Beatrice Wong Pharm D BCPS October 22 2013

Beatrice Wong, Pharm. D, BCPS October 22, 2013 GUIDELINE BASED THERAPY FOR HEART FAILURE

Objectives � Review current guidelines for the management of heart failure with emphasis on updates. � Discuss case scenarios and evaluate treatment strategies. � Review recent trials that have impacted clinical practice.

Heart Failure Syndrome caused by cardiac dysfunction, generally resulting from myocardial muscle dysfunction or loss and characterized by either LV (left ventricular) dilation or hypertrophy or both. Reduced EF REF HF- (ejection fraction ≤ 40%) Preserved EF HF-PEF (ejection fraction ≥ 50%) HFSA 2010 guidelines, ACC 2013 guidelines
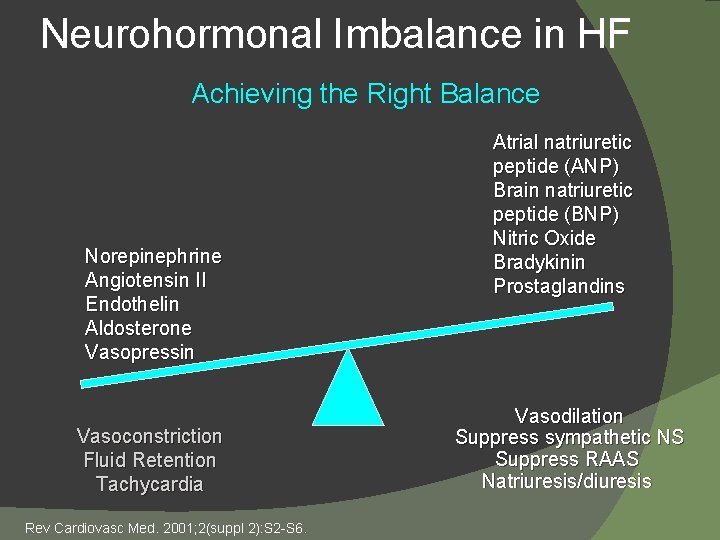
Neurohormonal Imbalance in HF Achieving the Right Balance Norepinephrine Angiotensin II Endothelin Aldosterone Vasopressin Vasoconstriction Fluid Retention Tachycardia Rev Cardiovasc Med. 2001; 2(suppl 2): S 2 -S 6. Atrial natriuretic peptide (ANP) Brain natriuretic peptide (BNP) Nitric Oxide Bradykinin Prostaglandins Vasodilation Suppress sympathetic NS Suppress RAAS Natriuresis/diuresis
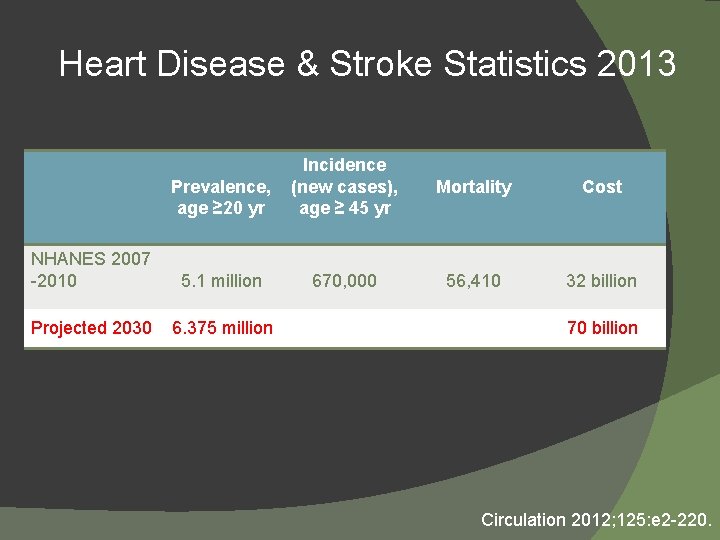
Heart Disease & Stroke Statistics 2013 Prevalence, age ≥ 20 yr Incidence (new cases), age ≥ 45 yr Mortality Cost NHANES 2007 -2010 5. 1 million 670, 000 56, 410 32 billion Projected 2030 6. 375 million 70 billion Circulation 2012; 125: e 2 -220.

Guidelines for Management American College of Cardiology Foundation/American Heart Association (ACCP/AHA) � Heart Failure Society of America (HFSA) � Canadian Cardiology Society (CCS) � European Society of Cardiology (ESC) � ACC/AHA 2009 ACC/AHA 2013 CCS 2012 HFSA 2010 ESC 2012

Classification of Recommendations � Classes of Recommendations � Class I- Evidence and/or general agreement that a given treatment or procedure is beneficial, useful, effective. � Class II- Conflicting evidence and/or divergence of opinion ○ IIa- Weight of evidence/opinion is in favour ○ IIb- Usefulness is less well established � Class III- Evidence or general agreement that given treatment is not useful/effective and in some cases may be harmful. � Level of Evidence � Level A- derived from multiple randomized clinical trials or meta-analyses � Level B- derived from a single randomized clinical trial or large nonrandomized studies � Level C- consensus of opinion of the experts and/or small studies, retrospective studies, or registries

Case 1 JK is a 65 yo male with ischemic cardiomyopathy with an EF of 35%. He presents with shortness of breath and fatigue even with walking up the 1 flight of stairs in his home. Meds: Furosemide 20 mg po daily Aspirin 81 mg po daily What class of heart failure does this patient have? What other medications should JK be on?

Types of Classification NYHA (New York Heart Association) ACC-AHA Stages of Heart Failure � I- Cardiac disease but no limitation of physical activity. � A- At high risk but no structural disease or symptoms. � II- Slight limitation of activity. Ordinary activity results in fatigue, palpitation, dyspnea, or anginal pain. � B- Asymptomatic, + structural heart disease (impaired LV [left ventricular] function, hypertrophy, or chamber distortion) � III- Marked limitation of physical activity. Less than ordinary activity causes fatigue, palpitation, dyspnea, or anginal pain. � C- Prior or current symptoms of HF, + structural heart disease � D- Refractory HF requiring specialized interventions � IV-Inability to carry on any physical activity without discomfort, maybe even present at rest.

Case 1 JK is a 65 yo male with ischemic cardiomyopathy with an EF of 35%. He presents with shortness of breath and fatigue even with walking up the 1 flight of stairs in his home. Meds: Furosemide 20 mg po daily Aspirin 81 mg po daily What class of heart failure does this patient have? Stage C, NYHA class III What other medications should JK be on?

Systolic Heart Failure � Goals of treatment �Reduction in symptoms �Decrease the rate of hospitalization �Prevention of premature death � Therapeutic approaches �Lifestyle modification �Implantable devices �Surgery – ventricular assist devices �Pharmacologic therapy

Loop Diuretics Bumetanide Furosemide Torsemide Equivalent dose (po) 1 mg 40 mg 20 mg “Maximum” dose Bioavailability T½ IV: PO conversion 10 mg 85% 1 -1. 5 hr 1: 1 400 mg 60% 1. 5 -3 hrs 1: 2 200 mg 85% 3 -6 hrs 1: 1 Additional characteristics � Torsemide is absorbed more rapidly/completely than bumetanide or furosemide � Furosemide – metabolized/excreted in the kidney � Torsemide/bumetanide – metabolized in liver � Ethacrynic acid (Edecrin) � Loop diuretic lacking sulfonamide moiety, ~ equipotent to furosemide � Dosing ○ Oral: 50 -100 mg/day in 1 -2 divided doses, max 400 mg/day ○ IV: 25 -100 mg/dose, dose qd to q 8 h as required � Side effect profile ○ Similar to other loops, except incidence of GI bleed and hematologic manifestations �

Diuretic Resistance � Decreased perfusion due to low cardiac output/vol. overload (require active transport to site of action) � Intestine – decreased/delayed absorption of diuretics � Kidney – decreased delivery of diuretics to the site of action � Decreased GFR results in decreased filtered load of sodium � Intravascular volume depletion activates plasma renin activity and stimulates SNS � Increased proximal tubule reabsorption of sodium, particularly in setting of elevated AII and elevated catecholamine levels � Increased distal reabsorbtion of sodium, stimulated by aldosterone � Na+ rebound - Elevation of AVP upregulation of Na+-K+-2 Cl � Associated with tubular hypertrophy: resetting basal rates of sodium reabsorbtion � Poor dosing strategies and patient compliance

Combination Therapy � Loop or Thiazide + Potassium Sparing �Rationale: prevent hypokalemia � Loop + Thiazide �Rationale: synergy when diuresis is inadequate with monotherapy or utilizing high doses of loops � Loop + Albumin �Rationale: questionable, may promote in UOP during oliguric renal failure

What else?
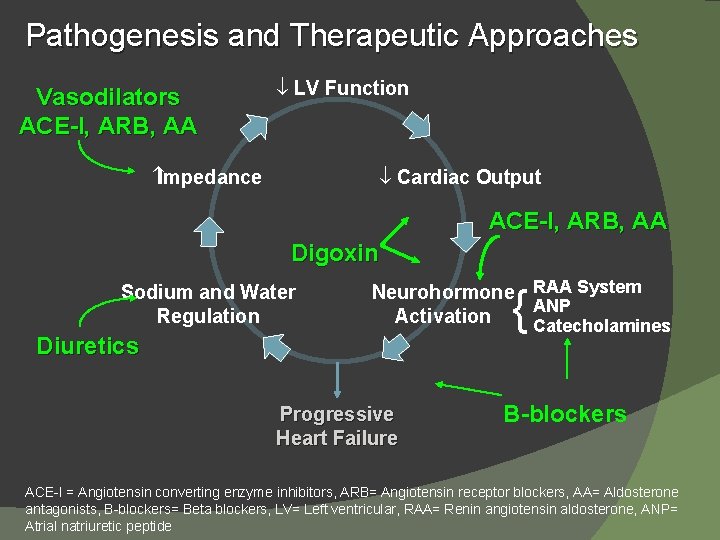
Pathogenesis and Therapeutic Approaches Vasodilators ACE-I, ARB, AA LV Function Impedance Cardiac Output ACE-I, ARB, AA Digoxin Sodium and Water Regulation Diuretics Neurohormone RAA System ANP Activation Catecholamines Progressive Heart Failure { B-blockers ACE-I = Angiotensin converting enzyme inhibitors, ARB= Angiotensin receptor blockers, AA= Aldosterone antagonists, B-blockers= Beta blockers, LV= Left ventricular, RAA= Renin angiotensin aldosterone, ANP= Atrial natriuretic peptide
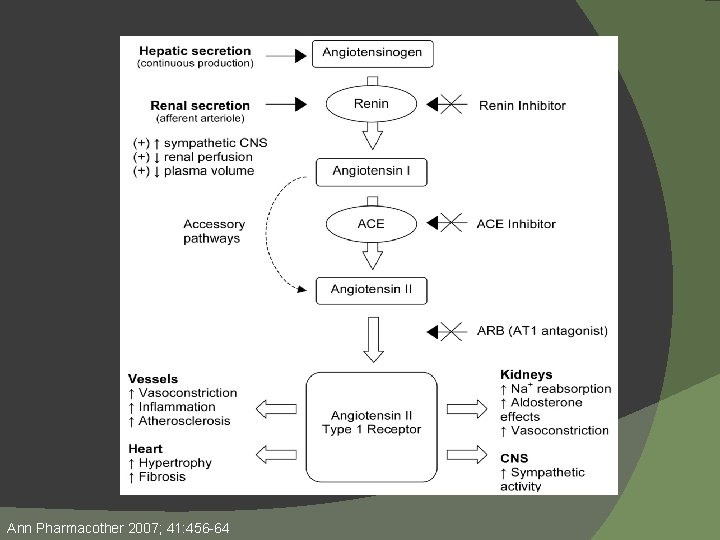
Ann Pharmacother 2007; 41: 456 -64
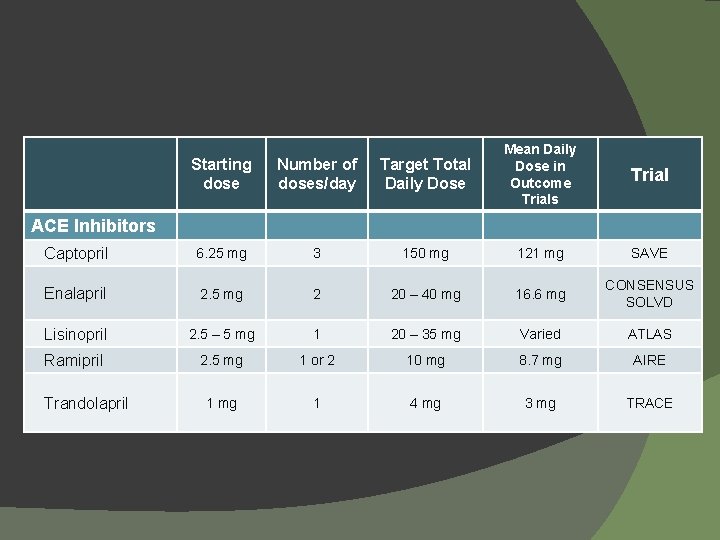
Starting dose Number of doses/day Target Total Daily Dose Mean Daily Dose in Outcome Trials Captopril 6. 25 mg 3 150 mg 121 mg SAVE Enalapril 2. 5 mg 2 20 – 40 mg 16. 6 mg CONSENSUS SOLVD Lisinopril 2. 5 – 5 mg 1 20 – 35 mg Varied ATLAS Ramipril 2. 5 mg 1 or 2 10 mg 8. 7 mg AIRE 1 mg 1 4 mg 3 mg TRACE Trial ACE Inhibitors Trandolapril
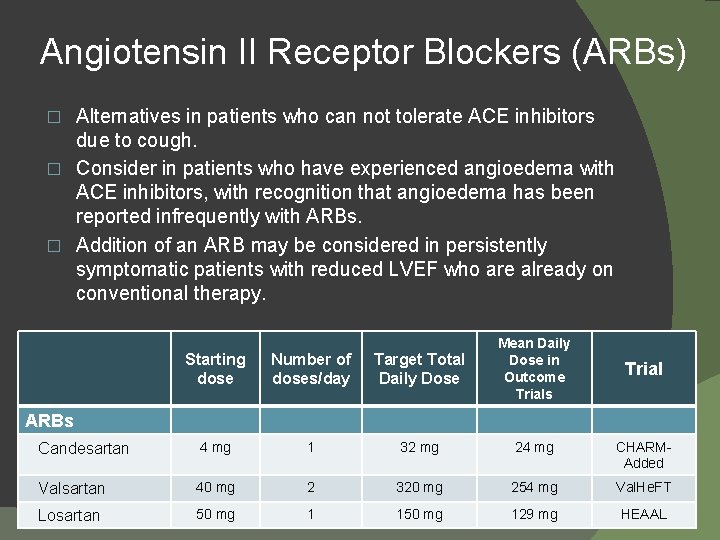
Angiotensin II Receptor Blockers (ARBs) Alternatives in patients who can not tolerate ACE inhibitors due to cough. � Consider in patients who have experienced angioedema with ACE inhibitors, with recognition that angioedema has been reported infrequently with ARBs. � Addition of an ARB may be considered in persistently symptomatic patients with reduced LVEF who are already on conventional therapy. � Starting dose Number of doses/day Target Total Daily Dose Mean Daily Dose in Outcome Trials Candesartan 4 mg 1 32 mg 24 mg CHARMAdded Valsartan 40 mg 2 320 mg 254 mg Val. He. FT Losartan 50 mg 1 150 mg 129 mg HEAAL Trial ARBs

Practical Considerations in Use of ACEi/ARBs � All agents � ↑K+, ↑ SCr, symptomatic/asymptomatic hypotension (↓ Na+, ↓ volume) ○ Change in SCr is to be expected - what is an acceptable % change? ? � Monitoring recommendations – SCr, K+ within 5 to 7 days of initiation and with dose titrations � ACE Inhibitors � Angioedema (≤ 1%) , non-productive cough (5 -15%), skin rash � ARBs � Angioedema cross-reactivity � Renin Inhibitors ○ Angioedema cross-reactivity ? ?

Practical Considerations � Drug Interactions � K+ supplements, K+ sparing diuretics � Aldosterone antagonists � “Low-salt” substitutes with high K+ content � NSAIDs � Lithium � Contraindications/Precautions/Warnings � History of angioedema � Bilateral renal artery stenosis � SCr > 2. 5 mg/d. L at baseline � Serum K+ level > 5 m. Eq/L � SBP < 90 mm. Hg � Pregnancy category C (1 st trimester)/ D (2 nd/3 rd trimester)

Beta-blockers � Beta-blockers mediate their effect by complex interactions with -adrenoreceptors (2 subtypes) � 1 -receptors ○ Heart and kidney (juxtaglomerular cells) ○ Stimulation - HR, contractility and renin release � 2 -receptors ○ Lungs, liver, pancreas, and arteriolar smooth muscle ○ Stimulation - bronchodilation and vasodilation � The ability to reduce plasma renin levels and thus angiotensin II concentrations – major cardiovascular benefit

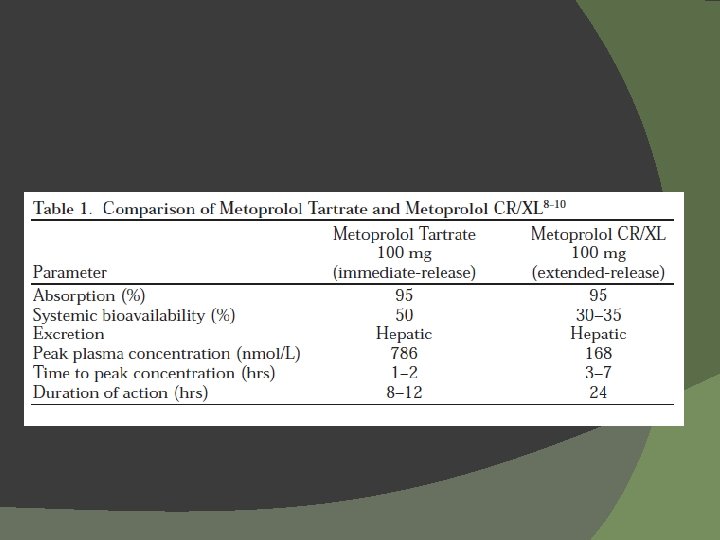
Beta-blockers Agent Starting Dose Max Dose Mean Dose Achieved in Trials Bisoprolol 1. 25 mg QD 10 mg QD 8. 6 mg/day Carvedilol 3. 125 mg BID 50 mg BID 37 mg/day Metoprolol XL 12. 5 -25 mg QD* 200 mg QD 159 mg/day *Metoprolol XL commonly dosed BID in clinical practice. Circulation. 2013; 128: e 240 -327.
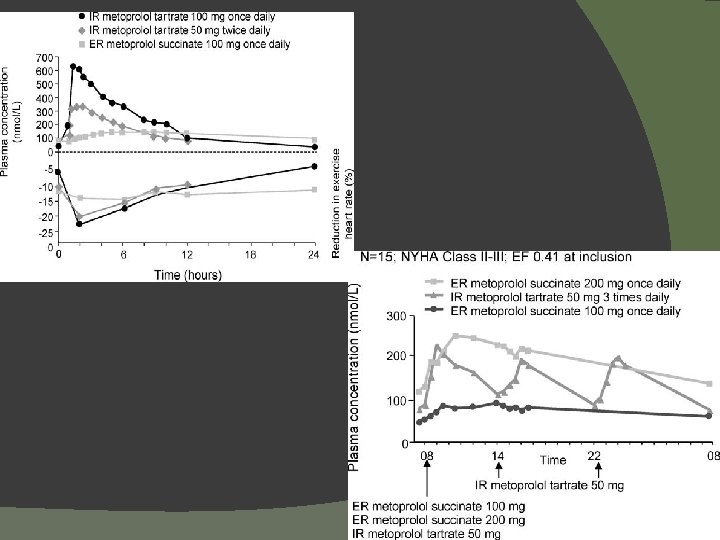
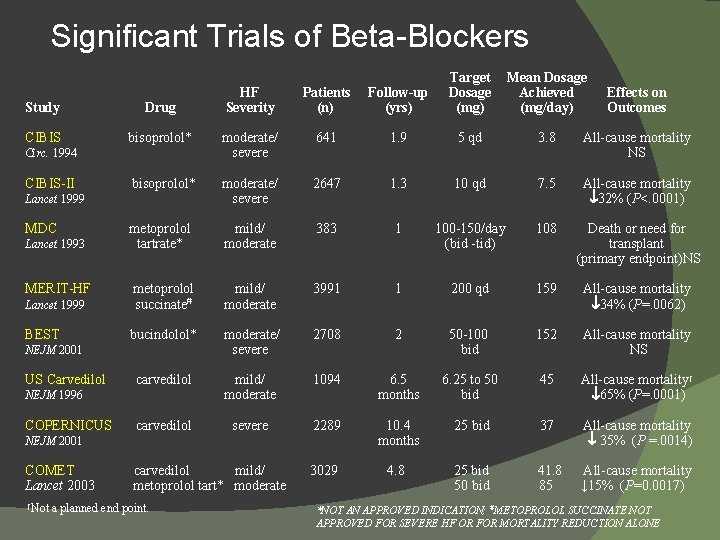
Significant Trials of Beta-Blockers HF Severity Patients (n) Follow-up (yrs) Target Dosage (mg) Mean Dosage Achieved (mg/day) Effects on Outcomes Study Drug CIBIS bisoprolol* moderate/ severe 641 1. 9 5 qd 3. 8 All-cause mortality NS bisoprolol* moderate/ severe 2647 1. 3 10 qd 7. 5 All-cause mortality 32% (P<. 0001) metoprolol tartrate* mild/ moderate 383 1 100 -150/day (bid -tid) 108 Death or need for transplant (primary endpoint)NS mild/ moderate 3991 1 200 qd 159 Lancet 1999 metoprolol succinate# All-cause mortality 34% (P=. 0062) BEST bucindolol* moderate/ severe 2708 2 50 -100 bid 152 All-cause mortality NS carvedilol mild/ moderate 1094 6. 5 months 6. 25 to 50 bid 45 All-cause mortality† 65% (P=. 0001) carvedilol severe 2289 10. 4 months 25 bid 37 All-cause mortality 35% (P =. 0014) 3029 4. 8 25 bid 50 bid 41. 8 85 All-cause mortality ↓ 15% (P=0. 0017) Circ. 1994 CIBIS-II Lancet 1999 MDC Lancet 1993 MERIT-HF NEJM 2001 US Carvedilol NEJM 1996 COPERNICUS NEJM 2001 COMET Lancet 2003 †Not carvedilol mild/ metoprolol tart* moderate a planned end point. *NOT AN APPROVED INDICATION; #METOPROLOL SUCCINATE NOT APPROVED FOR SEVERE HF OR FOR MORTALITY REDUCTION ALONE
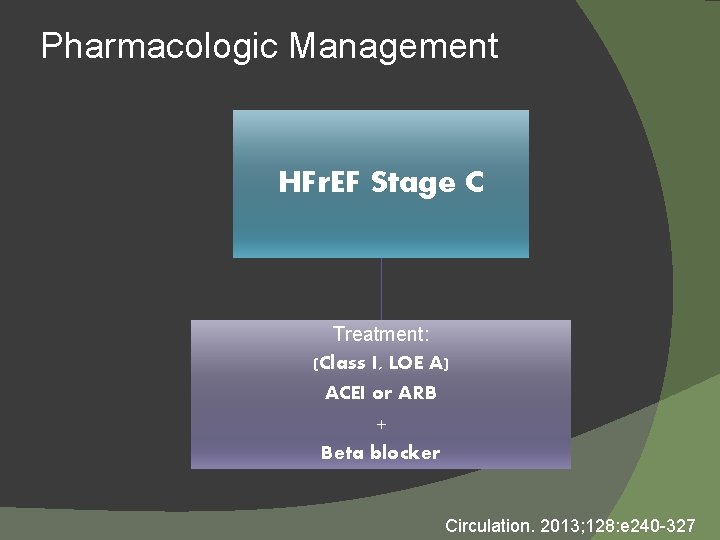
Pharmacologic Management HFr. EF Stage C Treatment: (Class I, LOE A) ACEI or ARB + Beta blocker Circulation. 2013; 128: e 240 -327

Case 1 (continued) JK is seeing you at clinic for f/u after recent hospitalization. He reports having to stop due to fatigue when he walks about a block. Current meds: Lisinopril 10 mg po BID Carvedilol 25 mg po BID Furosemide 20 mg po QD Aspirin 81 mg po QD EF 30% (echo in hospital) BP: 90/67, HR 72 Labs: Scr 1. 5 (stable), K 4. 2 What other meds could you consider adding at this point?

Otherapies: Which one do we add? � Aldosterone antagonists/Mineralocorticoid receptor antagonists (MRAs) � ARBs � Hydralazine/nitrates � Digoxin
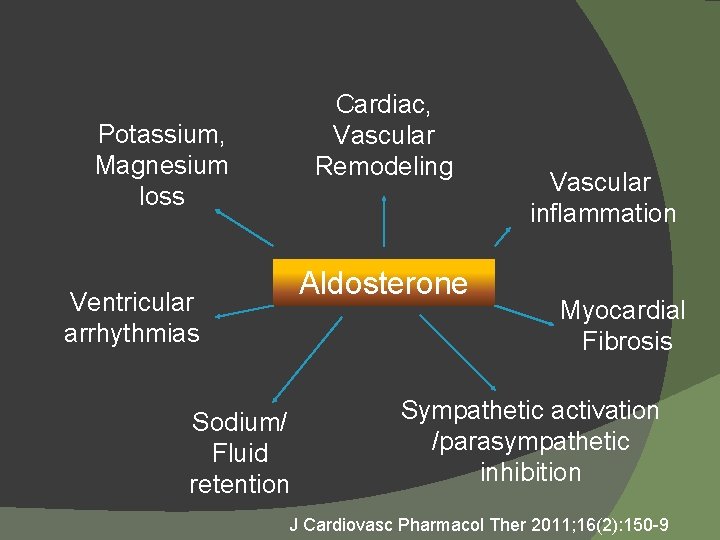
Cardiac, Vascular Remodeling Potassium, Magnesium loss Aldosterone Ventricular arrhythmias Sodium/ Fluid retention Vascular inflammation Myocardial Fibrosis Sympathetic activation /parasympathetic inhibition J Cardiovasc Pharmacol Ther 2011; 16(2): 150 -9

Aldosterone Antagonists � � AKA – “Mineralocorticoid Receptor Antagonists” Mechanism � Competitively inhibits the mineralocorticoid receptor in the collecting tubules � Spironolactone ○ Non-specific affinity for receptor ○ Possess antiadrogenic and progestational activity ○ Antiandrogenic and progestational activity � Men: gynecomastia and impotence � Women: menstrual irregularities ○ Other adverse effects: hyperkalemia � Eplerenone � Competitive and highly specific � Lacks hormone activity
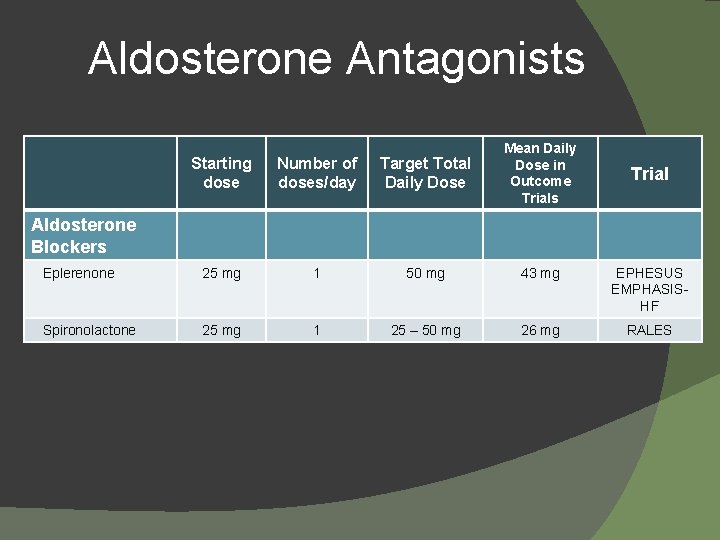
Aldosterone Antagonists Starting dose Number of doses/day Target Total Daily Dose Mean Daily Dose in Outcome Trials Eplerenone 25 mg 1 50 mg 43 mg EPHESUS EMPHASISHF Spironolactone 25 mg 1 25 – 50 mg 26 mg RALES Trial Aldosterone Blockers

EMPHASIS-HF NYHA Class II, age ≥ 55 yrs, EF ≤ 30% (randomized within 6 mo after hospitalization) N=2737 Eplerenone Placebo NEJM 2011; 364: 11 -21.
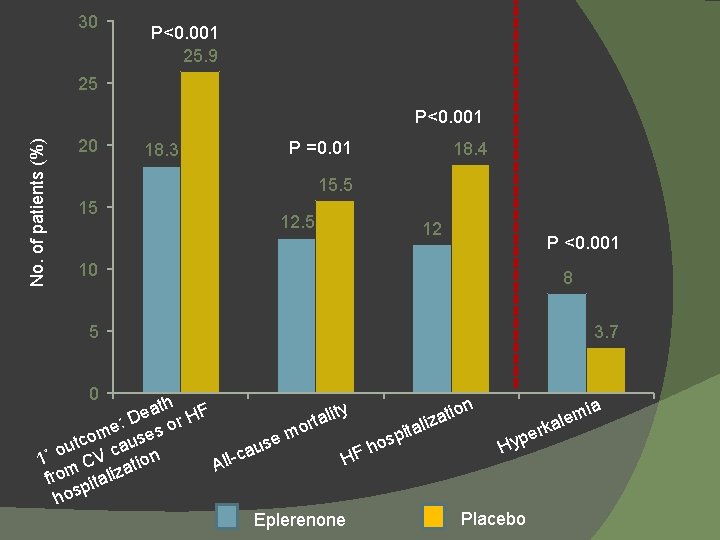
30 P<0. 001 25. 9 25 No. of patients (%) P<0. 001 20 18. 3 P =0. 01 18. 4 15. 5 15 10 5 0 12. 5 12 P <0. 001 8 3. 7 ia ath HF ity tion e m l a e a D l t z r e: es o or ka ali t r i m m e p o yp tc caus se os u u H h o a V tion HF 1˚ ll-c C A a from pitaliz hos Placebo Eplerenone

Hydralazine/nitrates Initial use of nitrates in combination with hydralazine was based on a hemodynamic concept designed to achieve a “nitroprussidelike” effect � Nitrates + Hydralazine � �Complementary hemodynamic action ○ Nitrates (preload) – via activation of guanylate cyclase c. GMP in vascular smooth muscle ○ Hydralazine (afterload) – direct-acting vasodilator �Effect on HF outcomes? ○ Survival benefit demonstrated in V-He. FT I and V-He. FT II ○ Demonstrated improvement in other markers for cardiac function including exercise tolerance and LVEF
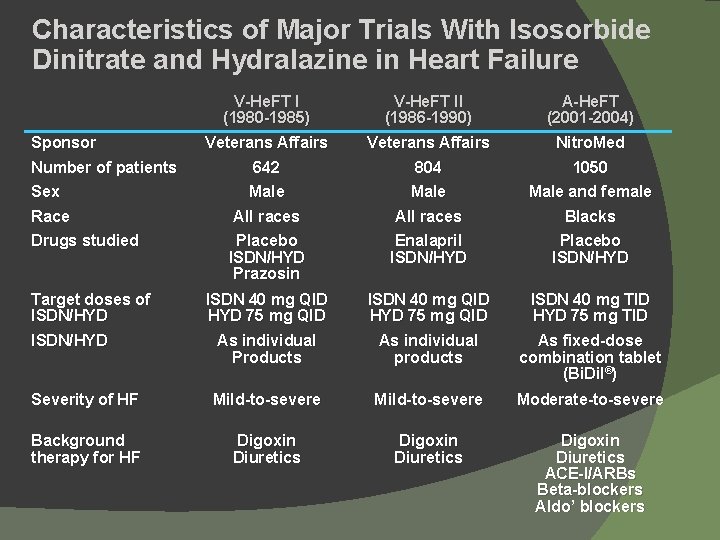
Characteristics of Major Trials With Isosorbide Dinitrate and Hydralazine in Heart Failure V-He. FT I (1980 -1985) V-He. FT II (1986 -1990) A-He. FT (2001 -2004) Veterans Affairs 642 Veterans Affairs 804 Nitro. Med 1050 Male All races Male and female Blacks Placebo ISDN/HYD Prazosin Enalapril ISDN/HYD Placebo ISDN/HYD ISDN 40 mg QID HYD 75 mg QID ISDN 40 mg TID HYD 75 mg TID ISDN/HYD As individual Products As individual products As fixed-dose combination tablet (Bi. Dil®) Severity of HF Mild-to-severe Moderate-to-severe Background therapy for HF Digoxin Diuretics ACE-I/ARBs Beta-blockers Aldo’ blockers Sponsor Number of patients Sex Race Drugs studied Target doses of ISDN/HYD
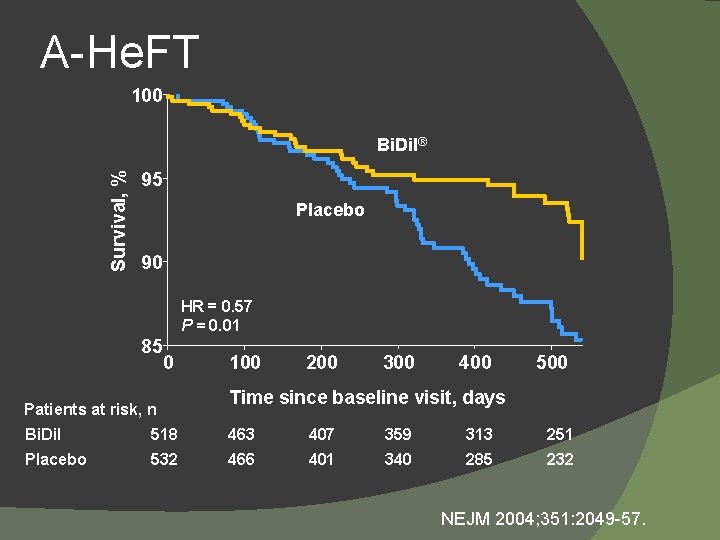
A-He. FT 100 Survival, % Bi. Dil® 95 Placebo 90 HR = 0. 57 P = 0. 01 85 0 Patients at risk, n 100 200 300 400 500 Time since baseline visit, days Bi. Dil 518 463 407 359 313 251 Placebo 532 466 401 340 285 232 NEJM 2004; 351: 2049 -57.

Practical Considerations � Effect of hydralazine on nitrate tolerance �HYD potentiates vasorelaxing effect of NTG and reduces the formation of nitrate-mediated vascular superoxide � Nitrates � Avoid use with PDE inhibitors such as sildenafil � Hydralazine � Reflex tachycardia/palpitations, flushing, exacerbation of angina, Na+/water retention � Immunological-type reactions ○ ○ Lupus-like syndrome (dose/duration related, > 6 months) Vasculitis, Dermatitis Glomerulonephritis Peripheral neuropathy, drug-induced hepatitis

Digoxin � Blocks Na+ / K+ ATPase => Ca+ + � “Inotropic” effect � No mortality benefit � Neurohormonal control - Plasma Noradrenaline - Peripheral nervous system activity - RAAS activity - Vagal tone - Normalizes arterial baroreceptors NEJM 1988; 318: 358

Digoxin in HF � Optimal serum concentrations �“Normal” serum level range: � HF patients ○ No added benefit with from 0. 7 -0. 8 to 1. 2 -1. 5 ng/m. L � EF, HF symptoms, exercise tolerance and neurohormone levels � Escalating levels beyond 0. 8 ng/m. L more likely harmful ○ PROVED/RADIANCE (retrospective analysis) � 0. 5 to 0. 9 ng/m. L; no added benefit with levels ○ DIG (retrospective analysis) � 0. 5 to 0. 8 ng/m. L; may be associated with mortality, whereas higher concentrations may mortality � Post-hoc analysis – risk of death in females with levels > 1. 2 ; likely similar effect in men with elevated digoxin levels but within normal range JAMA 2003; 289: 871 -8.

Case 1 (continued) JK is seeing you at clinic for f/u after recent hospitalization. He reports having to stop due to fatigue when he walks about a block. Current meds: Lisinopril 10 mg po BID Carvedilol 25 mg po BID Furosemide 20 mg po QD Aspirin 81 mg po QD EF 30% BP: 90/67, HR 72 Labs: Scr 1. 5 (stable), K 4. 2 What other meds could you consider adding at this point? Aldosterone antagonist

Aldosterone Antagonists � AHA 2013 � Aldosterone antagonists are recommended in patients with NYHA class II-IV and who have LVEF of 35% or less, unless contraindicated, to reduce morbidity and mortality. Patients with NYHA class II should have a history of prior cardiovascular hospitalization or elevated plasma natriuretic peptide levels to be considered for AA. Scr ≤ 2. 5 in men or ≤ 3 in women and K < 5 m. Eq/L. (Class I, Level A). � ESC 2012 � Recommended for all patients with persisting symptoms (NYHA II-IV) and an EF ≤ 35%, despite treatment with ACE inhibitor and a beta-blocker (Class I, Level A) Circulation 2013; 128: e 240 -327. Eur H J 2012; 33: 1787 -1847.
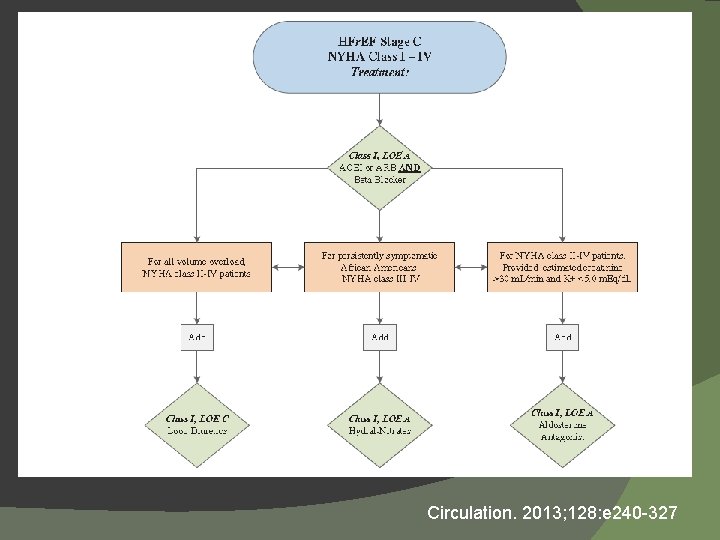
Circulation. 2013; 128: e 240 -327
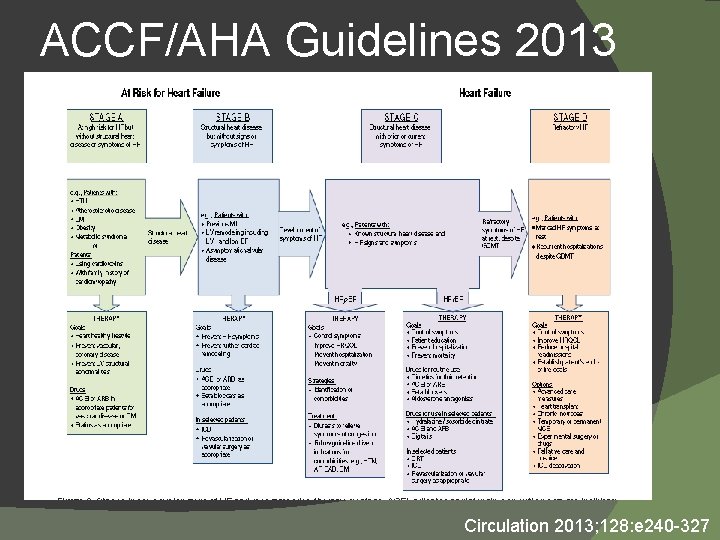
ACCF/AHA Guidelines 2013 Circulation 2013; 128: e 240 -327

Decompensated Heart Failure
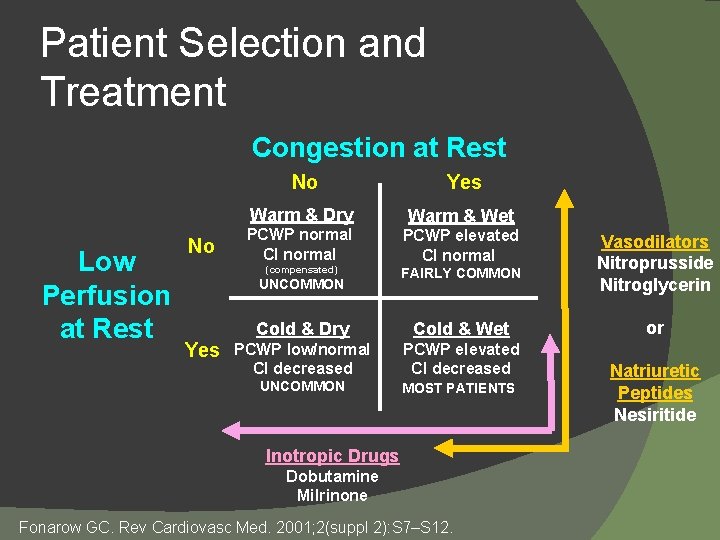
Patient Selection and Treatment Congestion at Rest Low Perfusion at Rest No No Yes Warm & Dry Warm & Wet PCWP normal CI normal PCWP elevated CI normal (compensated) UNCOMMON Yes FAIRLY COMMON Cold & Dry Cold & Wet PCWP low/normal CI decreased PCWP elevated CI decreased UNCOMMON MOST PATIENTS Inotropic Drugs Dobutamine Milrinone Fonarow GC. Rev Cardiovasc Med. 2001; 2(suppl 2): S 7–S 12. Vasodilators Nitroprusside Nitroglycerin or Natriuretic Peptides Nesiritide
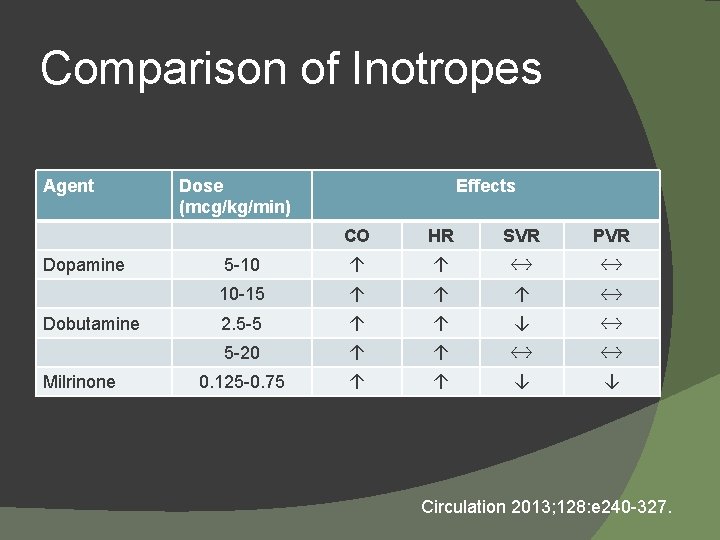
Comparison of Inotropes Agent Dopamine Dobutamine Milrinone Dose (mcg/kg/min) Effects CO HR SVR PVR 5 -10 ↑ ↑ ↔ ↔ 10 -15 ↑ ↑ ↑ ↔ 2. 5 -5 ↑ ↑ ↓ ↔ 5 -20 ↑ ↑ ↔ ↔ 0. 125 -0. 75 ↑ ↑ ↓ ↓ Circulation 2013; 128: e 240 -327.

Dobutamine � Cardiogenic shock, heart failure � Mechanism • Selective beta-2 agonist, ↑CO and HR � Dosing • 1 -20 mcg/kg/min • Dose > 10 mcg/kg/min rarely used � Considerations • Dose-related tachyarrhythmias • ↑ m. VO 2 – caution in ischemia, CAD pts

Milrinone � � “Inodilator” MOA • Phosphodiesterase inhibitor - ↑c. AMP levels leading to smooth muscle relaxation and increased cardiac contractility � Dosing • 0. 25 – 0. 75 mcg/kg/min � Considerations • Accumulates in renal dysfunction • Clinically significant hypotension – avoid loading • Good alternative to dobutamine in HF patients with ↑ PA pressures • Commonly used with vasopressin for milrinoneinduced hypotension

Nitroprusside �Direct active arterial vasodilator �Dosing � 0. 25 -5 mcg/kg/min (max 10 mcg/kg/min) �Half life 3 -5 minutes, fast onset 1 -10 mins �Main uses �Hypertension �Aortic dissection (use with beta-blocker) �CHF w/ “elevated” BP �Considerations �Renal insufficiency – accumulation of thiocyanate �Liver insufficiency – accumulation of cyanide �May increase ICP

Nitroglycerin �Primary role in pts with ACS, may be used in heart failure �MOA �Relaxation of smooth muscle �Dosing � 0. 05 -4 mcg/kg/min, can be titrated quickly for chest pain �Considerations �Tolerance occurs within 24 hrs �Headache �Rebound tachycardia

Nesiritide � Targeted for ADHF patients � MOA – numerous (see next slide) � Dosing • 0. 005 -0. 02 mcg/kg/min • Generally avoid bolus dosing � Considerations • Synergy with other vasodilators • Used in combination with inotropes

Nesiritide � Hemodynamic: • Vasodilation of arteries, veins, and coronaries • Preload and Cardiac Output • Lusitropy � Neuroendocrine: • Counter-regulatory effect on RAAS, Aldosterone • SNS activity, Endothelin � Renal: • GFR, Diuresis and Natriuresis Levin ER et al, NEJM 1998; Venugopal J, Journal Clinical Pharmacy and Therapeutics 2001

Other controversies in heart failure

Case 2 DP is a 40 yo male with non ischemic cardiomyopathy (EF of 18%), no atrial arrhythmias. The attending asked if you would like to start DP on Warfarin to prevent embolic events such as a stroke. What is your opinion on starting Warfarin? Yes, warfarin should be started. B. No, start Aspirin instead. C. No, start Clopidogrel instead. D. No, do not start any anticoagulant or antiplatelet agent. A.

Rationale � Heart failure is associated with hypercoagulability, formation of left ventricular thrombus, and stroke. � Associated with sudden death and progressive heart failure that may be due to atherothrombotic events. NEJM 2012; 366: 1859 -69.

Warfarin vs Aspirin vs Clopidogrel � WATCH trial �Randomized to open-label warfarin (inr 2. 5 - 3) or double blind Aspirin 162 mg or Clopidogrel 75 mg �N= 1587 patients with EF ≤ 35% and normal sinus rhythm �No difference in time to first occurrence of death, nonfatal MI, or nonfatal stroke. �Warfarin pts had fewer strokes ○ More major and minor bleeds than pts on Clopidogrel. Circulation 2009; 119: 1616 -24.

WARCEF � EF ≤ 35% and normal sinus rhythm � Randomized to warfarin INR goal 2. 75 (2 -3. 5) or Aspirin 325 mg a day � Outcome �Time to ischemic stroke, intracerebral hemorrhage, or death from any cause NEJM 2012; 366: 1859 -69.
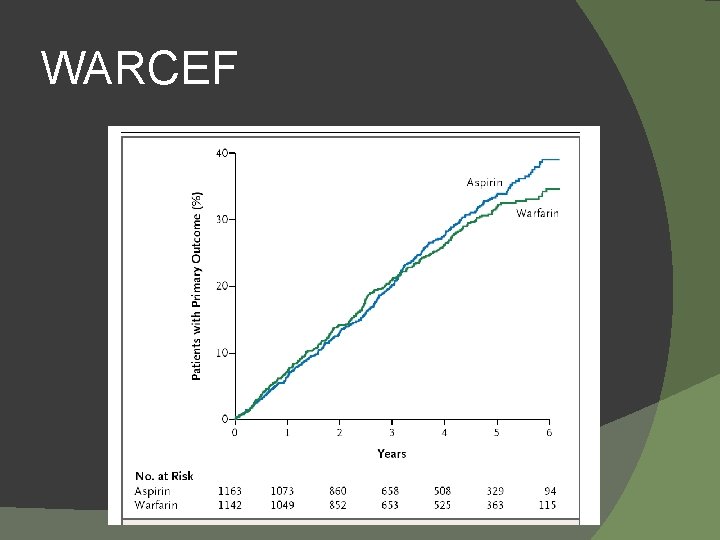
WARCEF
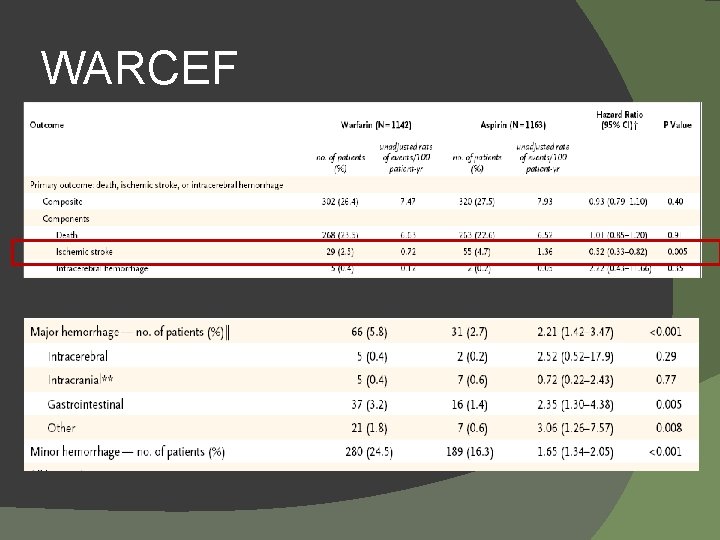
WARCEF

Anticoagulation in Heart Failure � ACC/AHA guidelines 2013 � Pts with chronic HF with permanent/persistent/paroxysmal AF and an additional risk factor for cardioembolic stroke (HTN, DM, prior TIA/stroke, or ≥ 75 yo) should receive chronic anticoagulant therapy (Class I, level A). � Reasonable for chronic HF with AF and no additional factor (Class IIa, level B). � ESC guidelines 2012 � Other than in patients with AF, there is no evidence that an oral anticoagulant reduces morbiditymortality compared with placebo or aspirin.

Case 2 DP is a 40 yo male with non ischemic cardiomyopathy (EF of 18%), no atrial arrhythmias. The attending asked if you would like to start DP on Warfarin to prevent embolic events such as a stroke. What is your opinion on starting Warfarin? Yes, warfarin should be started. B. No, start Aspirin instead. C. No, start Clopidogrel instead. D. No, do not start any anticoagulant or antiplatelet agent. A.

Case 3 � MJ is a 55 yo patient dobutaminedependent who has chronic atrial fibrillation managed with Amiodarone. The resident suggests switching to Dronedarone since it does not have the same long-term adverse effects as Amiodarone. Is this a good idea?
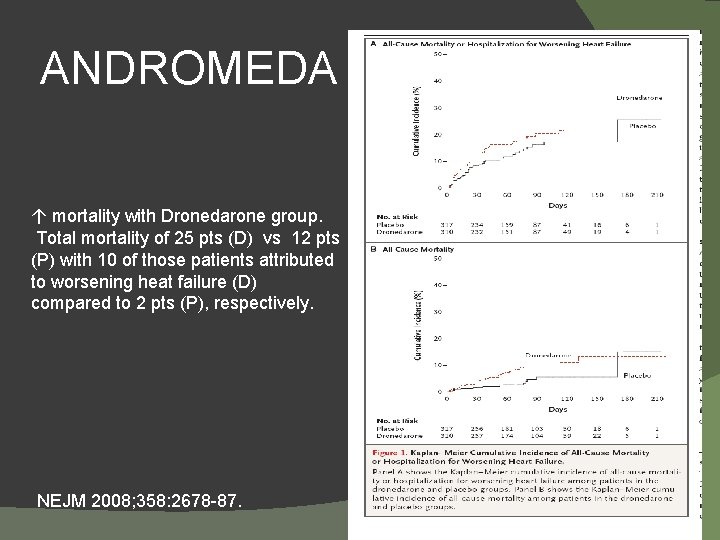
ANDROMEDA ↑ mortality with Dronedarone group. Total mortality of 25 pts (D) vs 12 pts (P) with 10 of those patients attributed to worsening heat failure (D) compared to 2 pts (P), respectively. NEJM 2008; 358: 2678 -87.

Vasopressin Antagonists � Should we use them in hyponatremia and when? � Tolvaptan (Samsca) �Bind to V 2 receptors of distal nephron selectively �Common adverse effects- thirst and dry mouth
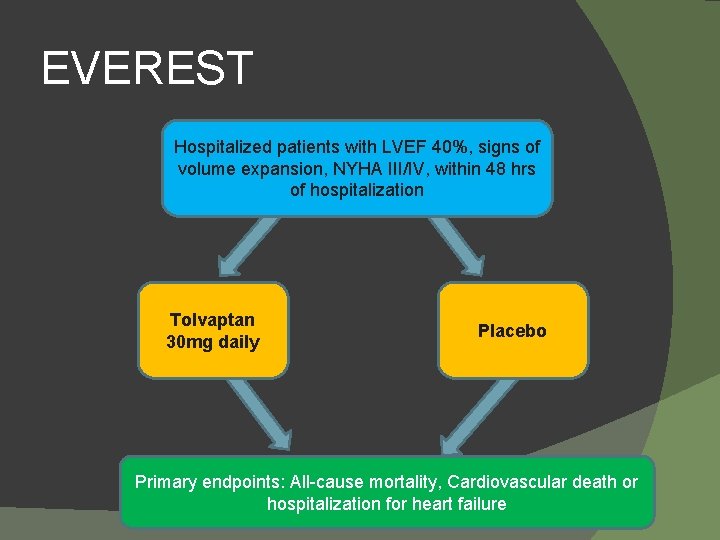
EVEREST Hospitalized patients with LVEF 40%, signs of volume expansion, NYHA III/IV, within 48 hrs of hospitalization Tolvaptan 30 mg daily Placebo Primary endpoints: All-cause mortality, Cardiovascular death or hospitalization for heart failure

EVEREST Results No significant differences in primary outcomes. � Tolvaptan pts had greater change in edema at 7 days (or discharge). � Among pts with baseline Na< 134 meq/L, mean Na increased by 5. 49 meq/L by day 7 (or discharge) with Tolvaptan vs 1. 85 meq/L (placebo). � Safety: � �Hypernatremia 1. 7% (tolvaptan) vs 0. 5% (placebo) JAMA 2007; 297: 1319 -31.

Diuretic Strategies in Patients with Acute Decompensated Heart Failure- DOSE trial � How much and what mode? � Presented within 24 hrs with ADHF � On oral diuretic of at least 1 month at dose of Furosemide 80 -240 mg/day or equivalent. � Excluded pts with SCr > 3 mg/d. L, IV vasodilators, inotropes (except Digoxin) � Randomized to Low-Dose, High-Dose, IV Bolus, or Continuous IV Infusion NEJM 2011; 364: 797 -805.
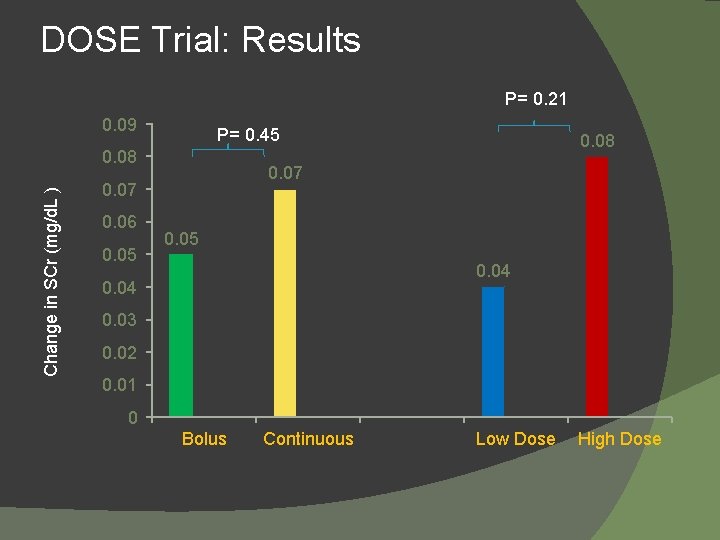
DOSE Trial: Results P= 0. 21 0. 09 P= 0. 45 Change in SCr (mg/d. L ) 0. 08 0. 07 0. 06 0. 05 0. 08 0. 05 0. 04 0. 03 0. 02 0. 01 0 Bolus Continuous Low Dose High Dose
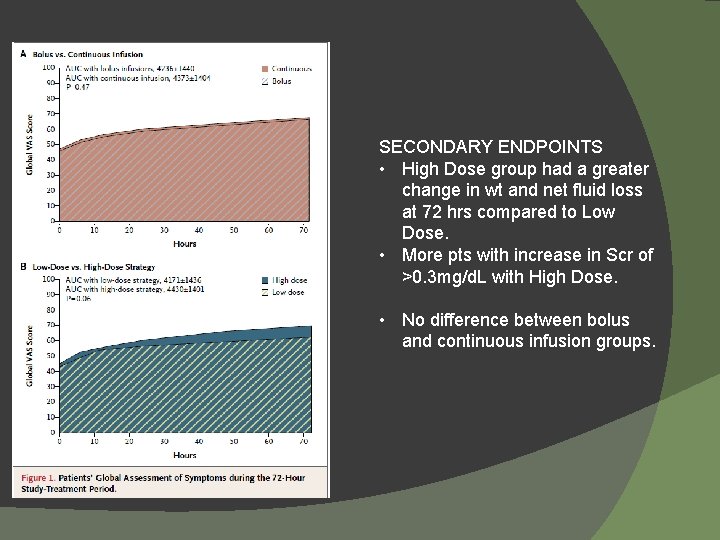
SECONDARY ENDPOINTS • High Dose group had a greater change in wt and net fluid loss at 72 hrs compared to Low Dose. • More pts with increase in Scr of >0. 3 mg/d. L with High Dose. • No difference between bolus and continuous infusion groups.

On the Horizon� Ivabradine (approved in Europe) �Slows heart rate by selective If current inhibition in sinus node �Reduced hospitalization in SHIFT trial �Most beneficial in patients with baseline HR ≥ 75 bpm in post-hoc analysis. � Serelaxin � Angiotensin receptor neprilysin inhibitor � Omecamtiv mecarbil Lancet 2010; 376: 886 -94.

- Slides: 72