Becoming apt at DAPT incorporating new dual antiplatelet

Becoming apt at DAPT: incorporating new dual antiplatelet therapy guidelines into clinical practice _________ Caitlin M. Gibson, Pharm. D, BCPS Assistant Professor, Department of Pharmacotherapy University of North Texas System College of Pharmacy
Learning objectives Pharmacists • Compare and contrast P 2 Y 12 receptor antagonists which may be used as part of a dual antiplatelet therapy (DAPT) regimen • Describe changes in recommended agents and duration of treatment for patients receiving DAPT for ischemic heart disease • Given a patient case, calculate a DAPT score and formulate a treatment recommendation for a patient receiving DAPT Technicians • Recognize the appropriate drugs and dosage forms to be used as part of a dual antiplatelet (DAPT) regimen • Identify appropriate DAPT regimens for patients with differing ischemic heart disease diagnoses and treatments • Identify standard treatment durations for patients receiving DAPT

Abbreviations • ACS = acute coronary syndromes • NNH = number needed to harm • ARR = absolute risk reduction • NNT = number needed to treat • ASA = aspirin • NSTE = non-ST elevation • BMS = bare metal stent • PCI = percutaneous coronary intervention • CABG = coronary artery bypass grafting • SIHD = stable ischemic heart disease • DAPT = dual antiplatelet therapy • STEMI = ST-elevation MI • DES = drug-eluting stent • TIA = transient ischemic attack • MACE = major adverse cardiovascular events • TIMI = thrombolysis in myocardial infarction • MI = myocardial infarction • USA = unstable angina
Outline Background Duration of DAPT Drug and dose selection Special populations
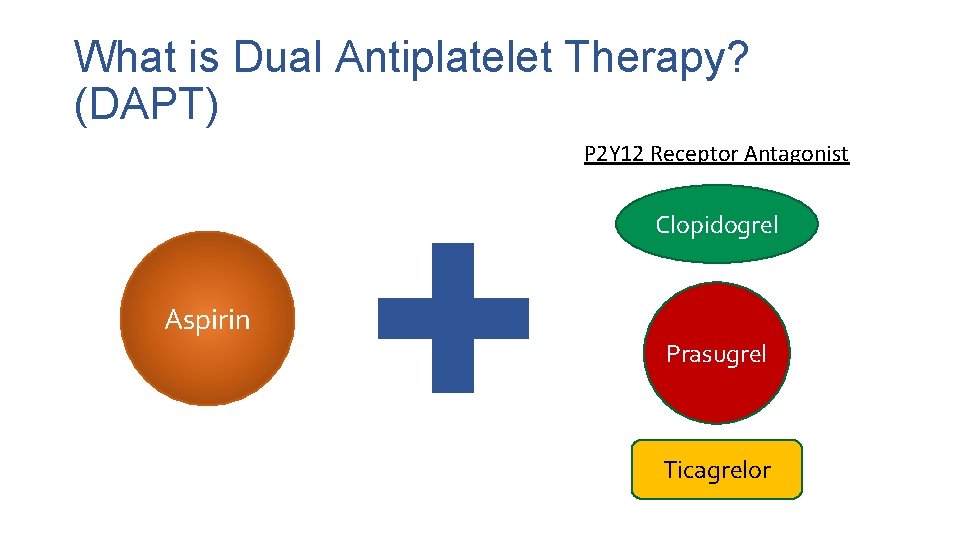
What is Dual Antiplatelet Therapy? (DAPT) P 2 Y 12 Receptor Antagonist Clopidogrel Aspirin Prasugrel Ticagrelor
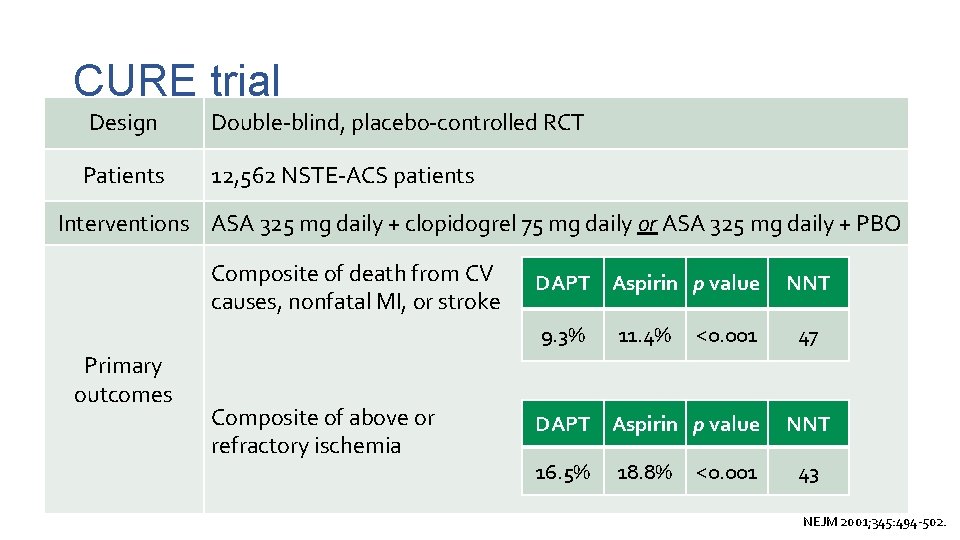
CURE trial Design Double-blind, placebo-controlled RCT Patients 12, 562 NSTE-ACS patients Interventions ASA 325 mg daily + clopidogrel 75 mg daily or ASA 325 mg daily + PBO Composite of death from CV causes, nonfatal MI, or stroke DAPT Aspirin p value 9. 3% Primary outcomes Composite of above or refractory ischemia 11. 4% <0. 001 DAPT Aspirin p value 16. 5% 18. 8% <0. 001 NNT 47 NNT 43 NEJM 2001; 345: 494 -502.
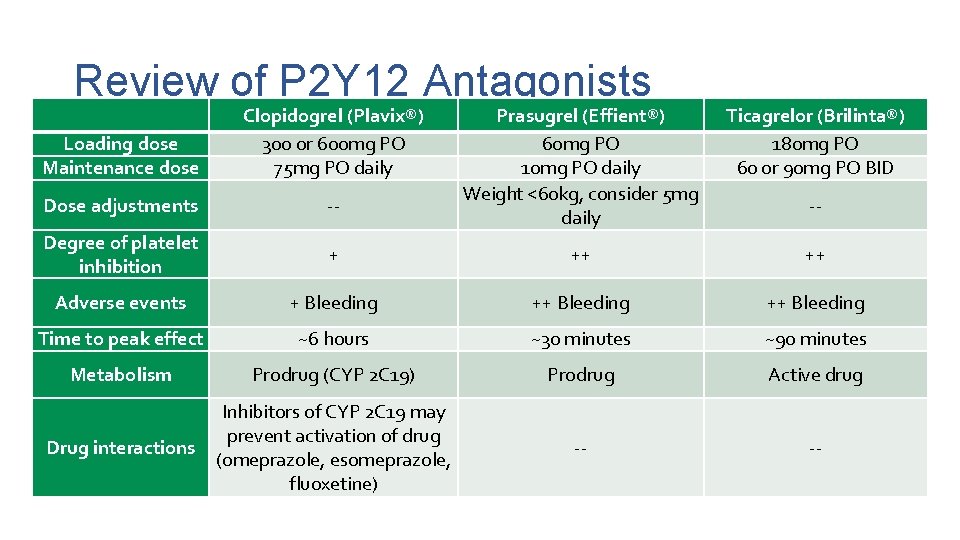
Review of P 2 Y 12 Antagonists Loading dose Maintenance dose Clopidogrel (Plavix®) 300 or 600 mg PO 75 mg PO daily Prasugrel (Effient®) 60 mg PO 10 mg PO daily Weight <60 kg, consider 5 mg daily Ticagrelor (Brilinta®) 180 mg PO 60 or 90 mg PO BID Dose adjustments -- Degree of platelet inhibition + ++ ++ Adverse events + Bleeding ++ Bleeding Time to peak effect ~6 hours ~30 minutes ~90 minutes Metabolism Prodrug (CYP 2 C 19) Prodrug Active drug Drug interactions Inhibitors of CYP 2 C 19 may prevent activation of drug (omeprazole, esomeprazole, fluoxetine) -- -- --

Review of P 2 Y 12 Antagonists Clopidogrel (Plavix®) Prasugrel (Effient®) Ticagrelor (Brilinta®) Contraindications Active bleeding History of CVA or TIA (stroke or mini-stroke) Active bleeding Pearls 25 -30% of patients have genetic polymorphism reducing efficacy of clopidogrel genetic or platelet reactivity testing Not for medical management Aspirin must be dosed <100 mg daily Can cause dyspnea Generic available? Yes No No

Where do these guidelines fit? • Updates to recommendations in various American Heart Association collaborative guidelines: SIHD PCI NSTE-ACS STEMI CABG Non-cardiac surgery
Outline Background Duration of DAPT Drug and dose selection Special populations
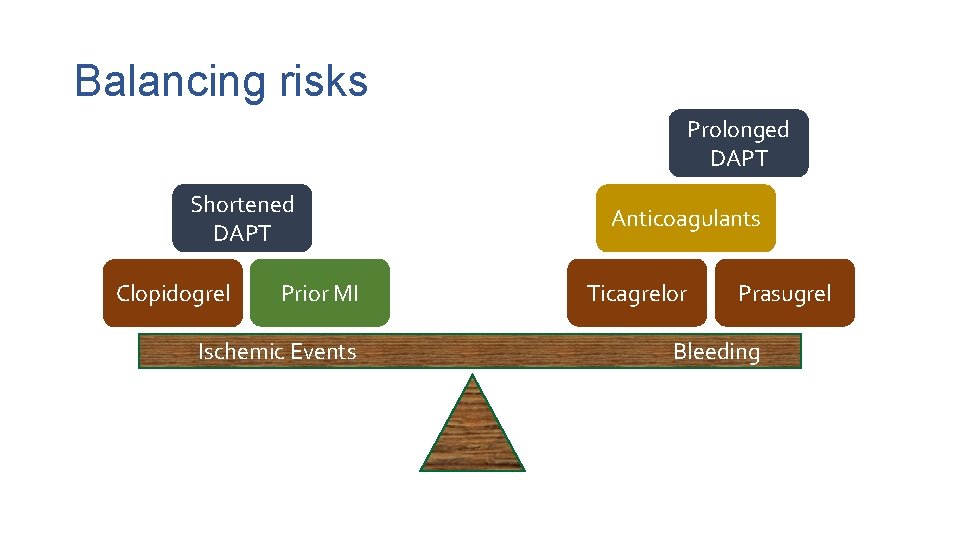
Balancing risks Prolonged DAPT Shortened DAPT Clopidogrel Prior MI Anticoagulants Ticagrelor Prasugrel Ischemic Events Bleeding
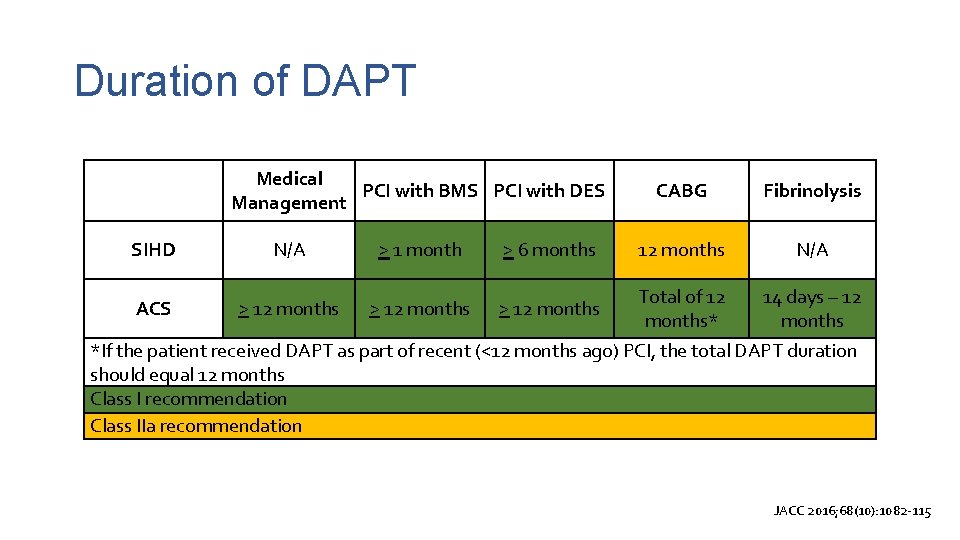
Duration of DAPT SIHD Medical PCI with BMS PCI with DES Management N/A > 1 month > 6 months CABG Fibrinolysis 12 months N/A Total of 12 14 days – 12 months* months *If the patient received DAPT as part of recent (<12 months ago) PCI, the total DAPT duration should equal 12 months Class I recommendation Class IIa recommendation ACS > 12 months JACC 2016; 68(10): 1082 -115

CABG recommendations • Mixed results in studies; most do not show benefit for DAPT • Largest meta-analysis showed DAPT vs. ASA alone resulted in: Reduced early saphenous vein graft occlusion RR=0. 59, 95% CI 0. 43 -0. 82, p=0. 002 Reduced incidence of 30 -day/inhospital mortality Incidence 0. 8% vs. 1. 9%, p<0. 001 Trend toward increase bleeding risk RR=1. 17, 95% CI 1. 00 -1. 37, p=0. 05 J Card Surg. 2013; 28: 109 -16.
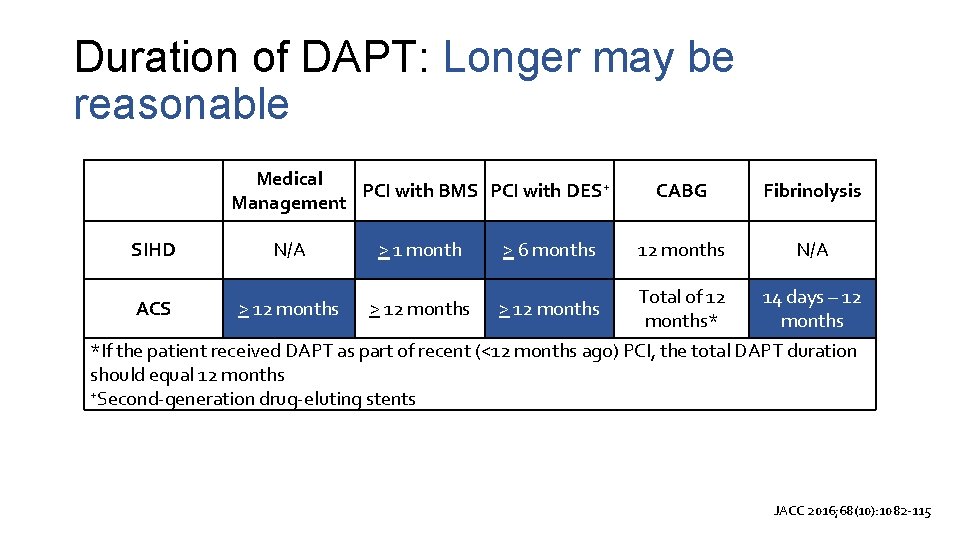
Duration of DAPT: Longer may be reasonable SIHD Medical PCI with BMS PCI with DES+ Management N/A > 1 month > 6 months CABG Fibrinolysis 12 months N/A Total of 12 14 days – 12 months* months *If the patient received DAPT as part of recent (<12 months ago) PCI, the total DAPT duration should equal 12 months +Second-generation drug-eluting stents ACS > 12 months JACC 2016; 68(10): 1082 -115

Duration of DAPT: Longer may be reasonable Trial Patients DAPT N = 9961 NEJM 2014; 371(23): 2155 -66. Any IHD with stent placed Intervention 30 vs. 12 mo DAPT after DES Drugs ASA 75162 mg Clopidogrel Prasugrel Endpoints Results: long vs. standard NNT/NNH Stent thrombosis 0. 4% vs. 1. 4% (p<0. 001) 100 Major adverse cardiovascular and cerebrovascular events 4. 3% vs. 5. 9% (p<0. 001) 63 Moderate-severe bleeding 2. 5% vs 1. 6% (p=0. 001) 111

Duration of DAPT: Shorter may be reasonable SIHD Medical PCI with BMS PCI with DES Management N/A > 1 month > 6 months CABG Fibrinolysis 12 months N/A Total of 12 14 days – 12 months* months *If the patient received DAPT as part of recent (<12 months ago) PCI, the total DAPT duration should equal 12 months ACS > 12 months JACC 2016; 68(10): 1082 -115
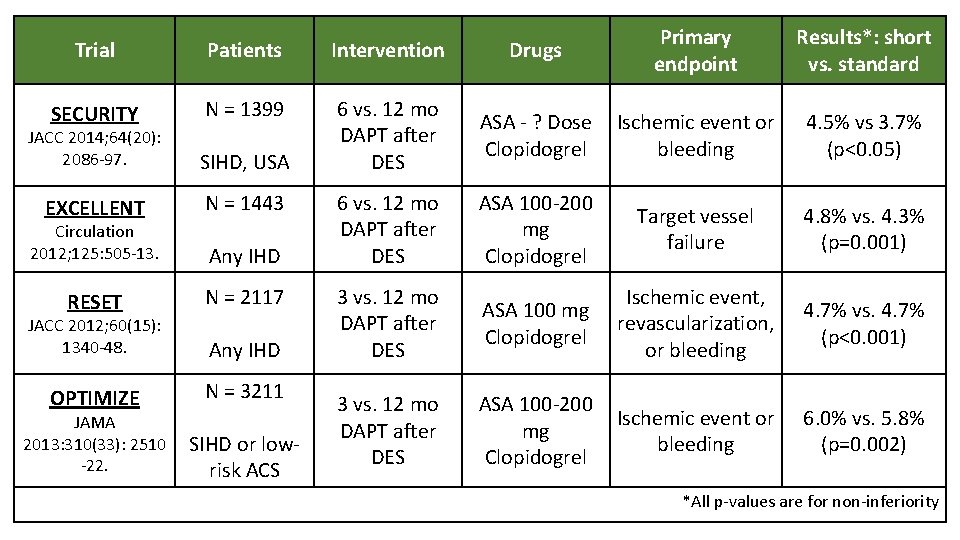
Trial Patients Intervention Drugs Primary endpoint Results*: short vs. standard SECURITY N = 1399 6 vs. 12 mo DAPT after DES ASA - ? Dose Clopidogrel Ischemic event or bleeding 4. 5% vs 3. 7% (p<0. 05) 6 vs. 12 mo DAPT after DES ASA 100 -200 mg Clopidogrel Target vessel failure 4. 8% vs. 4. 3% (p=0. 001) 3 vs. 12 mo DAPT after DES ASA 100 mg Clopidogrel Ischemic event, revascularization, or bleeding 4. 7% vs. 4. 7% (p<0. 001) 3 vs. 12 mo DAPT after DES ASA 100 -200 mg Clopidogrel Ischemic event or bleeding 6. 0% vs. 5. 8% (p=0. 002) JACC 2014; 64(20): 2086 -97. SIHD, USA EXCELLENT N = 1443 Circulation 2012; 125: 505 -13. Any IHD RESET N = 2117 JACC 2012; 60(15): 1340 -48. Any IHD OPTIMIZE N = 3211 JAMA 2013: 310(33): 2510 -22. SIHD or lowrisk ACS *All p-values are for non-inferiority

Risk vs. benefit of DAPT duration • Generally, extending duration of DAPT >12 months results in a 1 -2% decrease in late stent thrombosis & ischemic complications, but a 1% increase in risk of bleeding • For every 1000 patients, prolonged DAPT after DES implantation results in 6 fewer MIs and 3 fewer stent thomboses, but also 5 major bleeds • Prolonged DAPT is most likely to benefit patients with a history of MI JACC 2016; 68(10): 1082 -115
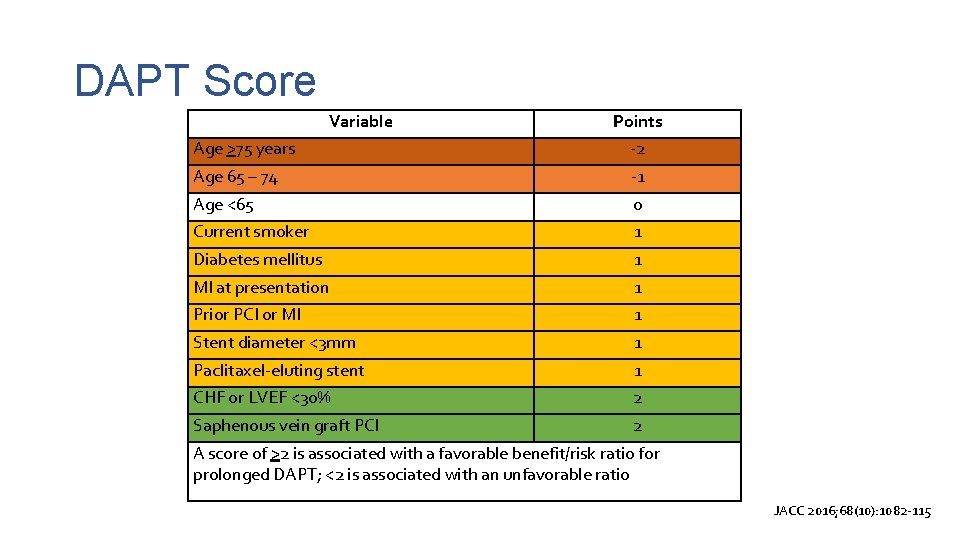
DAPT Score Variable Points Age >75 years -2 Age 65 – 74 -1 Age <65 0 Current smoker 1 Diabetes mellitus 1 MI at presentation 1 Prior PCI or MI 1 Stent diameter <3 mm 1 Paclitaxel-eluting stent 1 CHF or LVEF <30% 2 Saphenous vein graft PCI 2 A score of >2 is associated with a favorable benefit/risk ratio for prolonged DAPT; <2 is associated with an unfavorable ratio JACC 2016; 68(10): 1082 -115
Outline Background Duration of DAPT Drug and dose selection Special populations

Aspirin dosing in DAPT I A daily aspirin dose of 81 mg (range: 75 -100 mg) is recommended • Daily ASA doses as low as 30 -50 mg inactivate COX-1 and inhibit thromboxane production • Studies comparing low-dose to standard dose aspirin have consistently found comparable rates of ischemic events but higher bleeding rates with high dose aspirin: • TRANSLATE-ACS: NNT for 1 major bleed = 67 • PCI-CURE: NNT for 1 major bleed = 50 • Serenbrauny, et al: NNT for any bleed = 16
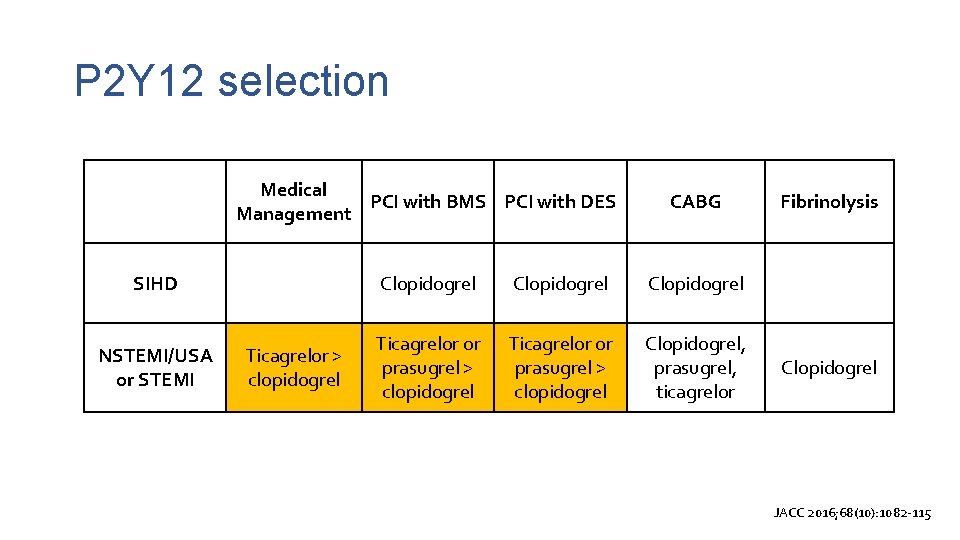
P 2 Y 12 selection Medical PCI with BMS PCI with DES Management CABG SIHD Clopidogrel Ticagrelor or prasugrel > clopidogrel Clopidogrel, prasugrel, ticagrelor NSTEMI/USA or STEMI Ticagrelor > clopidogrel Fibrinolysis Clopidogrel JACC 2016; 68(10): 1082 -115

Prasugrel – TRITON-TIMI 38 • Randomized, double-blind, multinational trial Prasugrel 60 mg 10 mg PO daily vs. Clopidogrel 300 mg 75 mg PO daily • Patients: ACS undergoing PCI (n=13, 608) Median age = 61 years; 74% male; 92% white race 74% NSTE-ACS; 26% STEMI 99% PCI (94% stented); 1% CABG New Eng J Med 2007; 357(20): 2001 -15.
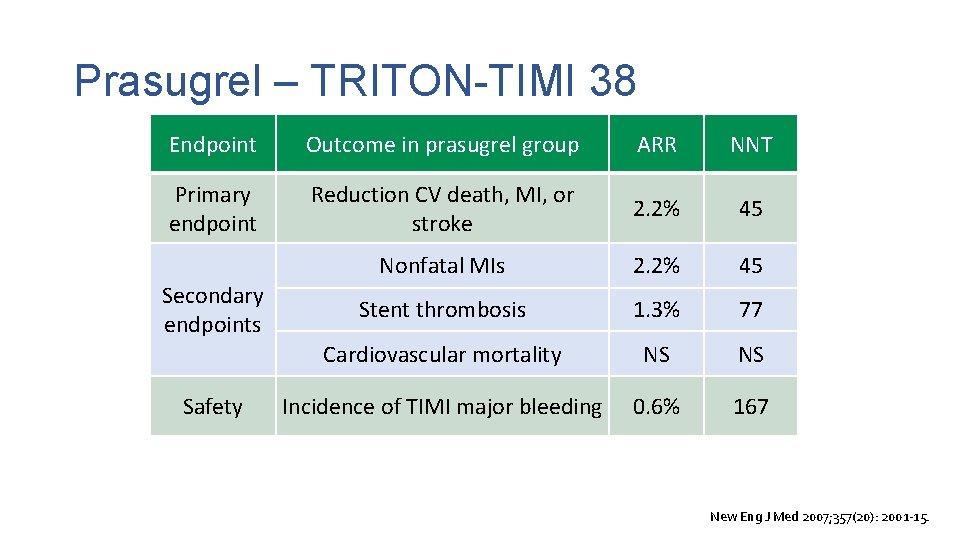
Prasugrel – TRITON-TIMI 38 Endpoint Outcome in prasugrel group ARR NNT Primary endpoint Reduction CV death, MI, or stroke 2. 2% 45 Nonfatal MIs 2. 2% 45 Stent thrombosis 1. 3% 77 Cardiovascular mortality NS NS Incidence of TIMI major bleeding 0. 6% 167 Secondary endpoints Safety New Eng J Med 2007; 357(20): 2001 -15.

- Patients with diabetes (HR 0. 70, 95% CI 0. 58 -0. 85) - History of stroke or transient ischemic attack (HR 1. 54, 95% CI 1. 02 -2. 32) - > 75 years - <60 kg No net benefit / net harm Net benefit Prasugrel – TRITON-TIMI 38
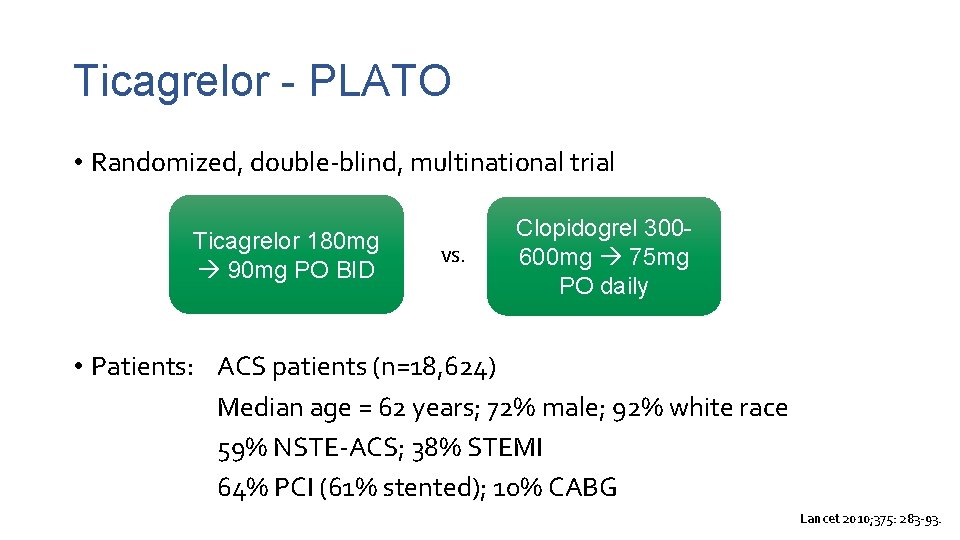
Ticagrelor - PLATO • Randomized, double-blind, multinational trial Ticagrelor 180 mg 90 mg PO BID vs. Clopidogrel 300600 mg 75 mg PO daily • Patients: ACS patients (n=18, 624) Median age = 62 years; 72% male; 92% white race 59% NSTE-ACS; 38% STEMI 64% PCI (61% stented); 10% CABG Lancet 2010; 375: 283 -93.
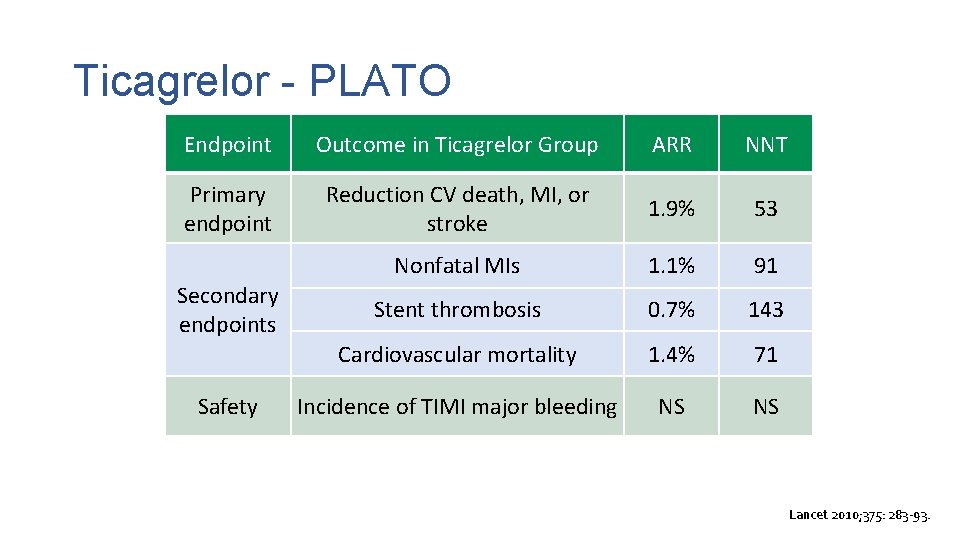
Ticagrelor - PLATO Endpoint Outcome in Ticagrelor Group ARR NNT Primary endpoint Reduction CV death, MI, or stroke 1. 9% 53 Nonfatal MIs 1. 1% 91 Stent thrombosis 0. 7% 143 Cardiovascular mortality 1. 4% 71 Incidence of TIMI major bleeding NS NS Secondary endpoints Safety Lancet 2010; 375: 283 -93.

Net benefit Prasugrel – TRITON-TIMI 38 No net benefit - Enrolled in North America (ASA dosing? ) - Less than median weight for sex - Not taking lipidlowering drugs

Ticagrelor – PEGASUS-TIMI 54 • Randomized, double-blind, 33 -months • Does long-term therapy with ticagrelor added to low-dose aspirin reduce the risk of major adverse cardiovascular events among stable patients with a history of myocardial infarction? Ticagrelor 90 mg PO BID + ASA vs. Ticagrelor 60 mg PO BID + ASA vs. Placebo + ASA • Patients: History of MI in prior 1 -3 years (n=21, 162) Median age = 65 years; 76% male; 87% white race 41% NSTE-ACS; 53% STEMI 83% PCI (61% stented); 10% CABG N Eng J Med 2015; 372(19): 1791 -1800.
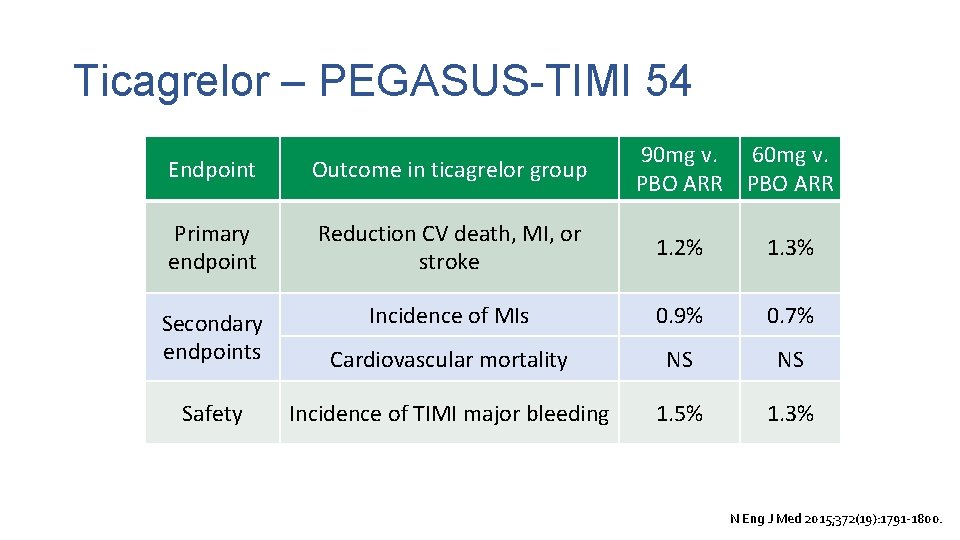
Ticagrelor – PEGASUS-TIMI 54 Endpoint Outcome in ticagrelor group 90 mg v. PBO ARR 60 mg v. PBO ARR Primary endpoint Reduction CV death, MI, or stroke 1. 2% 1. 3% Secondary endpoints Incidence of MIs 0. 9% 0. 7% Cardiovascular mortality NS NS Safety Incidence of TIMI major bleeding 1. 5% 1. 3% N Eng J Med 2015; 372(19): 1791 -1800.

Ticagrelor – PEGASUS-TIMI 54 • Patients had already received a year of DAPT • No head-to-head data against other P 2 Y 12 antagonists in this setting • FDA approved to reduce the rate of CV death, MI, and stroke in patients with a history of MI • Package insert: Administer 90 mg twice daily during the first year after an ACS event. After one year administer 60 mg twice daily
Outline Background Duration of DAPT Drug and dose selection Special populations

Triple therapy • Results in 2 -3 x increased bleeding risk • Recommendations: • • Assess ischemic and bleeding risks Keep duration as short as possible With warfarin, target INR of 2. 0 -2. 5 Clopidogrel is preferred P 2 Y 12 inhibitor JACC 2016; 68(10): 1082 -115

Proton pump inhibitors • Controversial drug interaction • Place in DAPT therapy: I History of prior gastrointestinal bleeding IIa Increased risk of gastrointestinal bleeding: • Advanced age • Concomitant warfarin, steroids, or nonsteroidal anti-inflammatory drugs III Routine use in low-risk patients JACC 2016; 68(10): 1082 -115

DAPT and elective non-cardiac surgery I Delay surgery 30 days after BMS implantation I Delay surgery optimally 6 months after DES implantation IIb Surgery may be considered after 3 months of DAPT III Surgery should not be performed within 30 days after BMS or 3 months of DES implantation JACC 2016; 68(10): 1082 -115

Take home points • Customizing duration of a DAPT regimen is reasonable • Weigh risk vs. benefit • Consider limitations of trials • Drug selection • Aspirin should be dosed at <100 mg in DAPT • Ticagrelor and prasugrel may be a reasonable preference over clopidogrel • Special populations • Use triple therapy sparingly • Reserve PPI use for high-risk patients

Becoming apt at DAPT: incorporating new dual antiplatelet therapy guidelines into clinical practice _________ Caitlin M. Gibson, Pharm. D, BCPS Assistant Professor, Department of Pharmacotherapy University of North Texas System College of Pharmacy
- Slides: 37