Be respectful no electronics please Announcements n Textbook

Be respectful – no electronics please! Announcements n Textbook homework tomorrow in discussion n Lab due tomorrow in discussion Lab again on Monday – Precipitation Reactions n n n Solutions manuals available for use in Chem Learning Center (2021 Chem Annex) Bring headphones or earbuds! Stoichiometry Workshop Tuesday March 12 Exam 2 on Thursday March 14

Solubility Rules (Page 167) 1. 2. 3. 4. 5. 6. Most nitrate salts are soluble. Most salts of sodium, potassium, and ammonium cations are soluble. Most chloride salts are soluble. Exceptions: Ag+ and Pb 2+. Most sulfate salts are soluble. Exceptions: Ca 2+, Ba 2+, and Pb 2+. Most hydroxide salts are only slightly soluble. Soluble ones are: Na+, K+, and Ca 2+. Most sulfide, carbonate, and phosphate salts are only slightly soluble.

Special Case #1 The one where it’s not on the solubility rules. 1. 2. 3. 4. 5. 6. Most nitrate salts are soluble. Most salts of sodium, potassium, and ammonium cations are soluble. Most chloride salts are soluble. Exceptions: Ag+ and Pb 2+. Most sulfate salts are soluble. Exceptions: Ca 2+, Ba 2+, and Pb 2+. Most hydroxide salts are only slightly soluble. Soluble ones are: Na+, K+, and Ca 2+. Most sulfide, carbonate, and phosphate salts are only slightly soluble.

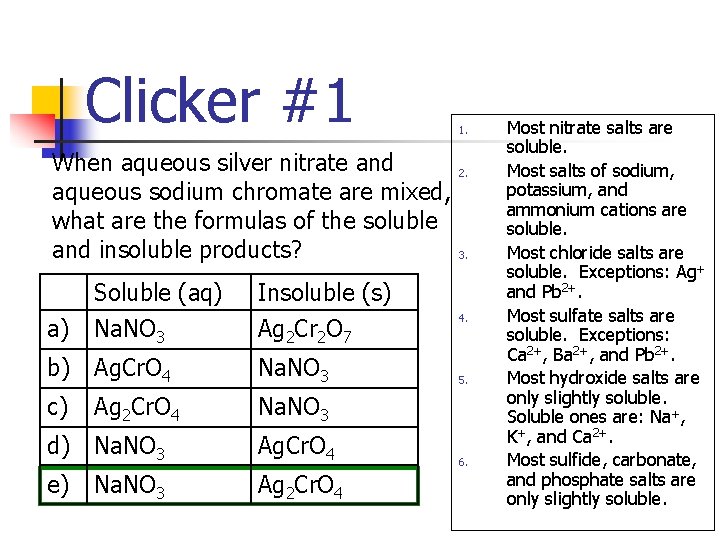
Clicker #1 When aqueous silver nitrate and aqueous sodium chromate are mixed, what are the formulas of the soluble and insoluble products? Soluble (aq) 1. 2. 3. Insoluble (s) a) Na. NO 3 Ag 2 Cr 2 O 7 b) Ag. Cr. O 4 Na. NO 3 c) Ag 2 Cr. O 4 Na. NO 3 d) Na. NO 3 Ag. Cr. O 4 e) Na. NO 3 Ag 2 Cr. O 4 4. 5. 6. Most nitrate salts are soluble. Most salts of sodium, potassium, and ammonium cations are soluble. Most chloride salts are soluble. Exceptions: Ag+ and Pb 2+. Most sulfate salts are soluble. Exceptions: Ca 2+, Ba 2+, and Pb 2+. Most hydroxide salts are only slightly soluble. Soluble ones are: Na+, K+, and Ca 2+. Most sulfide, carbonate, and phosphate salts are only slightly soluble.

Talk About It: What is incorrect about each of the net ionic equations shown below? 1) Ag 2+(aq) + Cr. O 4 -2(aq) Ag 2 Cr. O 4(s) 2) Ag 2+(aq) + Cr. O 4 -2(aq) Ag 2 Cr. O 4(s) 3) Ag+(aq) + Cr. O 4 -2(aq) Ag. Cr. O 4(s)

Clicker #2 n When potassium chloride and sodium nitrate are mixed, what precipitate will form? KCl(aq) + Na. NO 3(aq) → A) B) C) D) E) KNO 3 Na. Cl KCl Na. NO 3 No precipitate will form. n n § § Solubility Rules Most nitrate salts are soluble. Most salts of sodium, potassium, and ammonium cations are soluble. Most chloride salts are soluble. Exceptions: Ag+ and Pb 2+. Most sulfate salts are soluble. Exceptions: Ca 2+, Ba 2+, and Pb 2+. Most hydroxide salts are only slightly soluble. Soluble ones are: Na+, K+, and Ca 2+. Most sulfide, carbonate, and phosphate salts are only slightly soluble.

Special Case #2 The one where nothing happens. 1. 2. 3. 4. 5. 6. Most nitrate salts are soluble. Most salts of sodium, potassium, and ammonium cations are soluble. Most chloride salts are soluble. Exceptions: Ag+ and Pb 2+. Most sulfate salts are soluble. Exceptions: Ca 2+, Ba 2+, and Pb 2+. Most hydroxide salts are only slightly soluble. Soluble ones are: Na+, K+, and Ca 2+. Most sulfide, carbonate, and phosphate salts are only slightly soluble.

Special Case #3 The one where it looks like nothing happens, but something actually does happen. 1. 2. 3. 4. 5. 6. Most nitrate salts are soluble. Most salts of sodium, potassium, and ammonium cations are soluble. Most chloride salts are soluble. Exceptions: Ag+ and Pb 2+. Most sulfate salts are soluble. Exceptions: Ca 2+, Ba 2+, and Pb 2+. Most hydroxide salts are only slightly soluble. Soluble ones are: Na+, K+, and Ca 2+. Most sulfide, carbonate, and phosphate salts are only slightly soluble.
Acids & Bases

Clicker #3 – From the homework What acid and base must have reacted to produce these substances? _____ + _____ K 2 SO 4(aq) + 2 H 2 O(l) a) b) c) d) HSO 4 and KOH H 2 SO 4 and K 2 OH H 2 SO 4 and K 2 O

Solubility Rules (Page 127) 1. 2. 3. 4. 5. 6. Most nitrate salts are soluble. Most salts of sodium, potassium, and ammonium cations are soluble. Most chloride salts are soluble. Exceptions: Ag+ and Pb 2+. Most sulfate salts are soluble. Exceptions: Ca 2+, Ba 2+, and Pb 2+. Most hydroxide salts are only slightly soluble. Soluble ones are: Na+, K+, and Ca 2+. Most sulfide, carbonate, and phosphate salts are only slightly soluble.

Special Case #4 The one everything where happens. 1. 2. 3. 4. 5. 6. Most nitrate salts are soluble. Most salts of sodium, potassium, and ammonium cations are soluble. Most chloride salts are soluble. Exceptions: Ag+ and Pb 2+. Most sulfate salts are soluble. Exceptions: Ca 2+, Ba 2+, and Pb 2+. Most hydroxide salts are only slightly soluble. Soluble ones are: Na+, K+, and Ca 2+. Most sulfide, carbonate, and phosphate salts are only slightly soluble.

Toxicity of Barium Ions Marie Robards, Fort Worth Texas, 1993
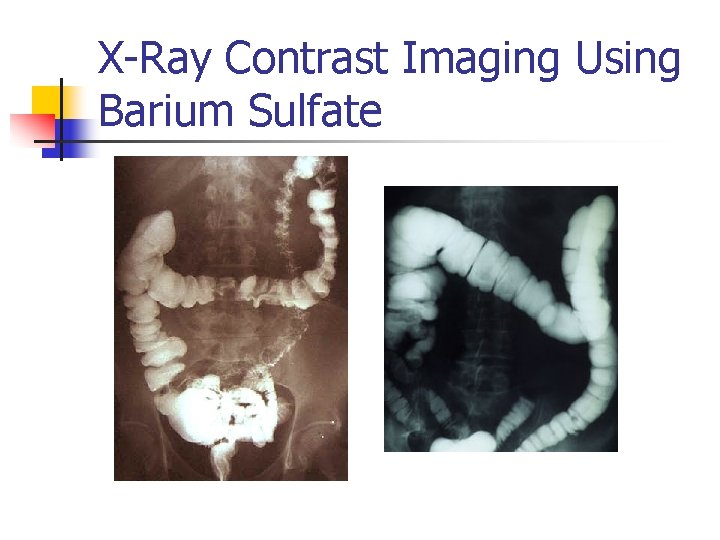
X-Ray Contrast Imaging Using Barium Sulfate

Clicker #4 What are the spectator ions in this reaction? A) B) C) D) E) H+, OHBa+2, SO 4 -2 H+ only Ba+2, SO 4 -2, H+, OHThere are no spectator ions.

Solubility Rules (Page 127) 1. 2. 3. 4. 5. 6. Most nitrate salts are soluble. Most salts of sodium, potassium, and ammonium cations are soluble. Most chloride salts are soluble. Exceptions: Ag+ and Pb 2+. Most sulfate salts are soluble. Exceptions: Ca 2+, Ba 2+, and Pb 2+. Most hydroxide salts are only slightly soluble. Soluble ones are: Na+, K+, and Ca 2+. Most sulfide, carbonate, and phosphate salts are only slightly soluble.

Stay positive… If you’re not part of the solution, you’re part of the precipitate.
- Slides: 19