BCMATargeted ADC MEDI 2228 FirstinHuman Phase I Study

BCMA-Targeted ADC MEDI 2228: First-in-Human Phase I Study in Patients With R/R Multiple Myeloma CCO Independent Conference Highlights of the 2020 Virtual ASH Annual Meeting, December 5 -8, 2020 *CCO is an independent medical education company that provides state-of-the-art medical information to healthcare professionals through conference coverage and other educational programs. Supported by educational grants from Amgen; Astra. Zeneca; Bristol-Myers Squibb; Epizyme, Inc. ; Glaxo. Smith. Kline; Incyte Corporation; Janssen Biotech; Karyopharm Therapeutics Inc. ; Novartis; Pharma. Essentia Corp. ; Seattle Genetics; and Takeda Oncology.
About These Slides § Please feel free to use, update, and share some or all of these slides in your noncommercial presentations to colleagues or patients § When using our slides, please retain the source attribution: Slide credit: clinicaloptions. com § These slides may not be published, posted online, or used in commercial presentations without permission. Please contact permissions@clinicaloptions. com for details

BCMA in Myeloma: Background § Patients with heavily pretreated R/R MM have median OS of < 9 mos, and novel treatment options are needed for this patient population[1] § Anti-BCMA therapies represent a novel class of therapy for MM ‒ In MM cells, BCMA expressed in all disease stages[2, 3] ‒ BCMA expression is restricted in normal cells to plasma cells, terminally differentiated B-cells, and plasmacytoid dendritic cells[4, 5] § MEDI 2228: ADC with fully humanized anti-BCMA antibody conjugated to pyrrolobenzodiazepine (DNA cross-linking agent) dimer tesirine ‒ BCMA-targeted antibody construct that preferentially binds membrane-bound BCMA § Current first-in-human phase I study sought to determine safety and tolerability of novel BCMA-targeted ADC MEDI 2228 in patients with R/R MM[6] 1. Gandhi. Leukemia. 2019; 33: 2266. 2. Lee. Br J Haematol. 2016; 911. 3. Cho. Front Immunol. 2018; 9: 1821. 4. Carpenter. Clin Cancer Res. 2013; 19: 2048. 5. Tai. Blood. 2016; 127: 3225. 6. Kumar. ASH 2020. Abstr 179. Slide credit: clinicaloptions. com
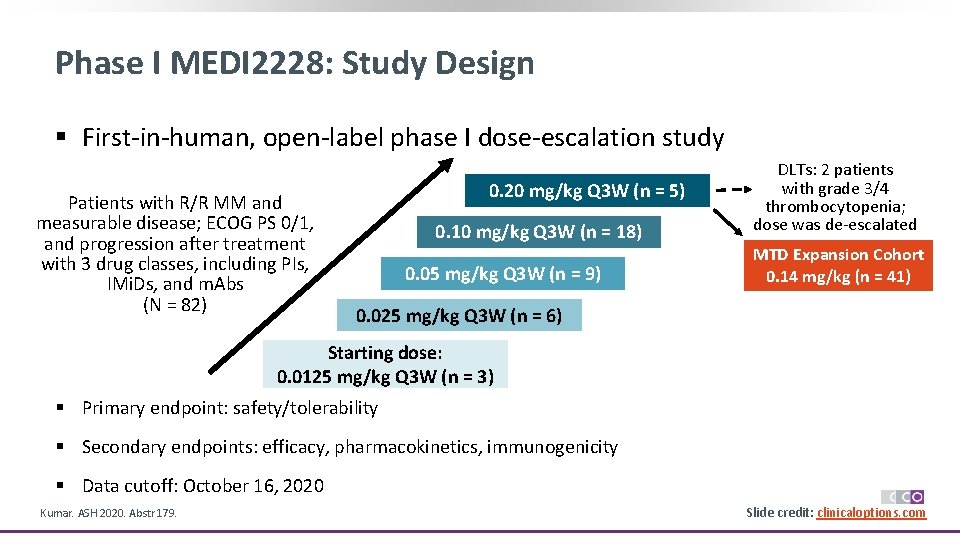
Phase I MEDI 2228: Study Design § First-in-human, open-label phase I dose-escalation study Patients with R/R MM and measurable disease; ECOG PS 0/1, and progression after treatment with 3 drug classes, including PIs, IMi. Ds, and m. Abs (N = 82) 0. 20 mg/kg Q 3 W (n = 5) 0. 10 mg/kg Q 3 W (n = 18) 0. 05 mg/kg Q 3 W (n = 9) DLTs: 2 patients with grade 3/4 thrombocytopenia; dose was de-escalated MTD Expansion Cohort 0. 14 mg/kg (n = 41) 0. 025 mg/kg Q 3 W (n = 6) Starting dose: 0. 0125 mg/kg Q 3 W (n = 3) § Primary endpoint: safety/tolerability § Secondary endpoints: efficacy, pharmacokinetics, immunogenicity § Data cutoff: October 16, 2020 Kumar. ASH 2020. Abstr 179. Slide credit: clinicaloptions. com
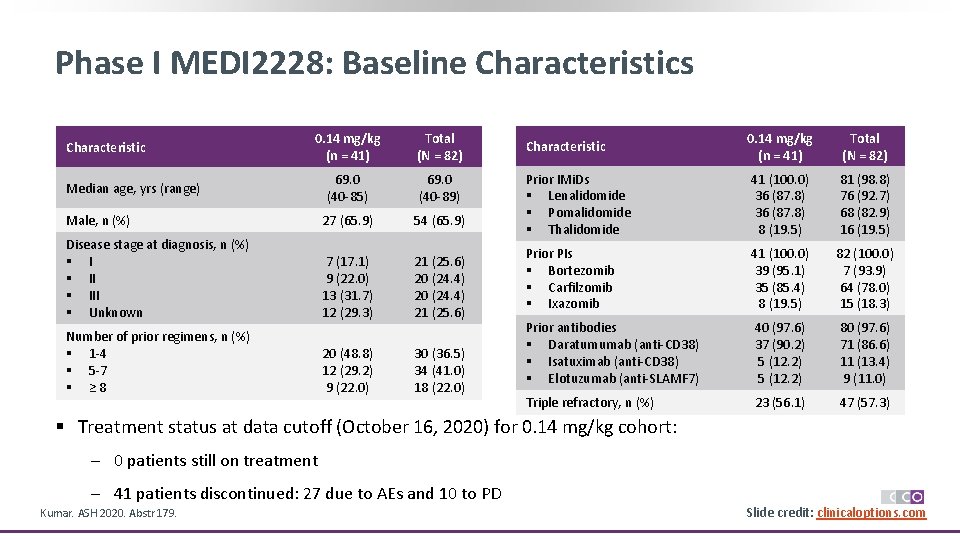
Phase I MEDI 2228: Baseline Characteristics 0. 14 mg/kg (n = 41) Total (N = 82) 69. 0 (40 -85) 69. 0 (40 -89) Male, n (%) 27 (65. 9) 54 (65. 9) Disease stage at diagnosis, n (%) § III § Unknown 7 (17. 1) 9 (22. 0) 13 (31. 7) 12 (29. 3) 21 (25. 6) 20 (24. 4) 21 (25. 6) Number of prior regimens, n (%) § 1 -4 § 5 -7 § ≥ 8 20 (48. 8) 12 (29. 2) 9 (22. 0) 30 (36. 5) 34 (41. 0) 18 (22. 0) Characteristic Median age, yrs (range) Characteristic 0. 14 mg/kg (n = 41) Total (N = 82) Prior IMi. Ds § Lenalidomide § Pomalidomide § Thalidomide 41 (100. 0) 36 (87. 8) 8 (19. 5) 81 (98. 8) 76 (92. 7) 68 (82. 9) 16 (19. 5) Prior PIs § Bortezomib § Carfilzomib § Ixazomib 41 (100. 0) 39 (95. 1) 35 (85. 4) 8 (19. 5) 82 (100. 0) 7 (93. 9) 64 (78. 0) 15 (18. 3) Prior antibodies § Daratumumab (anti-CD 38) § Isatuximab (anti-CD 38) § Elotuzumab (anti-SLAMF 7) 40 (97. 6) 37 (90. 2) 5 (12. 2) 80 (97. 6) 71 (86. 6) 11 (13. 4) 9 (11. 0) Triple refractory, n (%) 23 (56. 1) 47 (57. 3) § Treatment status at data cutoff (October 16, 2020) for 0. 14 mg/kg cohort: ─ 0 patients still on treatment ─ 41 patients discontinued: 27 due to AEs and 10 to PD Kumar. ASH 2020. Abstr 179. Slide credit: clinicaloptions. com
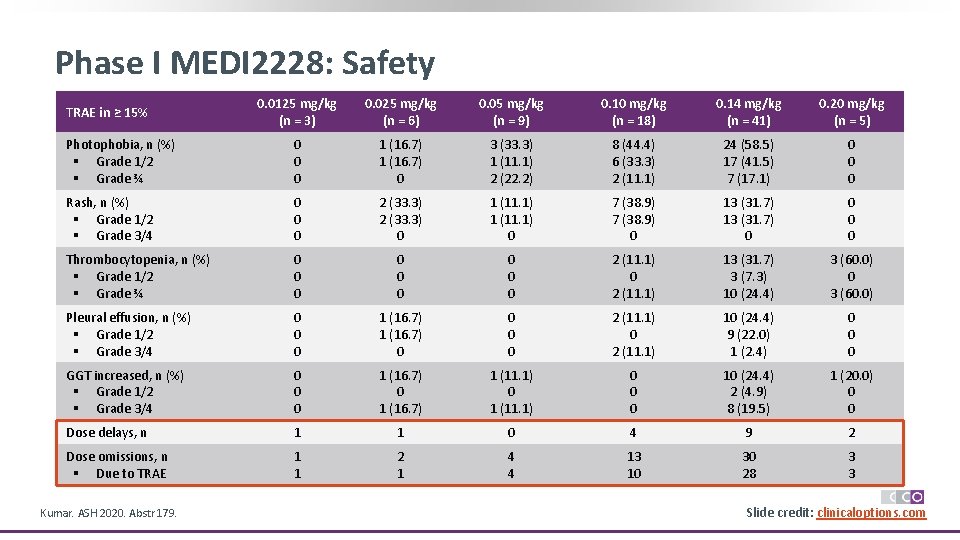
Phase I MEDI 2228: Safety 0. 0125 mg/kg (n = 3) 0. 025 mg/kg (n = 6) 0. 05 mg/kg (n = 9) 0. 10 mg/kg (n = 18) 0. 14 mg/kg (n = 41) 0. 20 mg/kg (n = 5) Photophobia, n (%) § Grade 1/2 § Grade ¾ 0 0 0 1 (16. 7) 0 3 (33. 3) 1 (11. 1) 2 (22. 2) 8 (44. 4) 6 (33. 3) 2 (11. 1) 24 (58. 5) 17 (41. 5) 7 (17. 1) 0 0 0 Rash, n (%) § Grade 1/2 § Grade 3/4 0 0 0 2 (33. 3) 0 1 (11. 1) 0 7 (38. 9) 0 13 (31. 7) 0 0 Thrombocytopenia, n (%) § Grade 1/2 § Grade ¾ 0 0 0 0 0 2 (11. 1) 13 (31. 7) 3 (7. 3) 10 (24. 4) 3 (60. 0) 0 3 (60. 0) Pleural effusion, n (%) § Grade 1/2 § Grade 3/4 0 0 0 1 (16. 7) 0 0 2 (11. 1) 10 (24. 4) 9 (22. 0) 1 (2. 4) 0 0 0 GGT increased, n (%) § Grade 1/2 § Grade 3/4 0 0 0 1 (16. 7) 1 (11. 1) 0 0 0 10 (24. 4) 2 (4. 9) 8 (19. 5) 1 (20. 0) 0 0 Dose delays, n 1 1 0 4 9 2 Dose omissions, n § Due to TRAE 1 1 2 1 4 4 13 10 30 28 3 3 TRAE in ≥ 15% Kumar. ASH 2020. Abstr 179. Slide credit: clinicaloptions. com
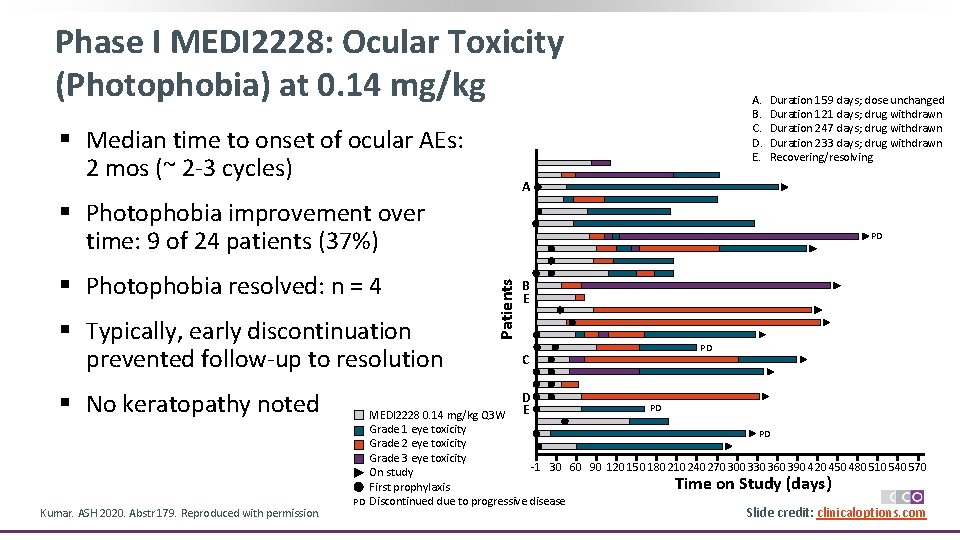
Phase I MEDI 2228: Ocular Toxicity (Photophobia) at 0. 14 mg/kg § Median time to onset of ocular AEs: 2 mos (~ 2 -3 cycles) A. B. C. D. E. A § Photophobia improvement over time: 9 of 24 patients (37%) § Typically, early discontinuation prevented follow-up to resolution § No keratopathy noted Kumar. ASH 2020. Abstr 179. Reproduced with permission. PD Patients § Photophobia resolved: n = 4 Duration 159 days; dose unchanged Duration 121 days; drug withdrawn Duration 247 days; drug withdrawn Duration 233 days; drug withdrawn Recovering/resolving B E PD C D E PD MEDI 2228 0. 14 mg/kg Q 3 W Grade 1 eye toxicity PD Grade 2 eye toxicity Grade 3 eye toxicity -1 30 60 90 120 150 180 210 240 270 300 330 360 390 420 450 480 510 540 570 On study Time on Study (days) First prophylaxis PD Discontinued due to progressive disease Slide credit: clinicaloptions. com
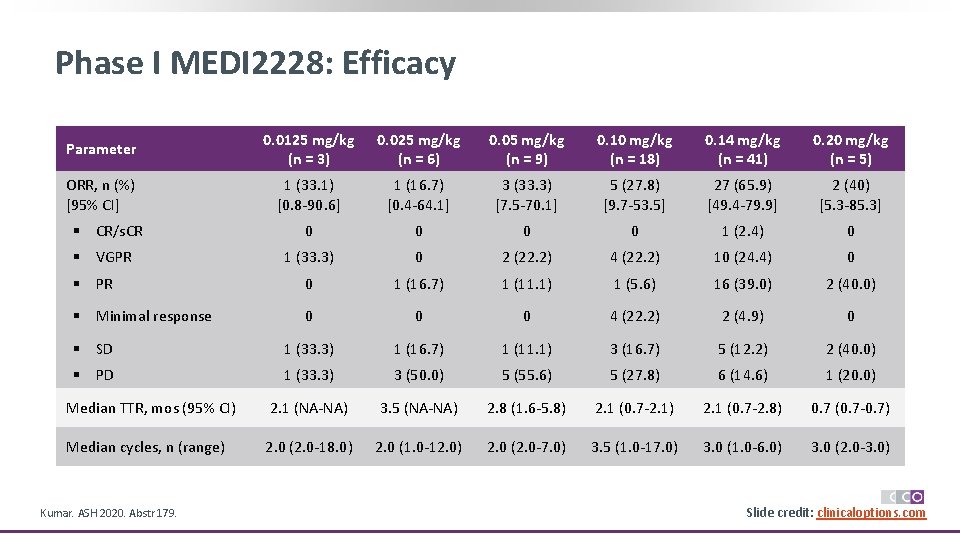
Phase I MEDI 2228: Efficacy Parameter 0. 0125 mg/kg (n = 3) 0. 025 mg/kg (n = 6) 0. 05 mg/kg (n = 9) 0. 10 mg/kg (n = 18) 0. 14 mg/kg (n = 41) 0. 20 mg/kg (n = 5) ORR, n (%) [95% CI] 1 (33. 1) [0. 8 -90. 6] 1 (16. 7) [0. 4 -64. 1] 3 (33. 3) [7. 5 -70. 1] 5 (27. 8) [9. 7 -53. 5] 27 (65. 9) [49. 4 -79. 9] 2 (40) [5. 3 -85. 3] 0 0 1 (2. 4) 0 1 (33. 3) 0 2 (22. 2) 4 (22. 2) 10 (24. 4) 0 § PR 0 1 (16. 7) 1 (11. 1) 1 (5. 6) 16 (39. 0) 2 (40. 0) § Minimal response 0 0 0 4 (22. 2) 2 (4. 9) 0 § SD 1 (33. 3) 1 (16. 7) 1 (11. 1) 3 (16. 7) 5 (12. 2) 2 (40. 0) § PD 1 (33. 3) 3 (50. 0) 5 (55. 6) 5 (27. 8) 6 (14. 6) 1 (20. 0) Median TTR, mos (95% CI) 2. 1 (NA-NA) 3. 5 (NA-NA) 2. 8 (1. 6 -5. 8) 2. 1 (0. 7 -2. 1) 2. 1 (0. 7 -2. 8) 0. 7 (0. 7 -0. 7) Median cycles, n (range) 2. 0 (2. 0 -18. 0) 2. 0 (1. 0 -12. 0) 2. 0 (2. 0 -7. 0) 3. 5 (1. 0 -17. 0) 3. 0 (1. 0 -6. 0) 3. 0 (2. 0 -3. 0) § CR/s. CR § VGPR Kumar. ASH 2020. Abstr 179. Slide credit: clinicaloptions. com

Phase I MEDI 2228: Efficacy MEDI 2228 0. 14 mg/kg Q 3 W ^ IFE negative (serum and urine) ^ IFE positive (serum) PD PD ^ VGPR PD PD PD Patients PR PR PD PR PR PR PD VGPR ^ PR PD PR PR VGPR PD VGPR PR PR VGPR ^ CR PD PD VGPR PR PR ^ VGPR PR PD PR ^†PD PD PR PD PD § Median Do. R: 5. 9 mos § Some patients showed deepening of response after discontinuing treatment § Loss to follow-up after discontinuing treatment due to AEs may have impacted Do. R § 17 of 23 patients who were triple-refractory (PI, IMi. D, and anti -CD 38) achieved response of ≥ PR 0 20 40 60 80 100 120 140 160 180 200 220 240 260 280 300 320 340 360 380 Time on Treatment (days) Kumar. ASH 2020. Abstr 179. Slide credit: clinicaloptions. com

Phase I MEDI 2228: Pharmacokinetics § Q 3 W dosing produced no significant accumulation (mean ratio: 1. 1 -2) § Free concentration of tesirine < 1% of intact MEDI 228 concentrations in majority of patients § Greater binding affinity for membrane-bound vs soluble BCMA Kumar. ASH 2020. Abstr 179. Reproduced with permission. MEDI 2228 Plasma Concentration Time Profile in Dose-Escalation Phase Mean Concentration (ng/m. L) § Linear PK at therapeutic dose not strongly affected by baseline soluble BCMA level Cohort 1: 0. 0125 mg/kg Cohort 2: 0. 025 mg/kg Cohort 3: 0. 05 mg/kg Cohort 4: 0. 1 mg/kg Cohort 4 a: 0. 14 mg/kg Cohort 5: 0. 2 mg/kg 5, 000 100 10 LLOQ 0 7 14 21 Days Slide credit: clinicaloptions. com

Phase I MEDI 2228: Investigator Conclusions § In patients with heavily pretreated R/R MM, treatment with anti-BCMA ADC MEDI 2228 showed manageable toxicity and promising efficacy at MTD of 0. 14 mg/kg Q 3 W ‒ Thrombocytopenia described as DLT: n = 2 at 0. 20 mg/kg ‒ Responses seen across all dose levels; ORR: 66% at 0. 14 mg/kg and median Do. R: 5. 9 mos, likely affected by loss of patients to follow-up § Early onset photophobia cause of discontinuations ‒ 4 cases resolved after drug discontinuation ‒ 37% with documented improvement over time § Expansion cohorts continue to be evaluated at 0. 14 mg/kg, with alternative dosing and scheduling to mitigate eye toxicity Kumar. ASH 2020. Abstr 179. Slide credit: clinicaloptions. com

Go Online for More CCO Coverage of ASH 2020! Short slideset summaries and additional CME-certified analyses with expert commentary on key studies in: § § § Acute and chronic leukemias Lymphomas and chronic lymphocytic leukemia Myelodysplastic syndromes and myeloproliferative neoplasms Myeloma Nonmalignant hematology clinicaloptions. com/oncology
- Slides: 12