Because of global warming growingseason temperatures have increased

• Because of global warming, growing-season temperatures have increased by an average of 2°C (3. 59°F) for most of the world's highquality wine regions over the last fifty years. • In tandem with this rise in temperatures, the quality of vintages has also improved. It is predicted that by 2049, the growing-season temperature will increase by an average of 2. 04°C (3. 67°F.


MOLAR HEAT OF NEUTRALIZATION • 300 ml of 0. 2 M aqueous KOH neutralizes 150 ml of aqueous 0. 2 M H 2 SO 4. The temperature rises from 22. 3 o. C to 29. 2 o. C. Calculate the molar heat of neutralization of KOH. (Assume the specific heat capacity of KOH and H 2 SO 4 is the same as water)

How to solve these types of problems! • 300 ml of 0. 2 M DH = -Q / n aqueous KOH neutralizes 150 ml of aqueous 0. 2 M H 2 SO 4. 1) Q=m x c x Dt =(300+150)*4. 19*(29. 2 -22. 3) The temperature rises = 13009. 95 J from 22. 3 o. C to 29. 2 o. C. Calculate the molar = 13. 01 k. J heat of neutralization of KOH. • Now we must find the # of moles.

• 300 ml of 0. 2 M Molarity = n aqueous KOH v neutralizes 150 ml of n=molarity * volume aqueous 0. 2 M H 2 SO 4. The temperature rises n = 0. 2 (0. 3 L) = 0. 06 moles of KOH from 22. 3 o. C to 29. 2 o. C. Calculate the molar heat of neutralization • Now we must of KOH. calculate DH

• 300 ml of 0. 2 M DH = -Q / n aqueous KOH = -13. 01 / 0. 06 neutralizes 150 ml of = -216. 83 k. J/mol aqueous 0. 2 M H 2 SO 4. The temperature rises from 22. 3 o. C to 29. 2 o. C. Calculate the molar heat of neutralization of KOH.



Calculating D H: Hess’ Law • The third way to calculate DH is to use a technique developed by Germain Henri Hess. • Hess' Law states that the enthalpy of a reaction is independent of whether the reaction occurs in one or several steps.

• Using Hess’ Law we can algebraically add the given equations and their accompanying DH's to obtain the DH for the desired or target equation.

Keep the following rules in mind • 1) If an equation is multiplied or divided by a number, that factor also applies to DH. • 2) If an equation is reversed, then the sign of DH changes.

• 3) Remember that enthalpy changes with different states of matter. Do not, for example, interchange H 2 O(l) with H 2 O(g). .

EXAMPLE #1 (do not copy!) • Carbon disulfide is a very flammable solvent. It burns according to the following equation: • CS 2(l) + 3 O 2(g) --> CO 2(g) + 2 SO 2(g)

CS 2(l) + 3 O 2(g) --> CO 2(g) + 2 SO 2(g) • Calculate DH for the above reaction using the following data: • (1) C(s) + 2 S(s) --> CS 2(l) • (2) C(s) + O 2(g) --> CO 2(g) • (3) S(s) + O 2(g) --> SO 2(g) DH = 88 k. J DH = -394 k. J DH = -297 k. J

How to solve this problem? ? • 1) First look at the final equation. Look at the reactants and look at the products. CS 2(l) + 3 O 2(g) --> CO 2(g) + 2 SO 2(g) • Reactants: CS 2(l) + 3 O 2(g) • Products: CO 2(g) + 2 SO 2(g)

CS 2(l) + 3 O 2(g) --> CO 2(g) + 2 SO 2(g) • 1)Take the first • CS 2(l) --> C(s) + 2 S(s) equation and flip it so DH = - 88 k. J/mol that CS 2 is on the reactant side. The sign of DH changes. • 2) Equation 2 does not need to be changed because it has CO 2 on the product side. • C(s) + O 2(g) --> CO 2(g) DH = -394 k. J/mol
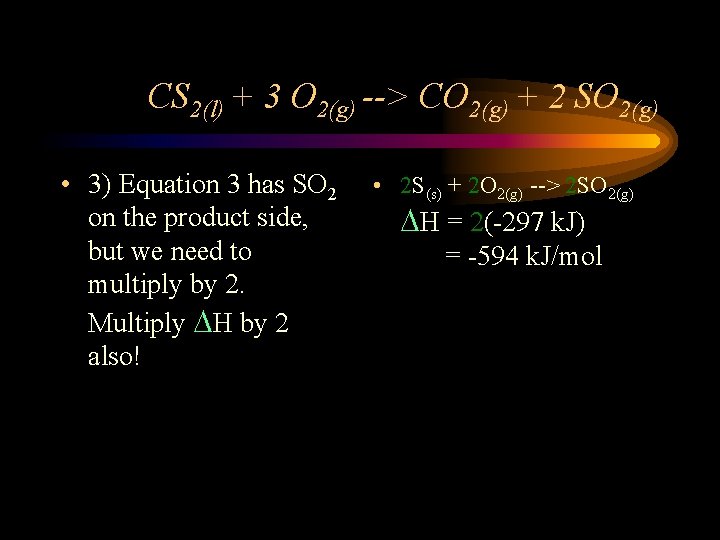
CS 2(l) + 3 O 2(g) --> CO 2(g) + 2 SO 2(g) • 3) Equation 3 has SO 2 on the product side, but we need to multiply by 2. Multiply DH by 2 also! • 2 S(s) + 2 O 2(g) --> 2 SO 2(g) DH = 2(-297 k. J) = -594 k. J/mol

• Add all three equations together and cancel the compounds that are the same but on opposite sides of the arrow. • Add the DH values and you will get your final answer. (DH = -1076 k. J/mol)
Calculating DH using Bond Energies

• Each type of bond has a characteristic bond energy • Breaking bonds requires energy. It is endothermic. • Making new bonds gives out energy. It is exothermic. • The energy associated for different bonds can be found on p. 156 & p. 419 of your textbook. • To calculate DH use the formula: DH = DH bonds broken - DH bonds formed

H 2(g) + Br 2(g) --> 2 HBr(g) • Calculate the DH for the above Bonds Broken: reaction. H-H 436 k. J/mol • You need to use the values of page 156 Br-Br 193 k. J/mol & 419 of your textbook. DH = DH bonds broken - DH bonds formed Bonds Formed: DH =(436+193) - (366+366) H-Br 366 k. J/mol DH = - 103 k. J/mol H-Br 366 k. J/mol
- Slides: 21