BDSI INVESTOR OVERVIEW A Rapidly Growing Commercial Stage

BDSI INVESTOR OVERVIEW A Rapidly Growing Commercial Stage Specialty Pharmaceutical Company Dedicated to Patients Living with Chronic Pain January 2019 © 2018 Bio. Delivery Sciences International, Inc. All Rights Reserved. January 2019 |

Forward Looking Statements Certain statements contained in this presentation or in other documents of Bio. Delivery Sciences International, Inc. (the “Company”), along with certain statements that may be made by management of the Company orally in presenting this material, may contain “forward-looking statements” as defined in the Private Securities Litigation Reform Act of 1995. These statements can be identified by the fact that they do not relate strictly to historic or current facts. They use words such as "estimate, " "expect, " "intend, " "believe, " "plan, " "anticipate, " “projected” and other words and terms of similar meaning in connection with any discussion of future operating or financial performance or condition. These statements are based upon the current beliefs and expectations of the Company's management and are subject to significant risks and uncertainties. Statements regarding future action, future performance and/or future results including, without limitation, those relating to the timing for completion, and results of, scheduled or additional clinical trials and the FDA’s or other regulatory review and/or approval and commercial launch and sales results (if any) of the Company’s formulations and products and regulatory filings related to the same, and receipt by the Company of milestone and royalty payments may differ from those set forth in the forward-looking statements. Peak sales and market size estimates have been determined on the basis of market research and comparable product analysis, but no assurances can be given that such sales levels will be achieved, if at all, or that such market size estimates will prove accurate. Because actual results are affected by these and other potential risks, contingencies and uncertainties, the Company cautions investors that actual results may differ materially from those expressed or implied in any forward-looking statement. It is not possible to predict or identify all such risks, contingencies and uncertainties. The Company identifies some of these factors in its Securities and Exchange Commission (“SEC”) filings on Forms 10 -K, 10 -Q and 8 -K, and investors are advised to consult the Company’s filings for a more complete listing of risk factors, contingencies and uncertainties effecting the Company and its business and financial performance. The Company assumes no obligation to update forward-looking statements as circumstances change. Investors are advised to consult further disclosures that the Company makes or has made on related subjects in the Company's Form 10 -K, 10 -Q and 8 -K reports. In presenting this material or responding to inquiries in connection with a presentation, management may refer to results, projections or performance measures that are not prepared in accordance with U. S. Generally Accepted Accounting Principles (“GAAP”) as reported in the Company’s SEC filings. These results, projections or performance measures are Non-GAAP measures and are not intended to replace or as a substitute for results measured under GAAP, but rather as supplement to the GAAP reported results. January 2019 | 2

Bio. Delivery Sciences (NASDAQ: BDSI) Significant Near Term Growth Potential A rapidly growing commercial stage specialty pharmaceutical company dedicated to patients living with chronic pain: • Portfolio of approved commercialized products • Capitalizing on the growth potential of BELBUCA® • Solid financial position January 2019 | 3

Company Fundamentals Accelerating BELBUCA Growth Organizational Development Financial Position • The first and only approved CIII long-acting opioid for the treatment of chronic pain utilizing buccal film technology • Addresses a significant unmet need in the management of chronic pain • Highly experienced senior management team in place • Reconstituted Board of exceptionally qualified independent directors • Cash & Equivalents: $49. 5 M (as of September 30, 2018) • Shares Outstanding: 70. 7 M shares (as of November 8, 2018) January 2019 | 4
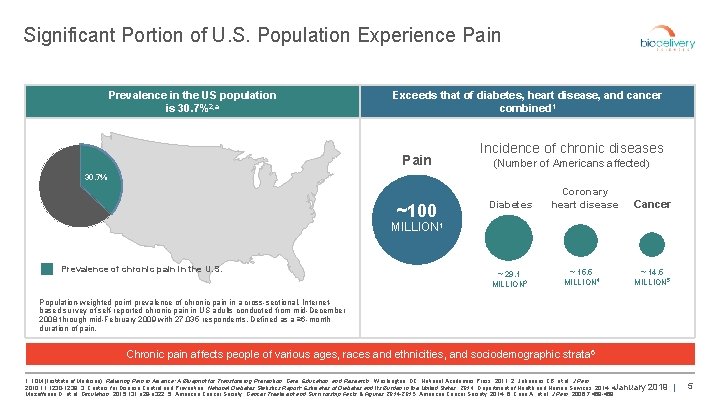
Significant Portion of U. S. Population Experience Pain Prevalence in the US population is 30. 7%2, a Exceeds that of diabetes, heart disease, and cancer combined 1 Pain Incidence of chronic diseases (Number of Americans affected) 30. 7% Diabetes ~100 Coronary heart disease Cancer MILLION 1 Prevalence of chronic pain in the U. S. ~ 29. 1 MILLION 3 ~ 15. 5 MILLION 4 ~ 14. 5 MILLION 5 Population-weighted point prevalence of chronic pain in a cross-sectional, Internetbased survey of self-reported chronic pain in US adults conducted from mid-December 2008 through mid-February 2009 with 27, 035 respondents. Defined as a ≥ 6 -month duration of pain. Chronic pain affects people of various ages, races and ethnicities, and sociodemographic strata 6 1. IOM (Institute of Medicine). Relieving Pain in America: A Blueprint for Transforming Prevention, Care, Education, and Research. Washington, DC: National Academies Press; 2011; 2. Johannes CB, et al. J Pain. 2010; 11: 1230 -1239; 3. Centers for Disease Control and Prevention. National Diabetes Statistics Report: Estimates of Diabetes and Its Burden in the United States, 2014. Department of Health and Human Services; 2014; 4. January 2019 | Mozaffarian D, et al. Circulation. 2015; 131: e 29 -e 322; 5. American Cancer Society. Cancer Treatment and Survivorship Facts & Figures 2014 -2015. American Cancer Society; 2014; 6. Cano A, et al. J Pain. 2006; 7: 459 -468. 5
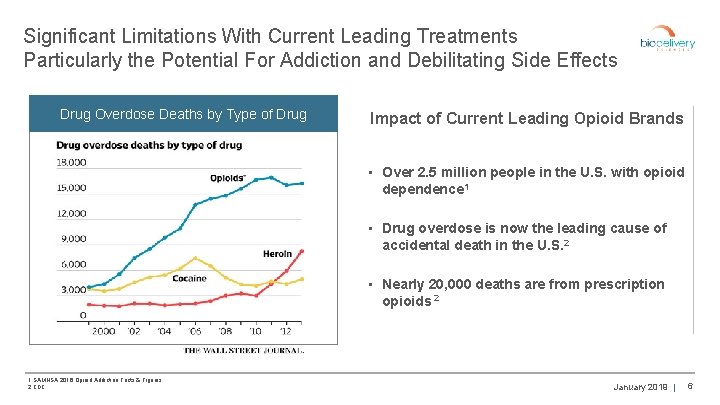
Significant Limitations With Current Leading Treatments Particularly the Potential For Addiction and Debilitating Side Effects Drug Overdose Deaths by Type of Drug Impact of Current Leading Opioid Brands • Over 2. 5 million people in the U. S. with opioid dependence 1 • Drug overdose is now the leading cause of accidental death in the U. S. 2 • Nearly 20, 000 deaths are from prescription opioids 2 1 SAMHSA 2016 Opioid Addiction Facts & Figures 2 CDC January 2019 | 6
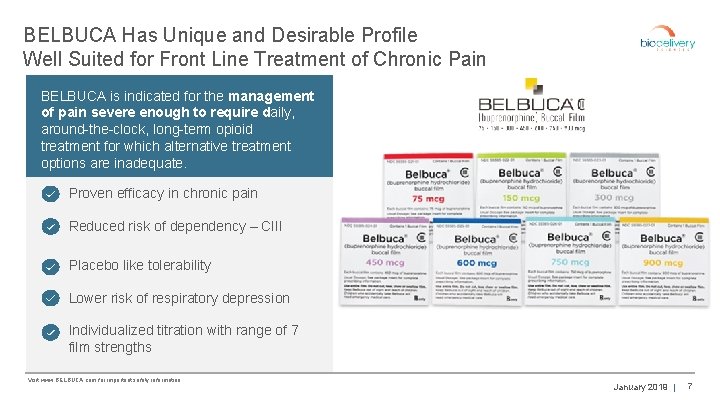
BELBUCA Has Unique and Desirable Profile Well Suited for Front Line Treatment of Chronic Pain BELBUCA is indicated for the management of pain severe enough to require daily, around-the-clock, long-term opioid treatment for which alternative treatment options are inadequate. Proven efficacy in chronic pain Reduced risk of dependency – CIII Placebo like tolerability Lower risk of respiratory depression Individualized titration with range of 7 film strengths Visit www. BELBUCA. com for important safety information. January 2019 | 7

BELBUCA® Growth Opportunity Well Positioned to Meet the Needs of Chronic Pain Patients • Highly effective with proven sustained pain relief • Unique delivery system that bypasses the first-pass metabolism in the gastrointestinal tract • Exclusivity through 2027 and high barriers to entry • Prevalence of chronic pain is growing • Significant long term market opportunity > $200 M 1 https: //www. ncbi. nlm. nih. gov/books/NBK 92516/ January 2019 | 8
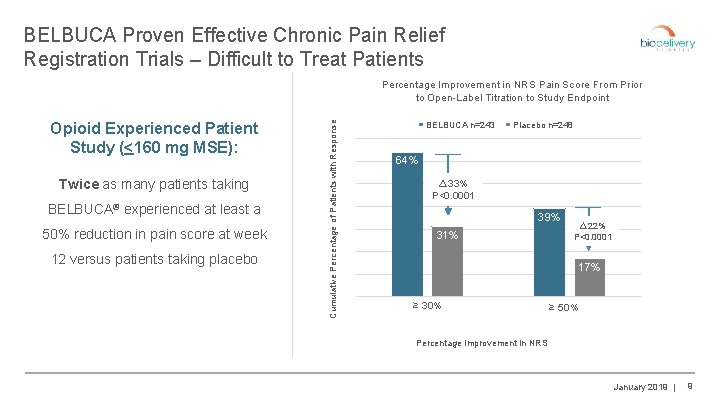
BELBUCA Proven Effective Chronic Pain Relief Registration Trials – Difficult to Treat Patients Opioid Experienced Patient Study (<160 mg MSE): Twice as many patients taking BELBUCA® experienced at least a 50% reduction in pain score at week 12 versus patients taking placebo Cumulative Percentage of Patients with Response Percentage Improvement in NRS Pain Score From Prior to Open-Label Titration to Study Endpoint BELBUCA n=243 Placebo n=248 64% △ 33% P<0. 0001 39% 31% △ 22% P<0. 0001 17% ≥ 30% ≥ 50% Percentage Improvement in NRS January 2019 | 9
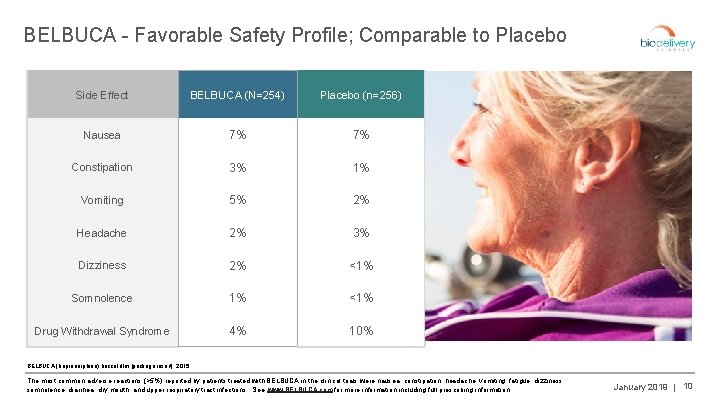
BELBUCA - Favorable Safety Profile; Comparable to Placebo Side Effect BELBUCA (N=254) Placebo (n=256) Nausea 7% 7% Constipation 3% 1% Vomiting 5% 2% Headache 2% 3% Dizziness 2% <1% Somnolence 1% <1% Drug Withdrawal Syndrome 4% 10% BELBUCA (buprenorphine) buccal film [package insert]. 2015. The most common adverse reactions (>5%) reported by patients treated with BELBUCA in the clinical trials were nausea, constipation, headache, vomiting, fatigue, dizziness, somnolence, diarrhea, dry mouth, and upper respiratory tract infections. See www. BELBUCA. com for more information including full prescribing information. January 2019 | 10
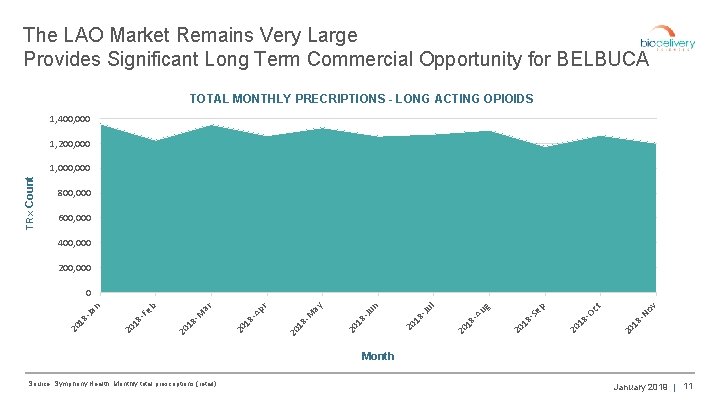
The LAO Market Remains Very Large Provides Significant Long Term Commercial Opportunity for BELBUCA TOTAL MONTHLY PRECRIPTIONS - LONG ACTING OPIOIDS 1, 400, 000 1, 200, 000 TRx Count 1, 000 800, 000 600, 000 400, 000 200, 000 ov 20 18 -N ct 20 18 -O p 18 -S e 18 20 20 Au g l Ju 20 18 - n Ju 20 18 - ay -M 20 18 r Ap 20 18 - ar 20 18 - M b 18 -F e 20 20 18 -Ja n 0 Month Source: Symphony Health, Monthly total prescriptions (retail) January 2019 | 11
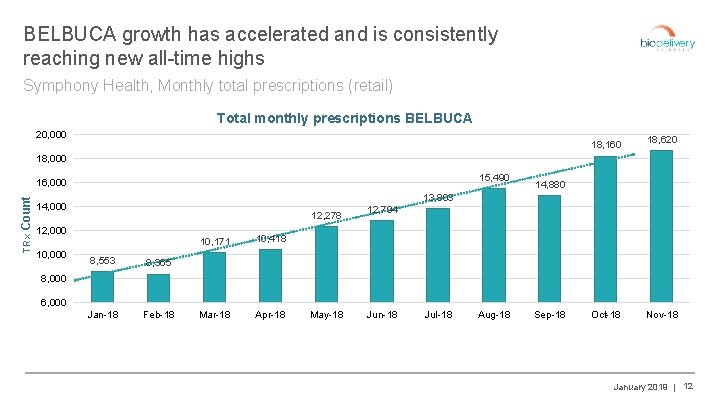
BELBUCA growth has accelerated and is consistently reaching new all-time highs Symphony Health, Monthly total prescriptions (retail) Total monthly prescriptions BELBUCA 20, 000 18, 160 18, 620 18, 000 15, 490 TRx Count 16, 000 13, 803 14, 000 12, 278 12, 000 10, 000 14, 880 8, 553 8, 365 Jan-18 Feb-18 10, 171 10, 418 Mar-18 Apr-18 12, 794 8, 000 6, 000 May-18 Jun-18 Jul-18 Aug-18 Sep-18 Oct-18 Nov-18 January 2019 | 12
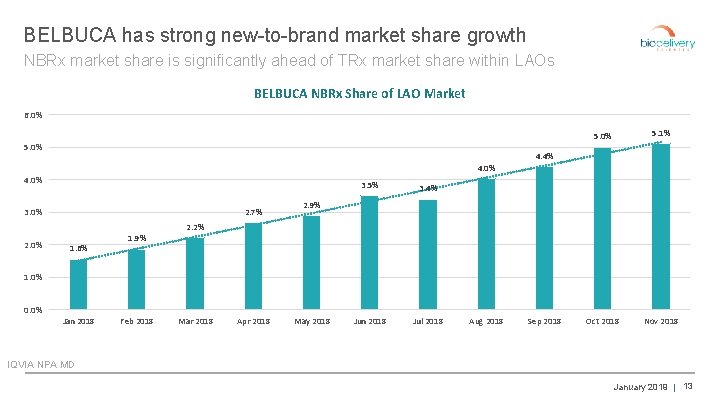
BELBUCA has strong new-to-brand market share growth NBRx market share is significantly ahead of TRx market share within LAOs BELBUCA NBRx Share of LAO Market 6. 0% 5. 1% Oct 2018 Nov 2018 4. 4% 4. 0% 2. 7% 3. 0% 3. 5% 3. 4% Jun 2018 Jul 2018 2. 9% 2. 2% 2. 0% 1. 6% 1. 9% 1. 0% 0. 0% Jan 2018 Feb 2018 Mar 2018 Apr 2018 May 2018 Aug 2018 Sep 2018 IQVIA NPA MD January 2019 | 13
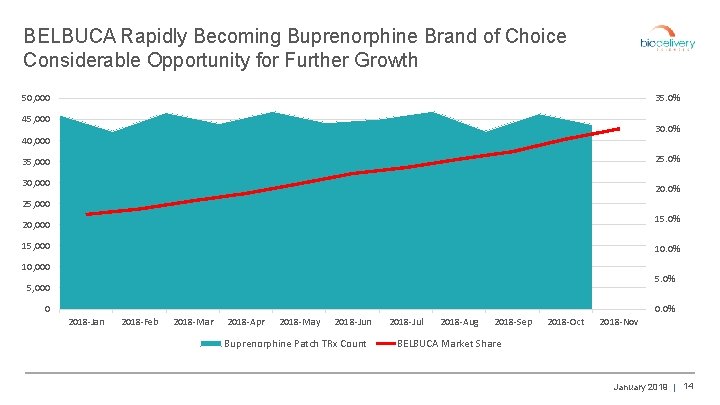
BELBUCA Rapidly Becoming Buprenorphine Brand of Choice Considerable Opportunity for Further Growth 50, 000 35. 0% 45, 000 30. 0% 40, 000 25. 0% 35, 000 30, 000 20. 0% 25, 000 15. 0% 20, 000 15, 000 10. 0% 10, 000 5. 0% 5, 000 0 0. 0% 2018 -Jan 2018 -Feb 2018 -Mar 2018 -Apr 2018 -May 2018 -Jun Buprenorphine Patch TRx Count 2018 -Jul 2018 -Aug 2018 -Sep 2018 -Oct 2018 -Nov BELBUCA Market Share January 2019 | 14
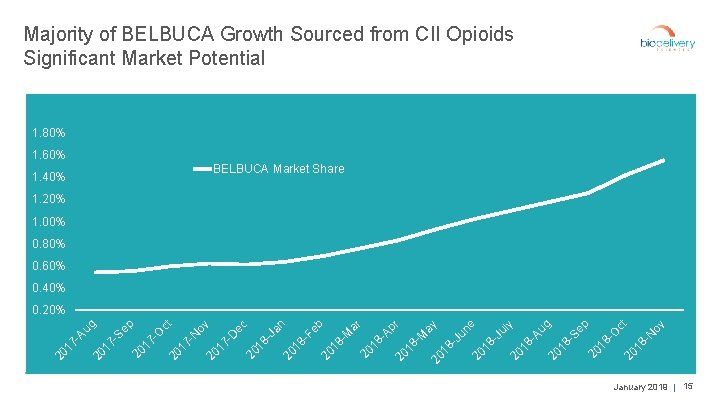
Majority of BELBUCA Growth Sourced from CII Opioids Significant Market Potential 1. 80% 1. 60% BELBUCA Market Share 1. 40% 1. 20% 1. 00% 0. 80% BELBUCA 0. 60% 0. 40% ov -N 18 20 18 -O ct p 20 -S e 18 20 18 -J -A u ul g y e 20 -J un 18 20 20 18 -M ay pr -A 18 20 18 -F -M ar eb n 20 18 -J a ec 20 17 -D ov -N 17 20 20 ct -O 17 20 -S e 17 20 20 17 -A u g p 0. 20% January 2019 | 15
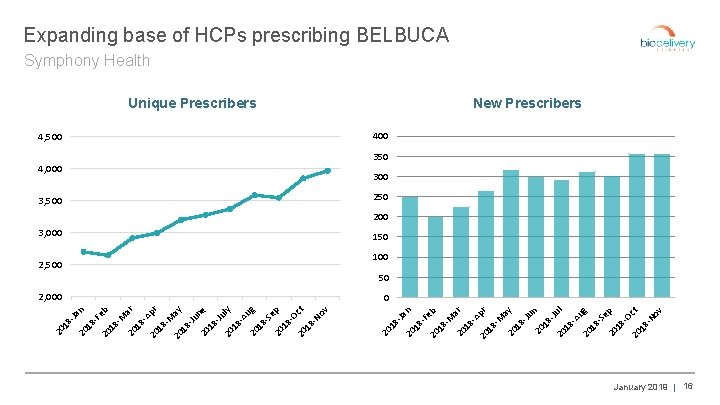
Unique Prescribers pr 20 18 -M ay 20 18 -Ju 20 n 18 -Ju 20 l 18 -A ug 20 18 -S ep 20 18 -O ct 20 18 -N ov ar M 18 -A 20 18 - Fe b 2, 000 20 2, 500 n 3, 000 18 - 3, 500 8 Ja 4, 000 20 4, 500 20 1 20 ar 18 -A p 20 18 r -M a 20 18 y -Ju 20 ne 18 -Ju ly 20 18 -A 20 ug 18 -S ep 20 18 -O ct 20 18 -N ov b Fe 18 -M 20 n 8 Ja 18 - 20 20 1 Expanding base of HCPs prescribing BELBUCA Symphony Health New Prescribers 400 350 300 250 200 150 100 50 0 January 2019 | 16

Expanding Patient Access to BELBUCA is a Key Priority to Further Growth – Remove HCP Obstacles Pull-through formulary wins Maximize Current Coverage Push-through prior authorization obstacles Preferred; no step-through Improve National Coverage Commercial Medicare Part D Regional Payers Expand Regional Coverage State Medicare Blues January 2019 | 17

Drove BELBUCA preferred access to over 100 M patients from 8 M during 2018 1 H 18 2 H 18 Pro. Care. Rx Added: Preferred, no step-through Express Scripts Added: Preferred; no stepthrough United Healthcare Improved: Removal of step-through Optum. Rx Added: Preferred; no step-through CVS/Caremark Improved: Preferred, no step-through, Butrans step-through BELBUCA Envision Added: Preferred; no step-through Humana Added: Preferred/No PA required, no stepthrough Ohio Workers Comp Added: Preferred, no step-through January 2019 | 18
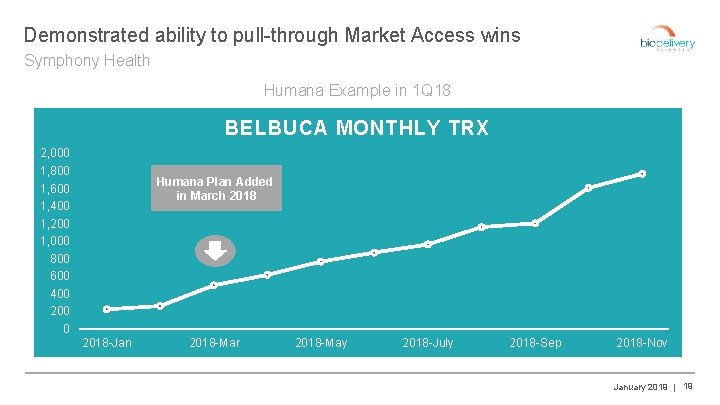
Demonstrated ability to pull-through Market Access wins Symphony Health Humana Example in 1 Q 18 BELBUCA MONTHLY TRX 2, 000 1, 800 1, 600 1, 400 1, 200 1, 000 800 600 400 200 0 Humana Plan Added in March 2018 -Jan 2018 -Mar 2018 -May 2018 -July 2018 -Sep 2018 -Nov January 2019 | 19
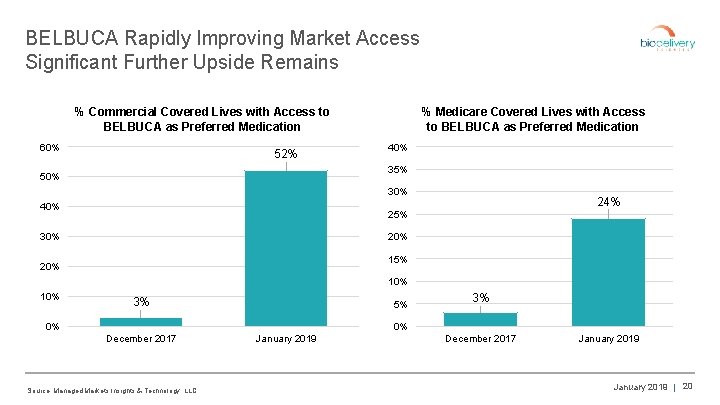
BELBUCA Rapidly Improving Market Access Significant Further Upside Remains % Commercial Covered Lives with Access to BELBUCA as Preferred Medication 60% 52% % Medicare Covered Lives with Access to BELBUCA as Preferred Medication 40% 35% 50% 30% 40% 24% 25% 30% 20% 15% 20% 10% 3% 5% 0% 3% 0% December 2017 Source: Managed Markets Insights & Technology, LLC. January 2019 December 2017 January 2019 | 20

Strategic Plan to Drive Long-Term Sustained BELBUCA Growth Implementation Starting 2 H 2018 Key Growth Levers: Sales Team Expansion Amplify Market Access Status Medical Information Development Consumer Engagement January 2019 | 21

Additional Commercialized Products - Contributing Revenue to Reinvest BUNAVAIL ® Indicated for the treatment of opioid dependency Leveraged our capabilities to capitalize on commercial synergies with BUNAVAIL ® for a focused commercial approach targeting identified healthcare providers ONSOLIS® Indicated for the treatment of breakthrough pain in opioid tolerant patients with cancer Marketed under the brand name BREAKYL™ in the E. U. and PAINKYL in Taiwan Assessing options for U. S. commercialization of ONSOLIS January 2019 | 22

Highly Experienced Executive Leadership Team Herm Cukier CEO Scott Plesha President Ernest De. Paolantonio CFO James Vollins General Counsel, CCO Joseph Lockhart SVP, Operations Thomas Smith, MD CMO January 2019 | 23

Investment Summary – Rapidly Growing and Well Positioned for Sustained Momentum NASDAQ: BDSI • BELBUCA® - The first and only CIII approved drug for the management of chronic pain using buccal technology • Addresses an important medical need in the treatment of chronic pain with a differentiated profile • Market exclusivity through 2027 and high barriers of entry • Strong cash position • Experienced Management Team • Longer term market opportunity > $200 M 1 https: //www. ncbi. nlm. nih. gov/books/NBK 92516/ January 2019 | 24
- Slides: 24