BCOR 011 Lecture 11 Chapter 8 The Flow

BCOR 011 Lecture 11 Chapter 8 The Flow of Energy in a Cell Sept 26, 2005 1 Figure 8. 1

Energy: the capacity to effect change Two types of energy Potential Energy Kinetic Energy -stored in height -energy of movement -stored in battery (conc/charge) -stored in BONDS -molecules colliding, vibrating -HEAT, light 2
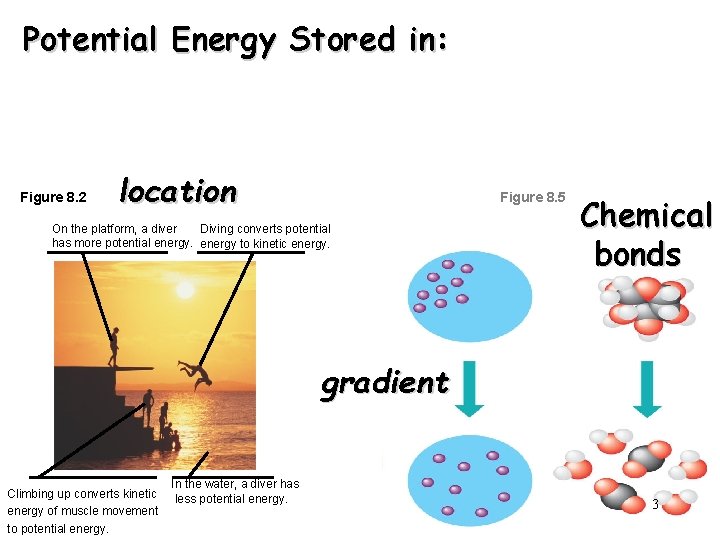
Potential Energy Stored in: Figure 8. 2 location Figure 8. 5 On the platform, a diver Diving converts potential has more potential energy to kinetic energy. Chemical bonds gradient Climbing up converts kinetic energy of muscle movement to potential energy. In the water, a diver has less potential energy. 3
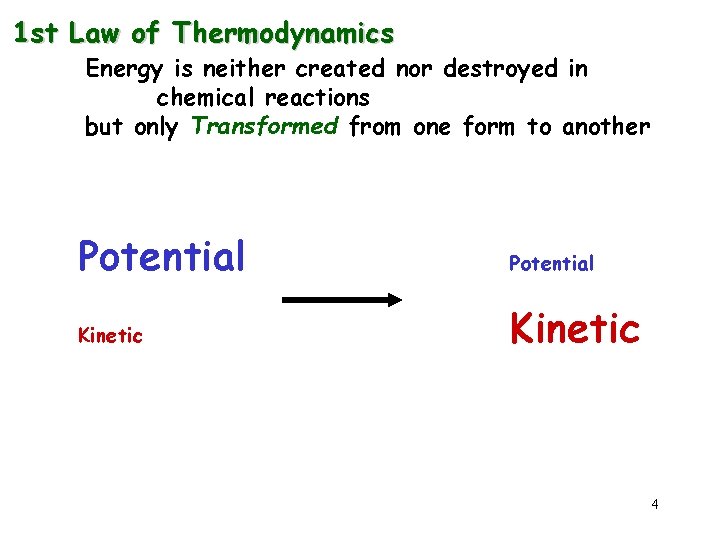
1 st Law of Thermodynamics Energy is neither created nor destroyed in chemical reactions but only Transformed from one form to another Potential Kinetic 4
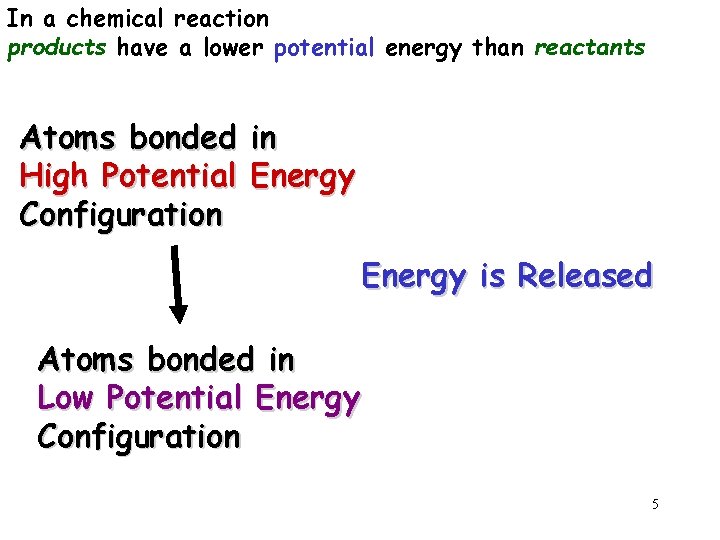
In a chemical reaction products have a lower potential energy than reactants Atoms bonded in High Potential Energy Configuration Energy is Released Atoms bonded in Low Potential Energy Configuration 5
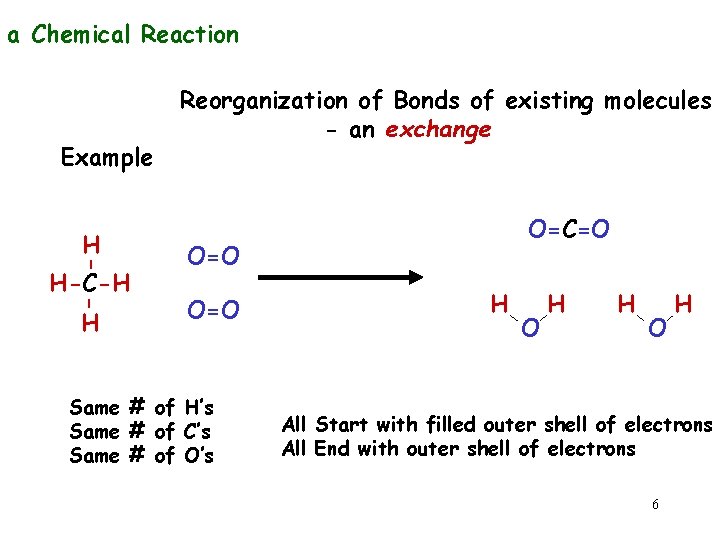
a Chemical Reaction Example - - H H-C-H H Reorganization of Bonds of existing molecules - an exchange O=C=O O=O Same # of H’s Same # of C’s Same # of O’s H O H All Start with filled outer shell of electrons All End with outer shell of electrons 6
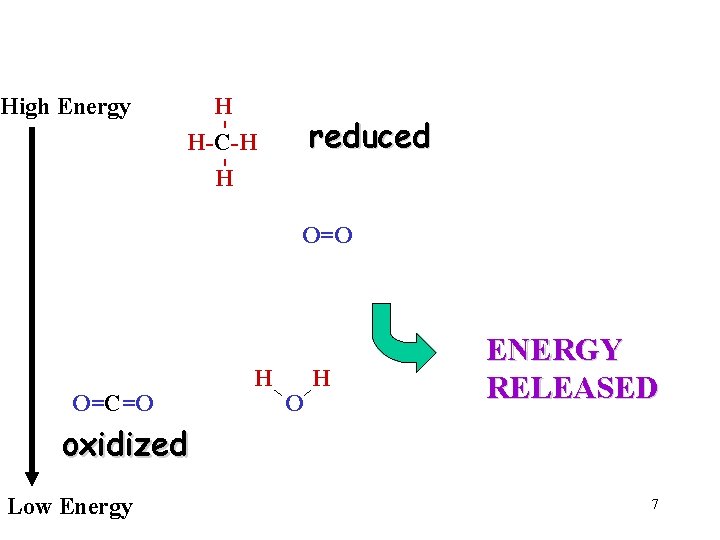
High Energy H H-C-H - reduced H O=O O=C=O H ENERGY RELEASED oxidized Low Energy 7

Energy that is released: Has the capacity to DO WORK Raise potential state of something else Or effect movement – heat, motion 8
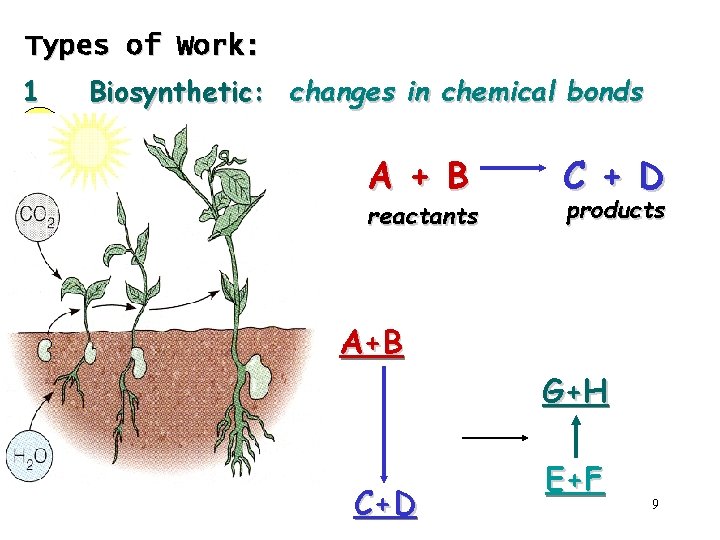
Types of Work: 1 Biosynthetic: changes in chemical bonds A + B reactants C + D products A+B G+H C+D E+F 9
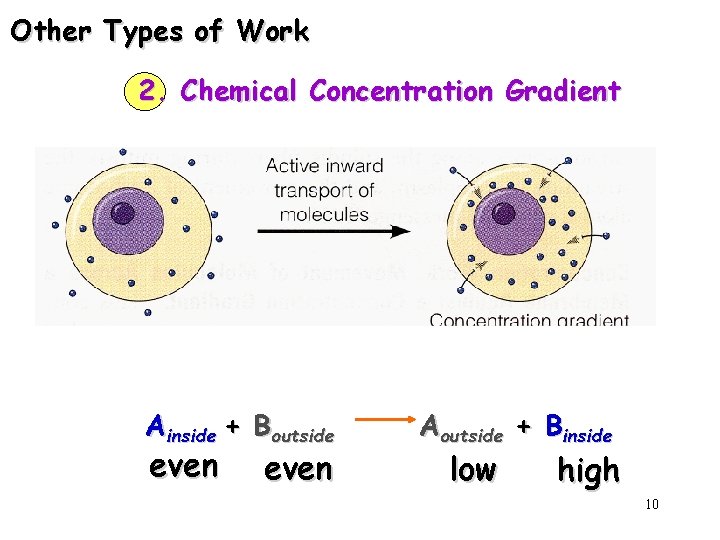
Other Types of Work 2. Chemical Concentration Gradient Ainside + Boutside even Aoutside + Binside low high 10
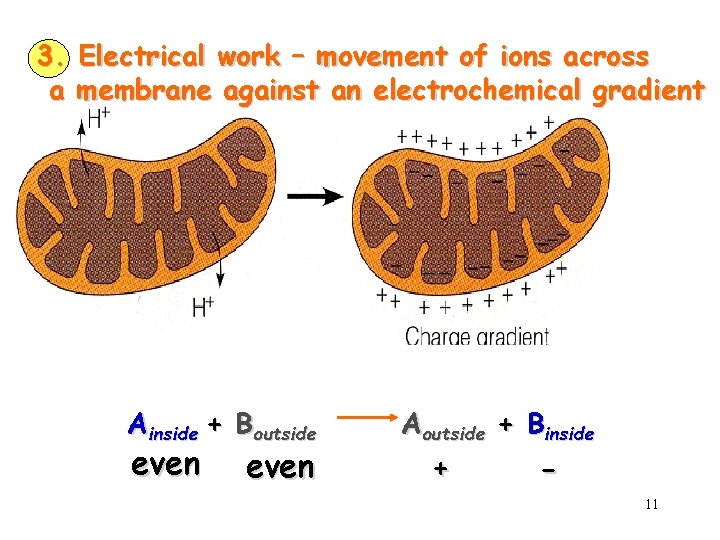
3. Electrical work – movement of ions across a membrane against an electrochemical gradient Ainside + Boutside even Aoutside + Binside + - 11
Other Types of Work 4 Mechanical Work: Movement, Motility 12
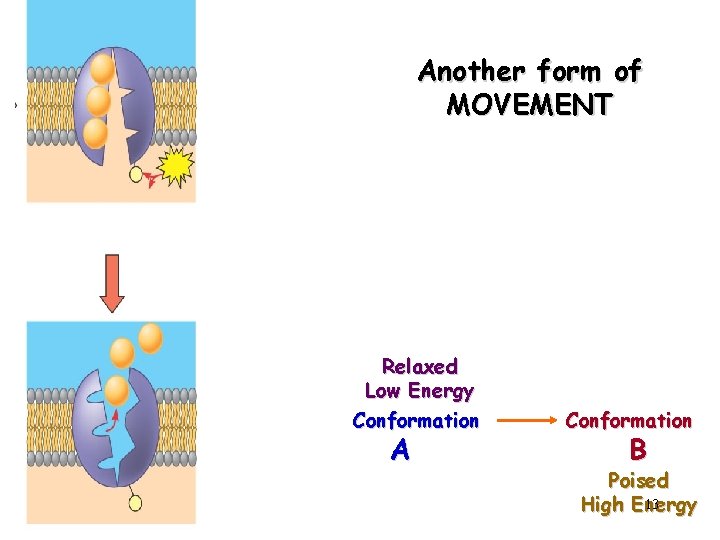
Another form of MOVEMENT Relaxed Low Energy Conformation A Conformation B Poised 13 High Energy

• Some organisms – Convert energy to light, as in bioluminescence Figure 8. 1 14

Energy that is released: Has the capacity to DO WORK Raise potential state of something else Or effect movement – heat, motion But some is always lost to disorder 15
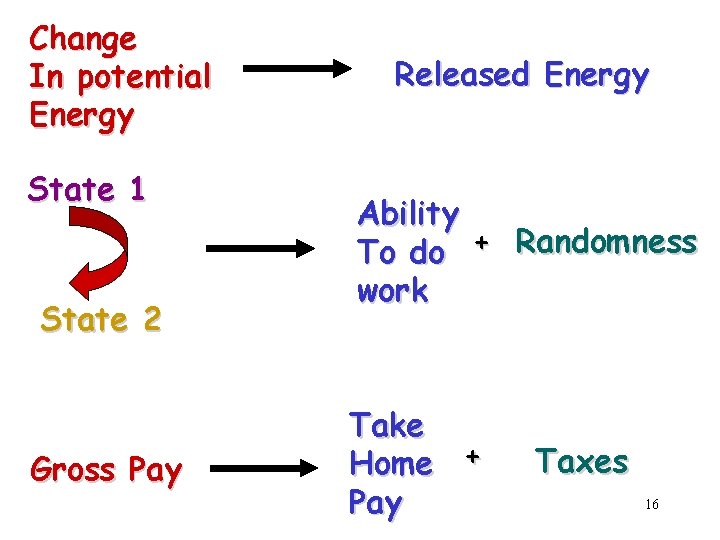
Change In potential Energy State 1 State 2 Gross Pay Released Energy Ability To do + Randomness work Take Home + Pay Taxes 16
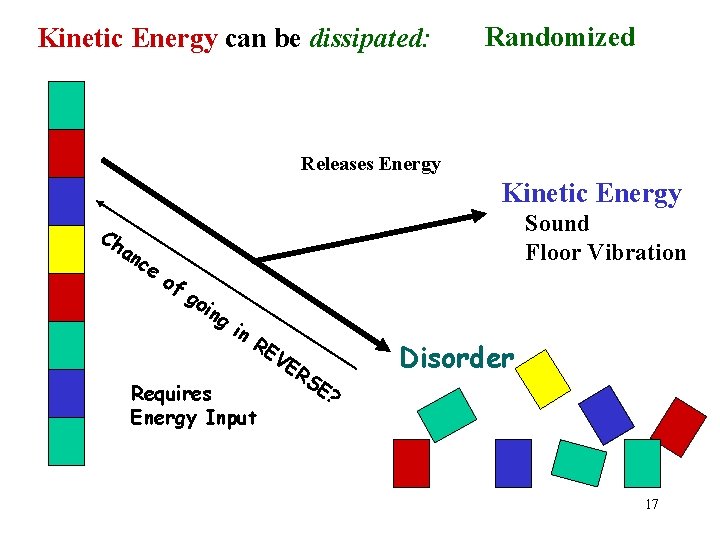
Kinetic Energy can be dissipated: Randomized Releases Energy Kinetic Energy Ch an ce of Sound Floor Vibration go ing in RE V Requires Energy Input Disorder ER SE ? 17

Second law of Thermodynamics: The Universe is proceeding to a State of MAXIMUM DISORDER Only time this is not true is when no movement anymore ie. at abosolute zero 18

0 o K - no motion, no “taxes” A Progressive Scale: Higher the temperature, the more that disorder comes into play higher proportion of energy lost to randomness 19
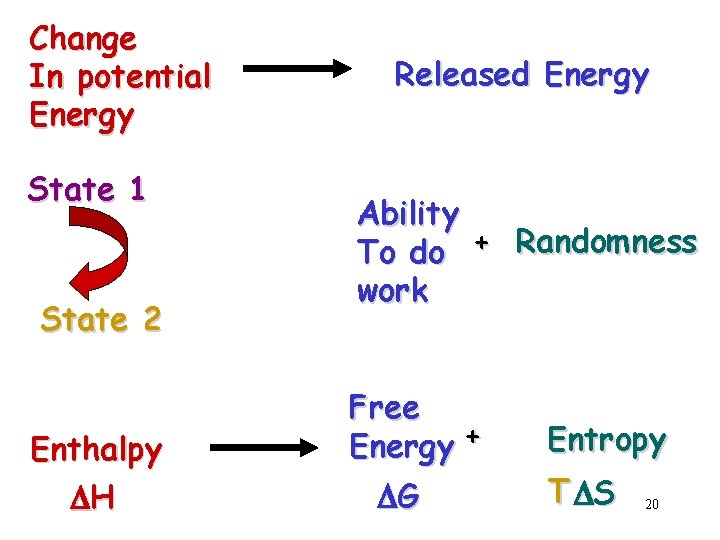
Change In potential Energy State 1 State 2 Enthalpy DH Released Energy Ability To do + Randomness work Free Energy + DG Entropy T DS 20

ENTHALPY DH Change in Chemical Bond Energy ENTROPY DS (disorder) Freedom of Movement or Position Randomness 21

Change in Chemical Bond Energy ENTHALPY DH Time High Potential Low Potential Glucose + 6 O 2 6 CO 2 + 6 H 2 O -D H Low Potential 6 Glucose 6 + 6 O 2 6 CO 2 + H 2 O 22
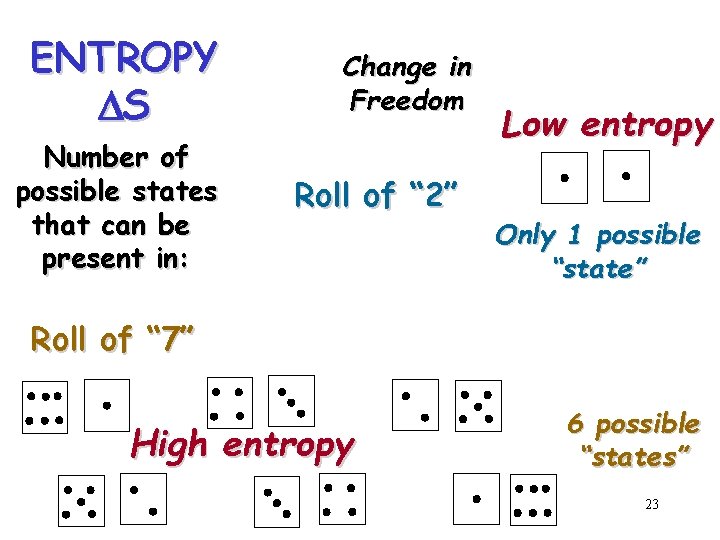
ENTROPY DS Number of possible states that can be present in: Change in Freedom Roll of “ 2” Low entropy Only 1 possible “state” Roll of “ 7” High entropy 6 possible “states” 23
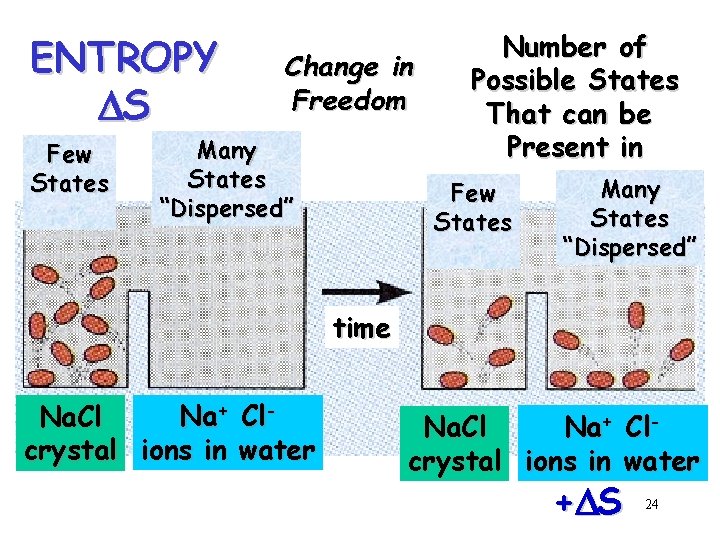
ENTROPY DS Few States Change in Freedom Many States “Dispersed” Number of Possible States That can be Present in Few States Many States “Dispersed” time Na+ Cl. Na. Cl crystal ions in water +D S 24

The Free Energy Change DG Dictates whether a reaction will Proceed spontaneously or not Whether a Reaction is Favorable or Unfavorable 25

Change in Chemical Bond Energy that Goes to Do Useful Work Energy that Goes to Randomness Dependent On Entropy Temperature Enthalpy “free energy” (Gibb’s Free Energy) Kinetic Movement DH = TDS DG = + DH - If DG = negative # reaction is energetically favorable DG TDS “spontaneous” 26
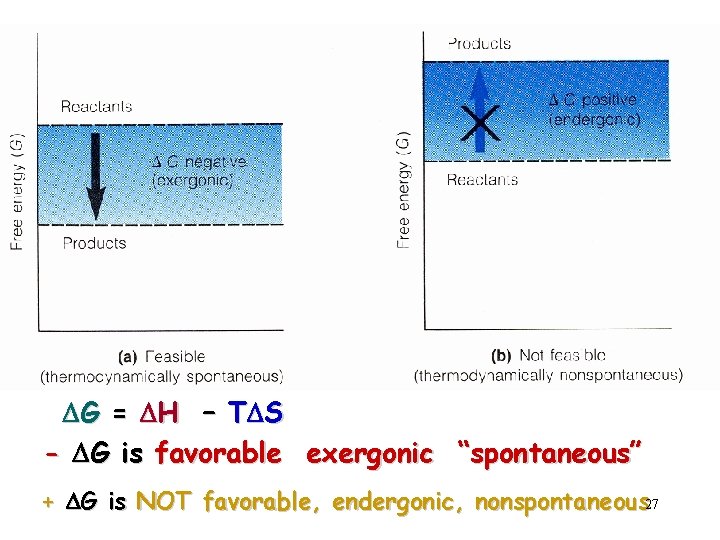
DG = DH – TDS - DG is favorable exergonic “spontaneous” + DG is NOT favorable, endergonic, nonspontaneous 27
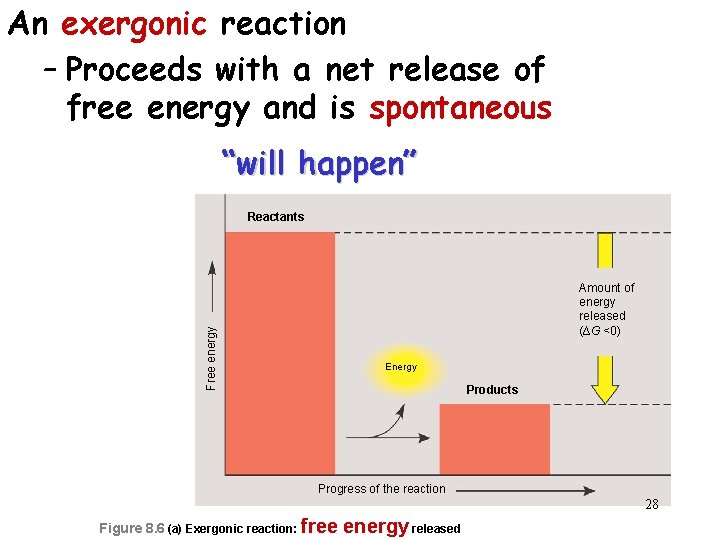
An exergonic reaction – Proceeds with a net release of free energy and is spontaneous “will happen” Reactants Free energy Amount of energy released (∆G <0) Energy Products Progress of the reaction 28 Figure 8. 6 (a) Exergonic reaction: free energy released
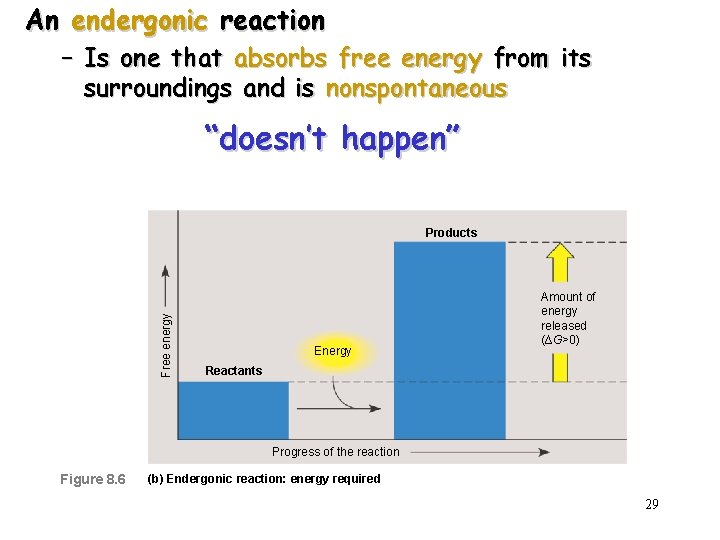
An endergonic reaction – Is one that absorbs free energy from its surroundings and is nonspontaneous “doesn’t happen” Free energy Products Energy Amount of energy released (∆G>0) Reactants Progress of the reaction Figure 8. 6 (b) Endergonic reaction: energy required 29
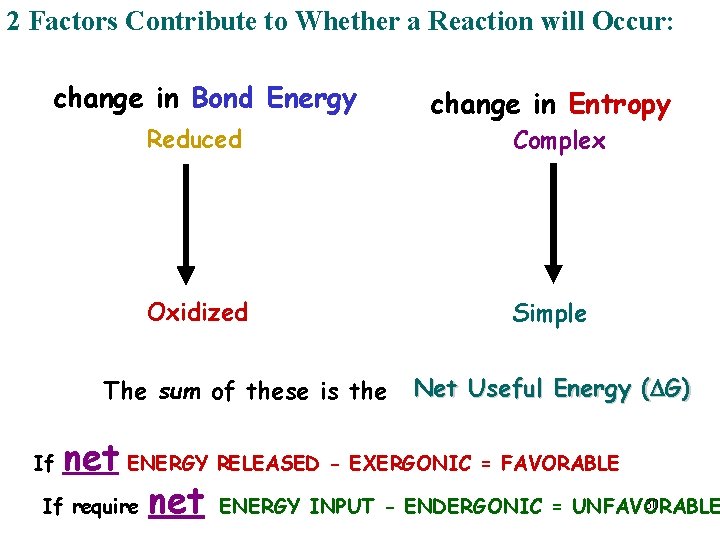
2 Factors Contribute to Whether a Reaction will Occur: change in Bond Energy Reduced Oxidized The sum of these is the change in Entropy Complex Simple Net Useful Energy (DG) net ENERGY RELEASED - EXERGONIC = FAVORABLE If require net ENERGY INPUT - ENDERGONIC = UNFAVORABLE If 30
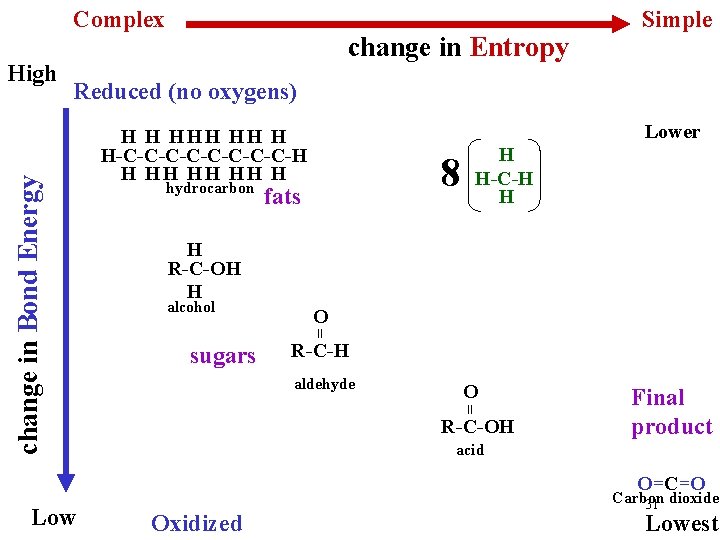
Complex Reduced (no oxygens) Lower H H HH H H-C-C-C-C-H H HH H hydrocarbon 8 fats H H-C-H H H R-C-OH H alcohol O = sugars R-C-H aldehyde O = change in Bond Energy High change in Entropy Simple R-C-OH Final product acid O=C=O Low Oxidized Carbon 31 dioxide Lowest

EXERGONIC REACTIONS gasoline burns iron rusts hydrogen and oxygen form water (explosive!) Either: go to bonding arrangement with lower potential energy Or: go from a more complex state to a simpler state 1 molecule of 8 carbons vs 8 molecules of 1 carbon 32
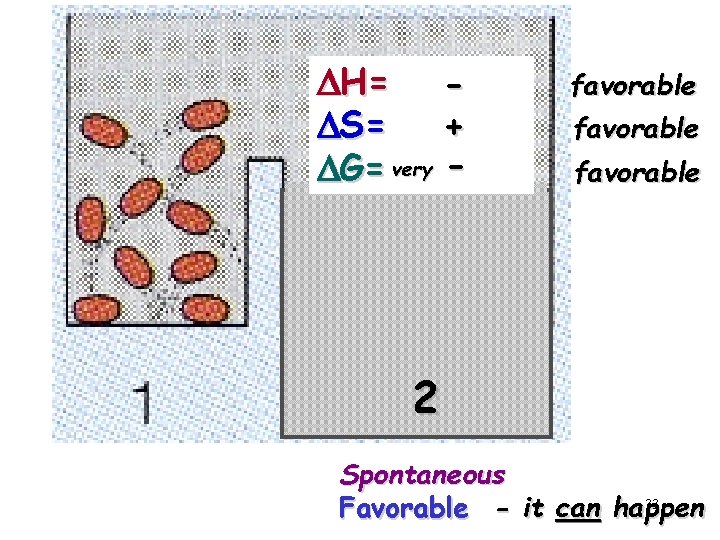
DH= DS= + DG= very - favorable 2 Spontaneous 33 Favorable - it can happen
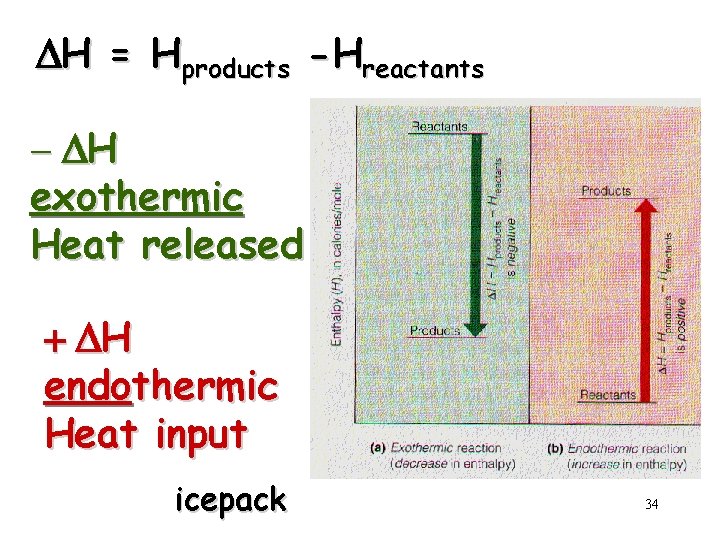
DH = Hproducts -Hreactants - DH exothermic Heat released + DH endothermic Heat input icepack 34
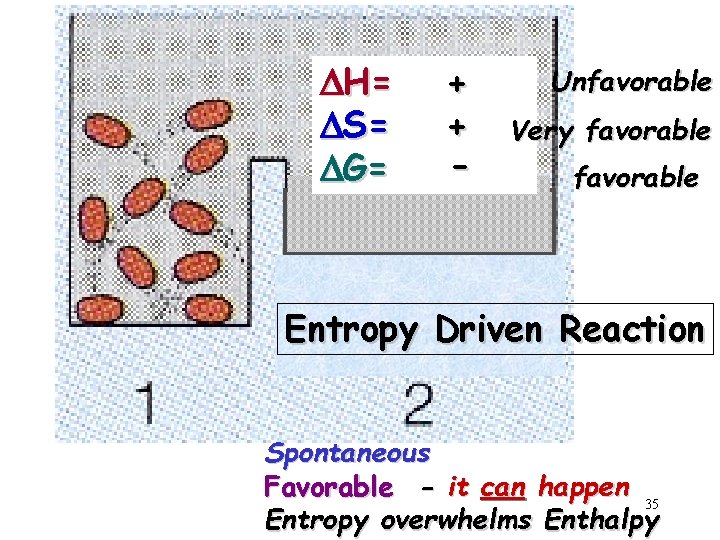
DH= DS= DG= + + - Unfavorable Very favorable Entropy Driven Reaction Spontaneous Favorable - it can happen 35 Entropy overwhelms Enthalpy
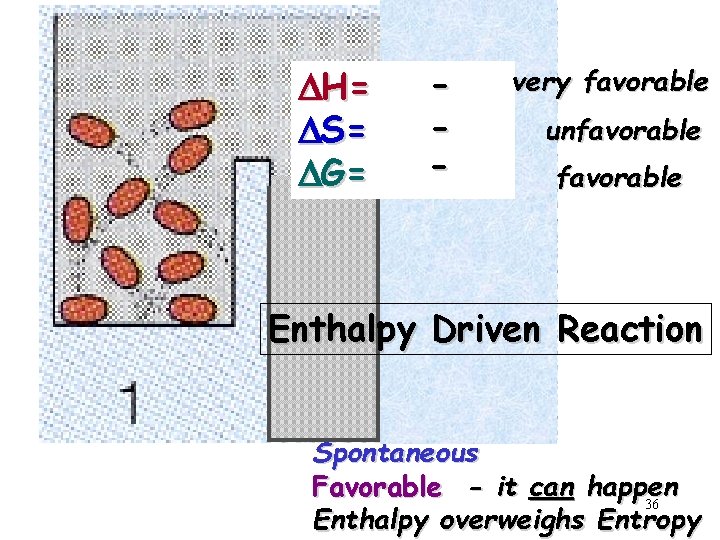
DH= DS= DG= - very favorable unfavorable Enthalpy Driven Reaction Spontaneous Favorable - it can happen 36 Enthalpy overweighs Entropy
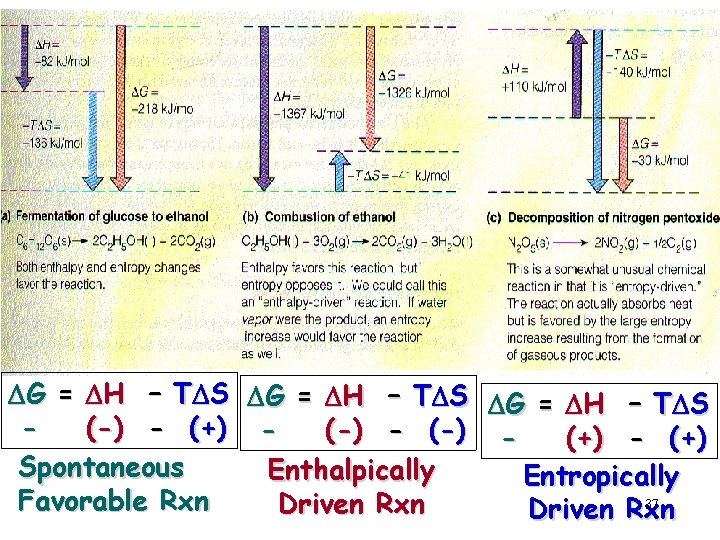
DG = DH – TDS (-) - (+) (-) - (-) (+) - (+) Spontaneous Enthalpically Entropically 37 Favorable Rxn Driven Rxn
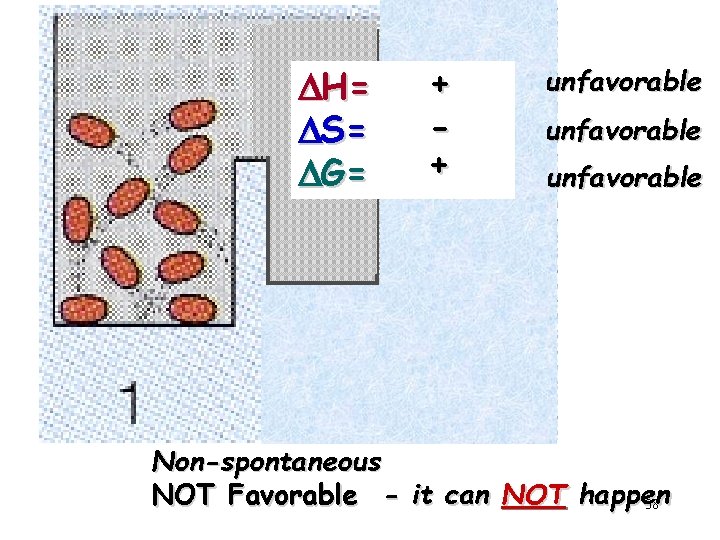
DH= DS= DG= + + unfavorable Non-spontaneous NOT Favorable - it can NOT happen 38

A typical ENDERGONIC/Unfavorable/Non. Spontaneous REACTION - building a polymer Monomer + Monomer Polymer + Water Requires 5. 5 energy units WILL NOT OCCUR How could we make it occur? If have a captured packet of energy of 7. 3 energy units Integrate an exergonic reaction with an endergonic reaction 39
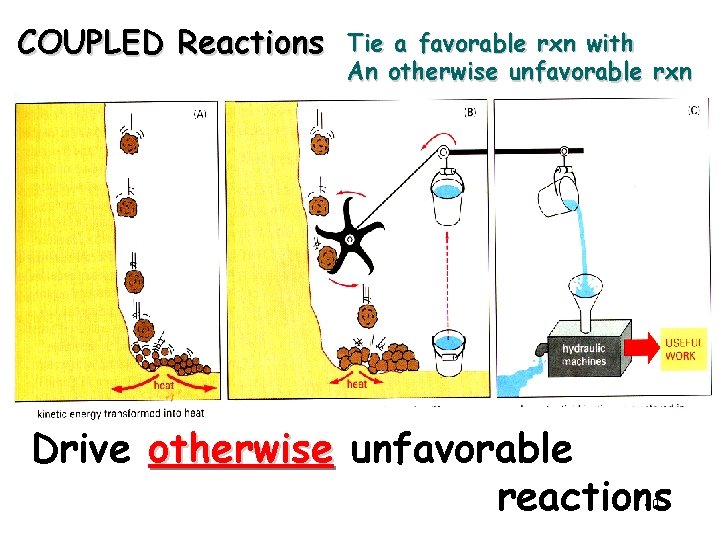
COUPLED Reactions Tie a favorable rxn with An otherwise unfavorable rxn Drive otherwise unfavorable reactions 40

DG = +5. 5 kcal/mole 1. 2. ATP+ H 2 O DG = -7. 3 kcal/mole ADP + Pi ATP ADP + Pi Favorable or unfavorable 41 ?
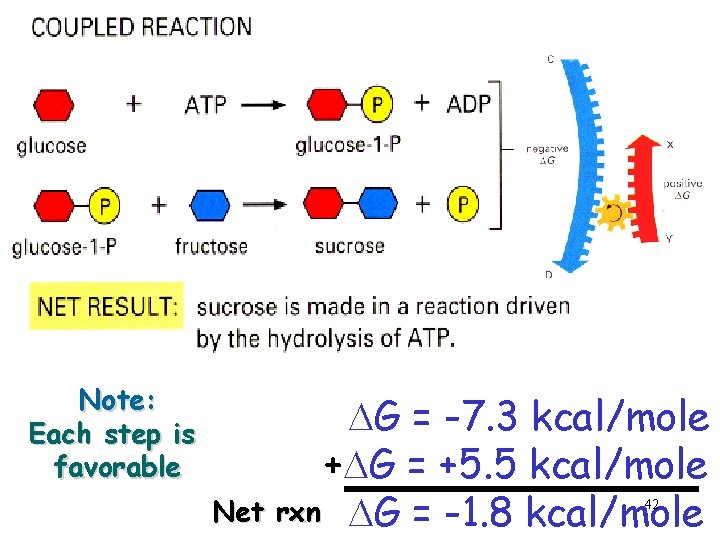
Note: Each step is favorable DG = -7. 3 kcal/mole +DG = +5. 5 kcal/mole Net rxn DG = -1. 8 kcal/mole 42
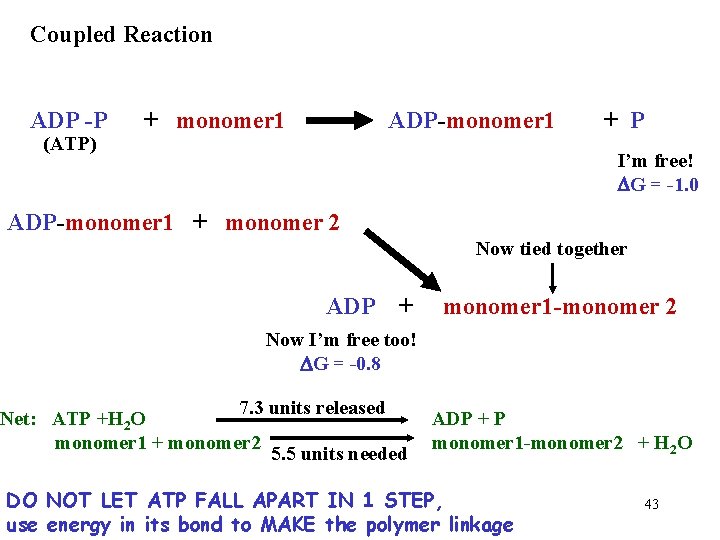
Coupled Reaction ADP -P (ATP) + monomer 1 ADP-monomer 1 + P I’m free! DG = -1. 0 ADP-monomer 1 + monomer 2 Now tied together ADP + monomer 1 -monomer 2 Now I’m free too! DG = -0. 8 7. 3 units released Net: ATP +H 2 O monomer 1 + monomer 2 5. 5 units needed ADP + P monomer 1 -monomer 2 + H 2 O DO NOT LET ATP FALL APART IN 1 STEP, use energy in its bond to MAKE the polymer linkage 43
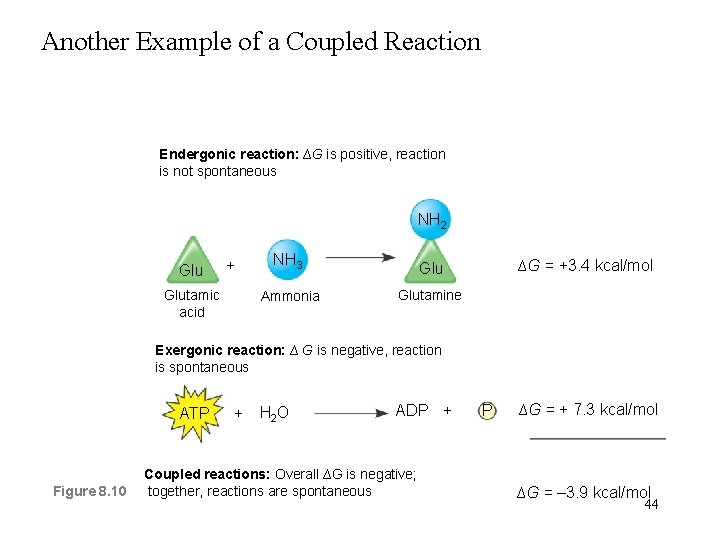
Another Example of a Coupled Reaction Endergonic reaction: ∆G is positive, reaction is not spontaneous NH 2 Glu + Glutamic acid NH 3 Glu Ammonia Glutamine ∆G = +3. 4 kcal/mol Exergonic reaction: ∆ G is negative, reaction is spontaneous ATP Figure 8. 10 + H 2 O ADP + Coupled reactions: Overall ∆G is negative; together, reactions are spontaneous P ∆G = + 7. 3 kcal/mol ∆G = – 3. 9 kcal/mol 44
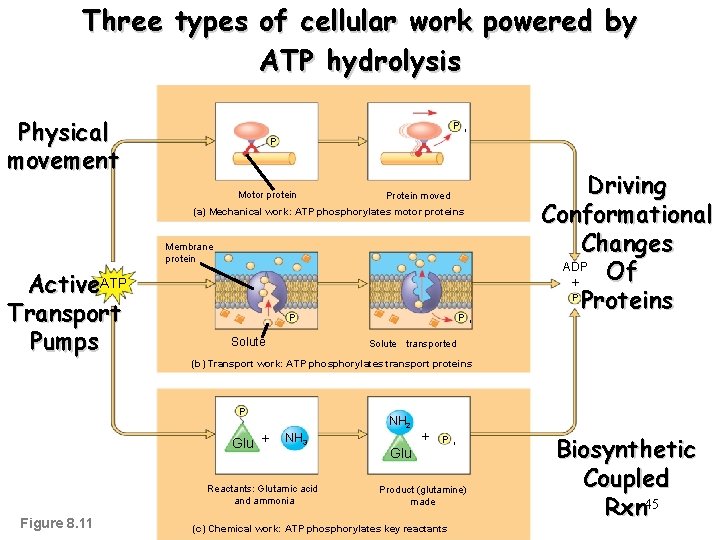
Three types of cellular work powered by ATP hydrolysis Physical movement P i P Motor protein Driving Conformational Changes ADP Of + P Proteins Protein moved (a) Mechanical work: ATP phosphorylates motor proteins Membrane protein Active. ATP Transport Pumps i P Solute P i Solute transported (b) Transport work: ATP phosphorylates transport proteins P Glu + NH 2 NH 3 Reactants: Glutamic acid and ammonia Figure 8. 11 + P Glu i Product (glutamine) made (c) Chemical work: ATP phosphorylates key reactants Biosynthetic Coupled Rxn 45
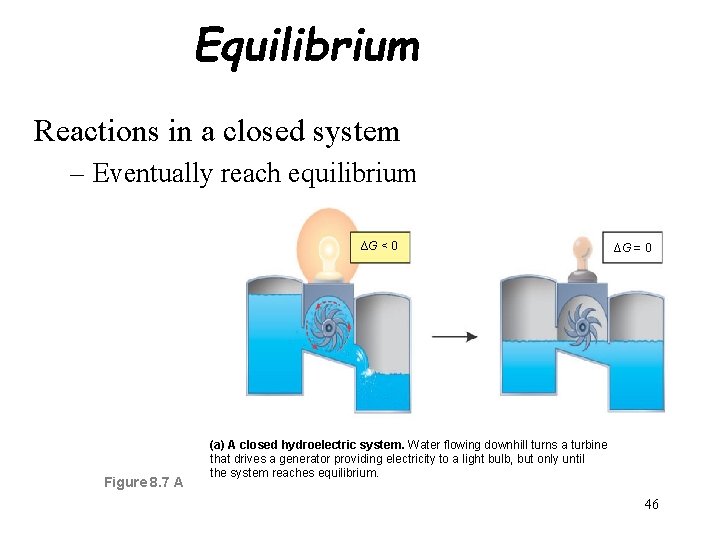
Equilibrium Reactions in a closed system – Eventually reach equilibrium ∆G < 0 Figure 8. 7 A ∆G = 0 (a) A closed hydroelectric system. Water flowing downhill turns a turbine that drives a generator providing electricity to a light bulb, but only until the system reaches equilibrium. 46
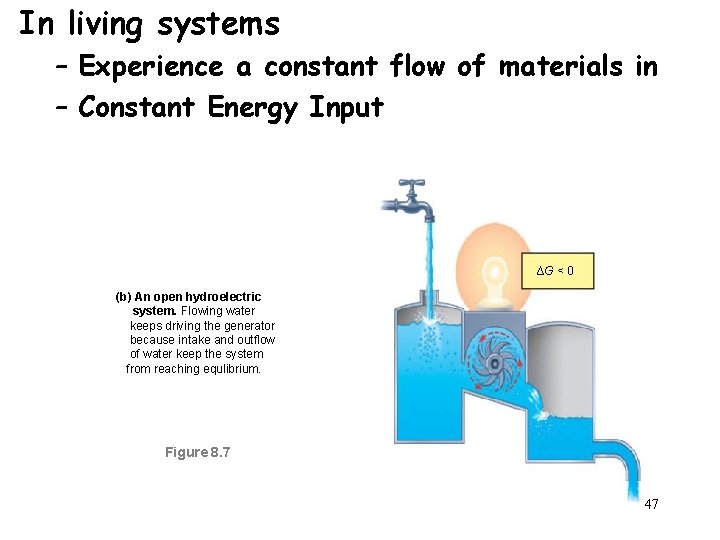
In living systems – Experience a constant flow of materials in – Constant Energy Input ∆G < 0 (b) An open hydroelectric system. Flowing water keeps driving the generator because intake and outflow of water keep the system from reaching equlibrium. Figure 8. 7 47
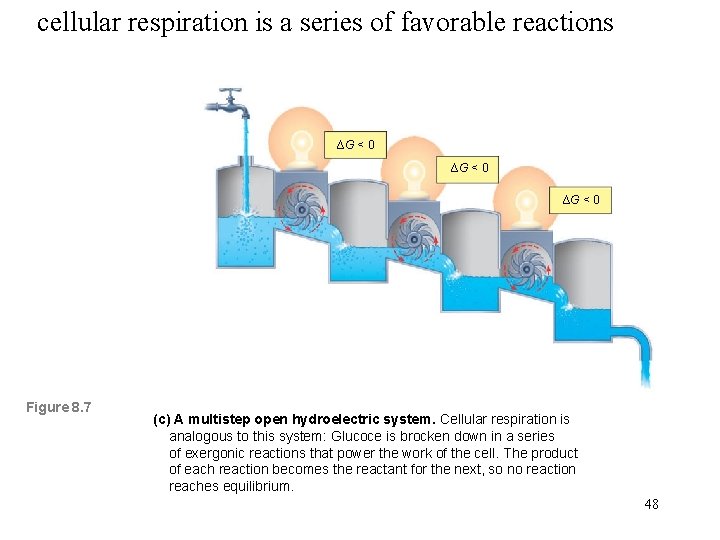
cellular respiration is a series of favorable reactions ∆G < 0 Figure 8. 7 (c) A multistep open hydroelectric system. Cellular respiration is analogous to this system: Glucoce is brocken down in a series of exergonic reactions that power the work of the cell. The product of each reaction becomes the reactant for the next, so no reaction reaches equilibrium. 48

For example, oxidation of glucose: C 6 H 12 O 6 (glucose) + 6 O 2 DG= -686 kcal/mol 6 CO 2 + 6 H 2 O DH = -673 kcal/mol TDS= -13 kcal/mol in the cell, this is done in >21 steps! Capture the energy in small packets ie, 36 ATP units of 7. 3 kcal 49

Summary: -matter is neither created nor destroyed -the universe is proceeding toward disorder DH = enthalpy (heat content, bond energy) DS = entropy (randomness) DG = free energy (available to do work) DG = DH - TDS - coupled reactions -biological systems always need constant energy input 50
- Slides: 50