BEERS LAW AP Free Response Questions 2003 Beers

BEER’S LAW AP Free Response Questions 2003

Beer’s Law A student is instructed to determine the concentration is a solution of Co. Cl 2 based on the absorption of light. (spectrophotometer/colorimeter method). The student is provided with a 0. 10 M Co. Cl 2 solution with which they are to prepare standard solutions with concentrations of 0. 020 M, 0. 040 M, 0. 060 M and 0. 080 M. Describe the procedure for diluting the 0. 10 M solution to a concentration of 0. 020 M using distilled water, a 100 ml volumetric flask, and a pipet or buret. Include Specific amounts where appropriate.

Solution: 1 pt for Co. Cl 2; 1 pt for H 2 O M 1 V 1 = M 2 V 2 0. 10 M(V 1) = 0. 02 M(100 ml) V 1 = 20 ml Co. Cl 2 80 ml = H 2 O Pipet 20 ml of the Co. Cl 2 into a volumetric flask. Add enough distilled water to the flask to reach the 100 ml marking on the flask. Mix well.
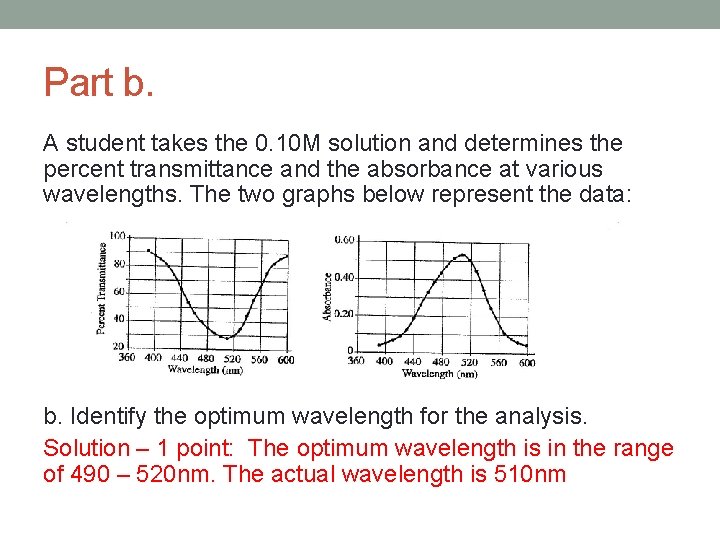
Part b. A student takes the 0. 10 M solution and determines the percent transmittance and the absorbance at various wavelengths. The two graphs below represent the data: b. Identify the optimum wavelength for the analysis. Solution – 1 point: The optimum wavelength is in the range of 490 – 520 nm. The actual wavelength is 510 nm

Part c The student measure the absorbance of the 0. 020 M, 0. 040 M, 0. 060 M, 0. 080 M and 0. 10 M solutions. The data are plotted below. c. The absorbance of the unknown solution is 0. 275. What is the concentration of the solution? Solution – 1 point: The acceptable range is 0. 045 M – 0. 055 M. The actual value is 0. 050 M.

d. Beer’s Law is an expression that includes three factors that determine the amount of light that passes through a solution. Identify two of these factors. Solution: 1 point each factor ID’d and explained A=abc a=molar absorbtivity b=path length of the cuvette c=concentration

Parts e. e. The student handles the sample container (cuvette) that holds the unknown solution and leaves fingerprints in the path of the light beam. How will this affect the calculated concentration of the unknown? Explain your answer. Solution e. The presence of fingerprints will scatter or absorb light. Since less light reaches the detector, the solutions will have a higher apparent absorbance (1 point), and therefore a higher reported concentration. (1 point)

Part f f. Why is this method of determining the concentration of Co. Cl 2 solution appropriate, whereas using the same method for measuring the concentration of Na. Cl solution would not be appropriate? Solution f. Co. Cl 2 is a colored solution with will absorb visible light (1 point) and Na. Cl is a colorless solution and will not absorb visible light (1 point).
- Slides: 8