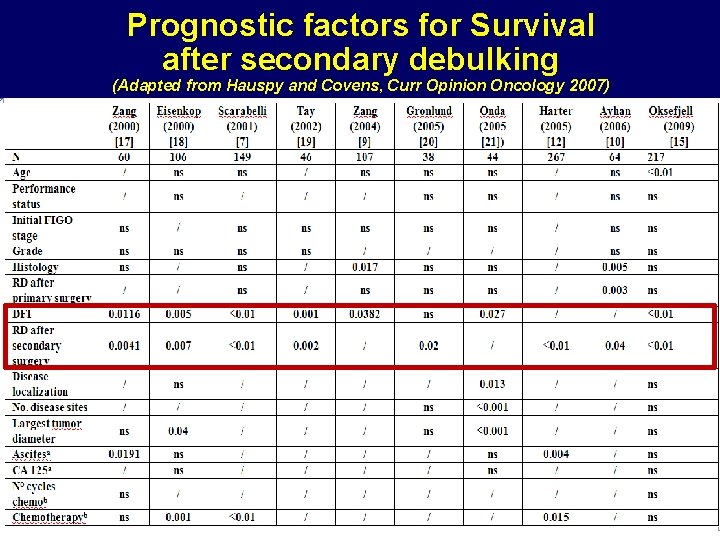
Prognostic factors for Survival after secondary debulking (Adapted from Hauspy and Covens, Curr Opinion Oncology 2007) AIOM 2000
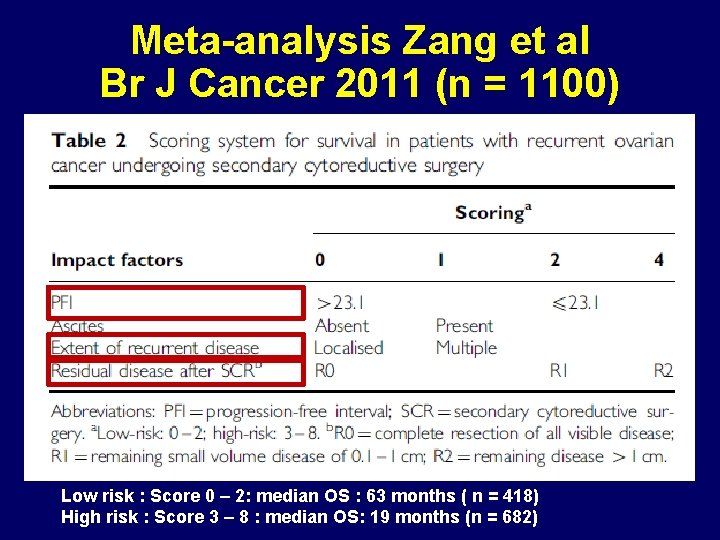
Meta-analysis Zang et al Br J Cancer 2011 (n = 1100) Low risk : Score 0 – 2: median OS : 63 months ( n = 418) High risk : Score 3 – 8 : median OS: 19 months (n = 682)

Secondary Debulking Surgery 1. Besides the AGO score (ECOG, no ascites and no residual tumor after first surgery), time to relapse and spread of the disease should be considered when selecting patients for secondary debulking surgery. 2. Whole body diffusion-weighted MRI (and if not available PET-CT or laparoscopy) have an added value to CT.
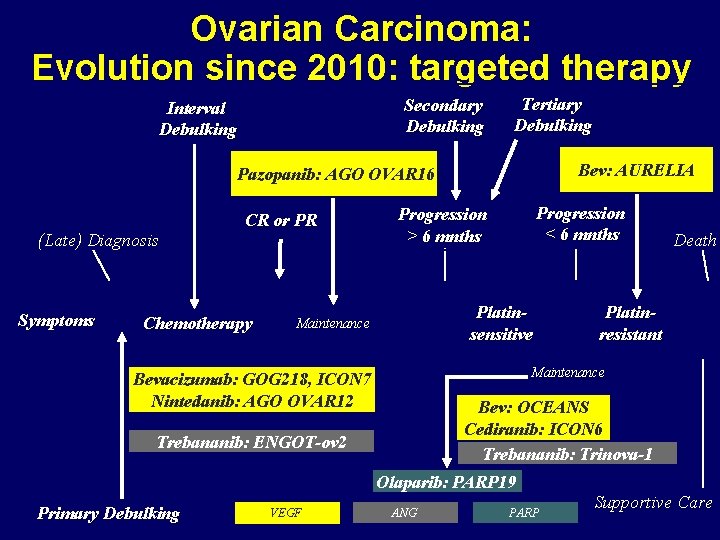
Ovarian Carcinoma: Evolution since 2010: targeted therapy Secondary Debulking Interval Debulking Tertiary Debulking Bev: AURELIA Pazopanib: AGO OVAR 16 (Late) Diagnosis Symptoms CR or PR Chemotherapy Progression < 6 mnths Progression > 6 mnths Platinsensitive Maintenance Death Platinresistant Maintenance Bevacizumab: GOG 218, ICON 7 Nintedanib: AGO OVAR 12 Bev: OCEANS Cediranib: ICON 6 Trebananib: Trinova-1 Trebananib: ENGOT-ov 2 Olaparib: PARP 19 Primary Debulking VEGF ANG PARP Supportive Care
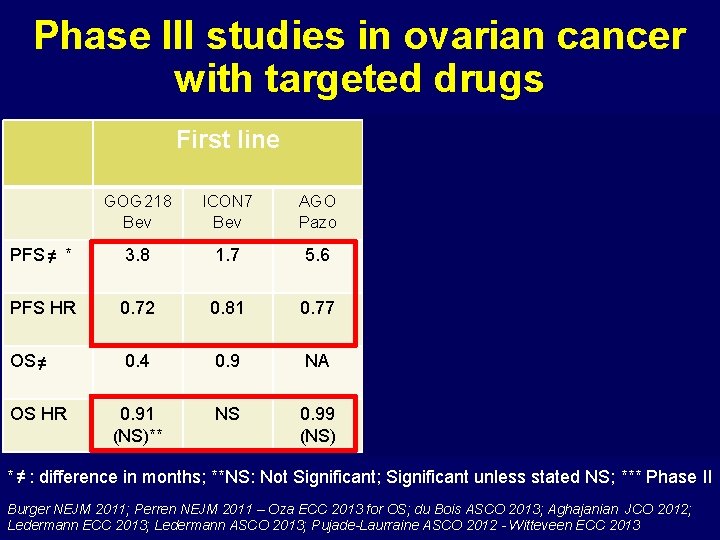
Phase III studies in ovarian cancer with targeted drugs First line Platin-sensitive Plresist GOG 218 Bev ICON 7 Bev AGO Pazo OCEANS Bev ICON 6 Ced PARP* Ola AURELIA Bev PFS ≠ * 3. 8 1. 7 5. 6 4. 0 3. 1 4. 0 3. 3 PFS HR 0. 72 0. 81 0. 77 0. 84 0. 57 0. 35 0. 48 OS ≠ 0. 4 0. 9 NA - 1. 8 2. 7 2. 0 3. 3 0. 91 (NS)** NS 0. 99 (NS) 1. 03 (NS) 0. 70 0. 88 (NS) 0. 85 (NS) OS HR * ≠ : difference in months; **NS: Not Significant; Significant unless stated NS; *** Phase II Burger NEJM 2011; Perren NEJM 2011 – Oza ECC 2013 for OS; du Bois ASCO 2013; Aghajanian JCO 2012; Ledermann ECC 2013; Ledermann ASCO 2013; Pujade-Laurraine ASCO 2012 - Witteveen ECC 2013
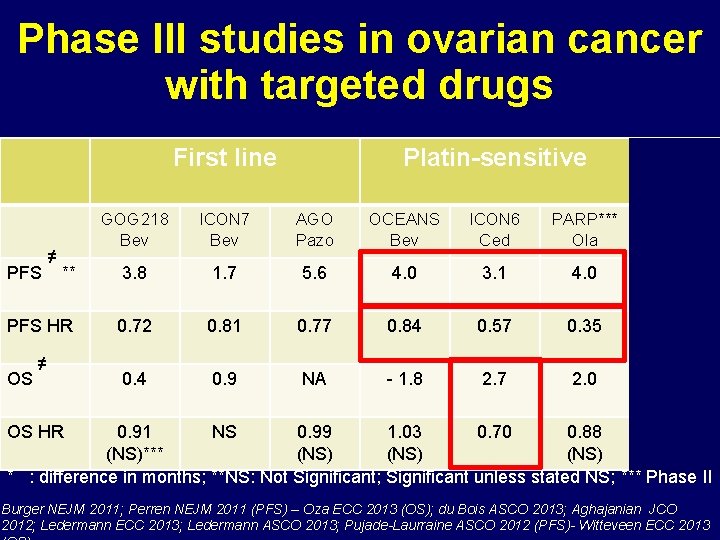
Phase III studies in ovarian cancer with targeted drugs First line Platin-sensitive Plresist GOG 218 Bev ICON 7 Bev AGO Pazo OCEANS Bev ICON 6 Ced PARP*** Ola AURELIA Bev ** 3. 8 1. 7 5. 6 4. 0 3. 1 4. 0 3. 3 PFS HR 0. 72 0. 81 0. 77 0. 84 0. 57 0. 35 0. 48 0. 4 0. 9 NA - 1. 8 2. 7 2. 0 3. 3 PFS OS ≠ ≠ OS HR 0. 91 NS 0. 99 1. 03 0. 70 0. 88 0. 85 (NS)*** (NS) * : difference in months; **NS: Not Significant; Significant unless stated NS; *** Phase II Burger NEJM 2011; Perren NEJM 2011 (PFS) – Oza ECC 2013 (OS); du Bois ASCO 2013; Aghajanian JCO 2012; Ledermann ECC 2013; Ledermann ASCO 2013; Pujade-Laurraine ASCO 2012 (PFS)- Witteveen ECC 2013
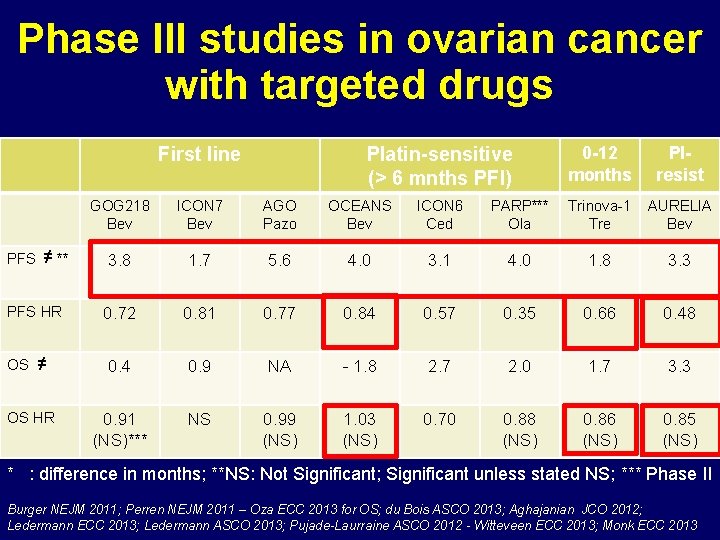
Phase III studies in ovarian cancer with targeted drugs First line PFS ≠ ** PFS HR OS ≠ OS HR Platin-sensitive (> 6 mnths PFI) 0 -12 months Plresist GOG 218 Bev ICON 7 Bev AGO Pazo OCEANS Bev ICON 6 Ced PARP*** Ola Trinova-1 Tre AURELIA Bev 3. 8 1. 7 5. 6 4. 0 3. 1 4. 0 1. 8 3. 3 0. 72 0. 81 0. 77 0. 84 0. 57 0. 35 0. 66 0. 48 0. 4 0. 9 NA - 1. 8 2. 7 2. 0 1. 7 3. 3 0. 91 (NS)*** NS 0. 99 (NS) 1. 03 (NS) 0. 70 0. 88 (NS) 0. 86 (NS) 0. 85 (NS) * : difference in months; **NS: Not Significant; Significant unless stated NS; *** Phase II Burger NEJM 2011; Perren NEJM 2011 – Oza ECC 2013 for OS; du Bois ASCO 2013; Aghajanian JCO 2012; Ledermann ECC 2013; Ledermann ASCO 2013; Pujade-Laurraine ASCO 2012 - Witteveen ECC 2013; Monk ECC 2013
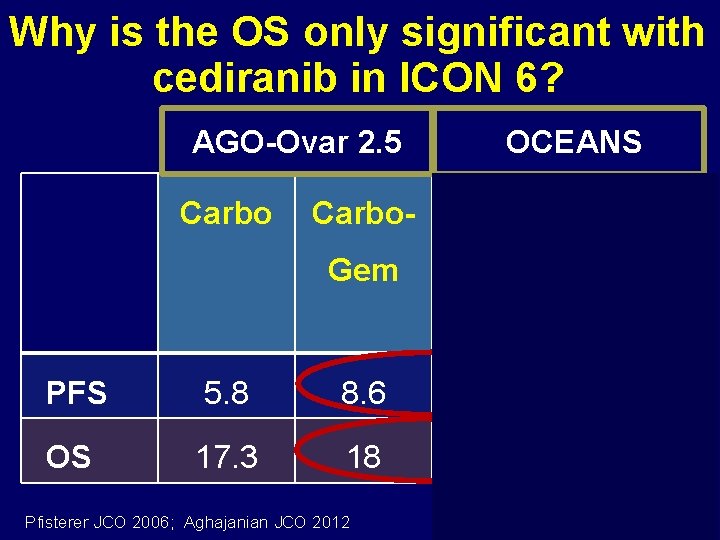
Why is the OS only significant with cediranib in ICON 6? AGO-Ovar 2. 5 Carbo OCEANS Carbo- Gem Gem + Bev PFS 5. 8 8. 6 8. 4 12. 4 OS 17. 3 18 35. 2 33. 3 Pfisterer JCO 2006; Aghajanian JCO 2012
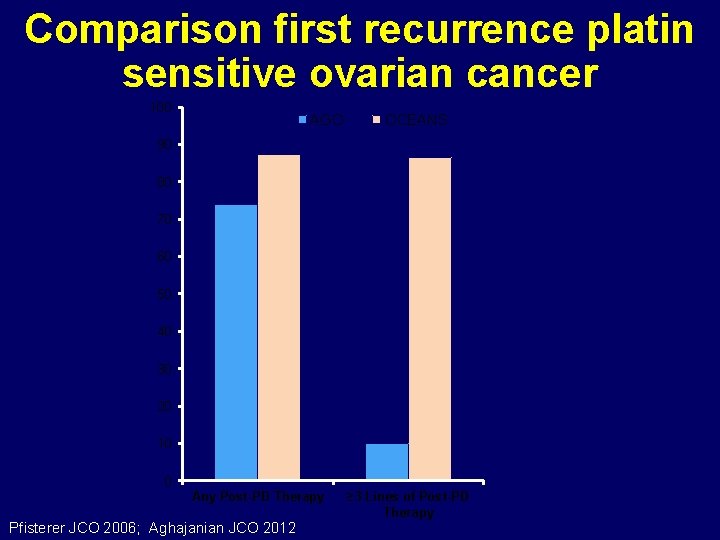
Comparison first recurrence platin sensitive ovarian cancer 100 AGO OCEANS 90 80 70 60 50 40 30 20 10 0 Any Post-PD Therapy Pfisterer JCO 2006; Aghajanian JCO 2012 ≥ 3 Lines of Post-PD Therapy
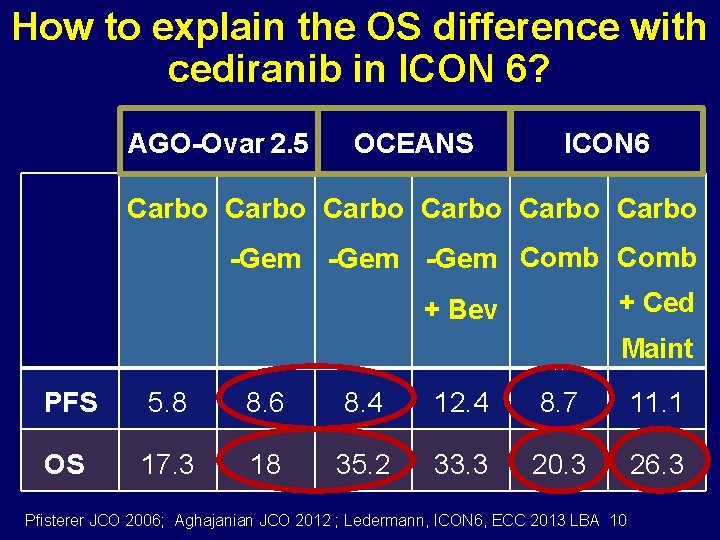
How to explain the OS difference with cediranib in ICON 6? AGO-Ovar 2. 5 OCEANS ICON 6 Carbo Carbo -Gem Comb + Ced + Bev Maint PFS 5. 8 8. 6 8. 4 12. 4 8. 7 11. 1 OS 17. 3 18 35. 2 33. 3 20. 3 26. 3 Pfisterer JCO 2006; Aghajanian JCO 2012 ; Ledermann, ICON 6, ECC 2013 LBA 10

Targeted therapy in ovarian cancer What we know: • New anti-angiogenetic drugs, both VEGF and Angiopoetin targeting drugs are active in prolonging PFS of ovarian cancer. • PARP inhibition is very interesting in high-grade serous ovarian cancer and especially in BRCA patients. • Current ongoing studies on targeted therapy base the targeted therapy on genetic profile of the tumor (often linked to the histological type). OVARIAN CANCER = MANY DISEASES!

Targeted therapy in ovarian cancer What we do not know: • When should an angiogenese inhibitor be given (first-line, platin-sensitive, platin-resistant)? Or should we give them in all lines? • Which angiogenesis inhibitor should be prefered in which group of ovarian cancer patients? • Biomarkers for efficacy of antiangiogenesis are still needed. • Potential for combination of VEGF inhibitors with other classes of antiangiogenetic drugs (e. g. ang inhibitors) or other targeted therapy such as e. g. PARP inhibitors.
- Slides: 18