BEERS LAW COLORIMETRY COLLIGATIVE PROPERTIES REVIEW Objectives Today

BEER’S LAW COLORIMETRY COLLIGATIVE PROPERTIES REVIEW

Objectives Today I will be able to: Apply Beer’s Law to determining the concentration of an unknown solution Informal assessment – monitor student questions and interactions as they complete the practice problems Formal assessment – analyze student responses to the practice and exit ticket

Lesson Sequence Evaluate: Warm – Up Explain: Beer’s Law Notes/Sample Data Elaborate: Colligative Properties Review Evaulate: Exit Ticket

Warm Up Think back to the Kool-Aid lab… What happened to the color of the solutions as the molarity increased? How would this observation impact the amount of light transmitted

Objectives Today I will be able to: Apply Beer’s Law to determining the concentration of an unknown solution

Homework Wear closed toe shoes Wednesday Study for Solutions Exam – Friday, May 15

Agenda Warm – Up Beer’s Law Notes/Sample Data Colligative Properties Review Exit Ticket

BEER’S LAW NOTES, INTRO TO COLORIMETRY

Beer’s Law quantifies the relationship between color and concentration Formula: A = ebc e is molar absorptivity (unique for a given compound) b is path length c concentration

WE CAN USE BEER’S LAW TO DETERMINE THE CONCENTRATIONS OF UNKNOWN SOLUTIONS

How do we do this? We use a technique called spectroscopy
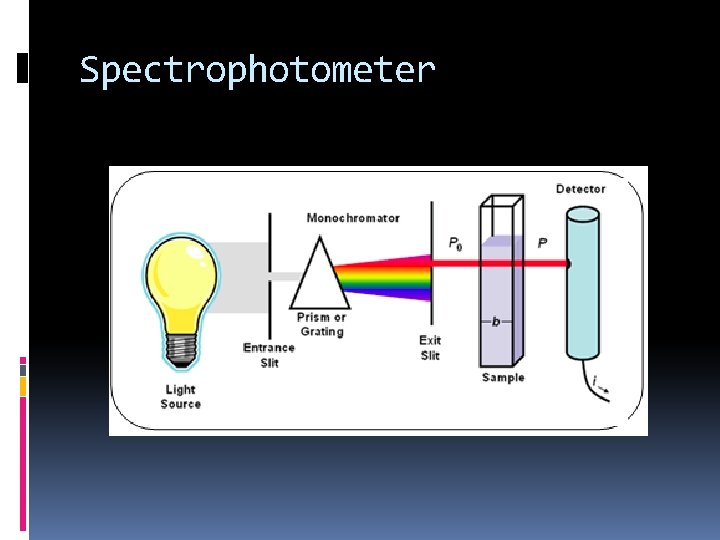
Spectrophotometer

How Do We Do This in lab? We make several solutions with known concentrations We determine which wavelength or color of light the solution responds to best We measure that transmittance and/or absorbance for the known solutions and the unknown solution We graph the knowns and interpolate for the unknown

SAMPLE SCENARIO

Sample Scenario A series of standard solutions containing a red dye was made by diluting a stock solution and then measuring the percent transmittance of each solution at 505 nm (greenish blue). This wavelength was selected by examining its absorption spectrum. If the solution looks red, it is absorbing red's complementary color of light, which is greenish blue. The results, after conversion to absorbance, are shown below.
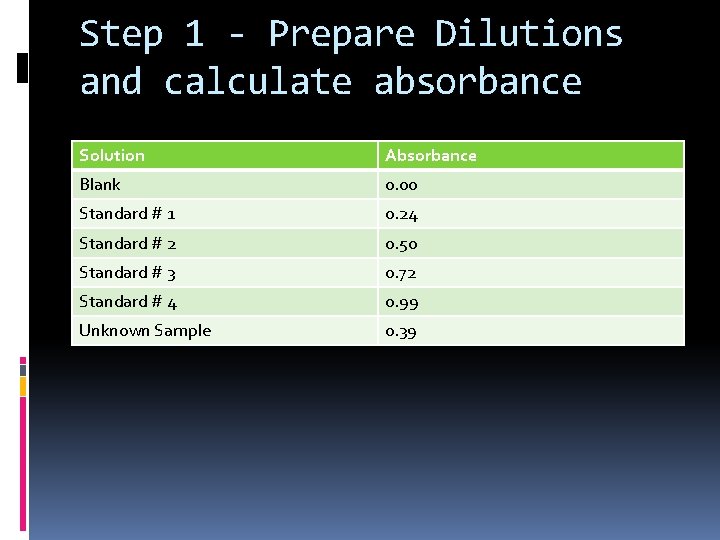
Step 1 - Prepare Dilutions and calculate absorbance Solution Absorbance Blank 0. 00 Standard # 1 0. 24 Standard # 2 0. 50 Standard # 3 0. 72 Standard # 4 0. 99 Unknown Sample 0. 39

Step 2 – Calculate Concentrations using Beer’s Law A = ebc Solution Absorbance Concentration Blank 0. 00 M Standard # 1 0. 24 0. 15 M Standard # 2 0. 50 0. 30 M Standard # 3 0. 72 0. 45 M Standard # 4 0. 99 0. 60 M Unknown Sample 0. 39 ? ? M Note: You would need to know the e-value and calculate the pathlength (b)
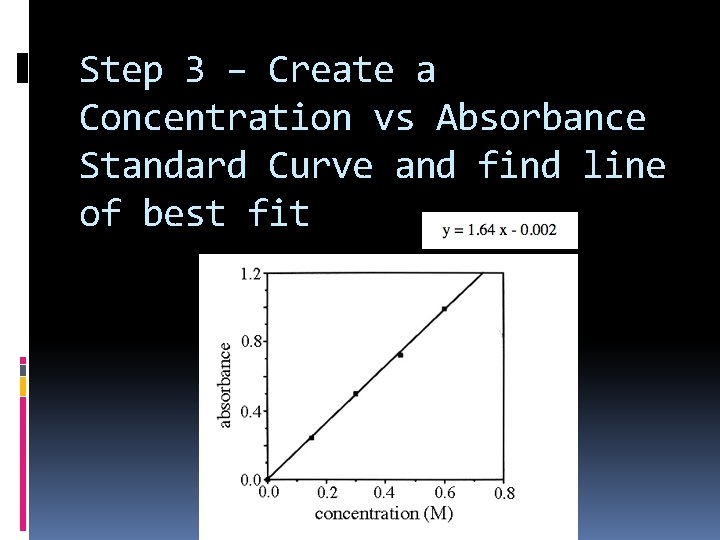
Step 3 – Create a Concentration vs Absorbance Standard Curve and find line of best fit

Step 4 – Determine the concentration of the unknown using the line of best fit

FINISH COLLIGATIVE PROPERTIES CALCULATIONS FROM FRIDAY

Exit Ticket What is the purpose of creating a standard curve when using a spectrophotometer? What steps do you take to determine the concentration of an unknown?
- Slides: 21