BCHS 3304 General Biochemistry I Section 07553 Spring

BCHS 3304: General Biochemistry I, Section 07553 Spring 2003 1: 00 -2: 30 PM Mon. /Wed. AH 101 http: //www. uh. edu/sibs/faculty/glegge Instructor: Glen B. Legge, Ph. D. , Cambridge UK Phone: 713 -743 -8380 Fax: 713 -743 -2636 E-mail: glegge@uh. edu Office hours: Mon. and Wed. (2: 30 -4: 00 PM) or by appointment 353 SR 2 (Science and Research Building 2) 1

SIBS program • Monday Chat room on Webct: 8: 00 -10: 00 PM Tuesday Workshop: 5: 00 -7: 00 PM in 101 AH Wednesday Office Hours: 3: 00 -4: 45 PM in 114 S Wednesday Workshop: 5: 00 -7: 00 PM in 116 SR 1 • Students must activate their webct accounts. • SIBS will not print out exam reviews • Jerry Johnson (BCHS 3304 workshops) contact email: MYSTIK 1775@aol. com

Acids and Bases January 27 2003

Ionization of water • Although neutral water has a tendency to ionize H 2 O <-> H+ + OH • The free proton is associated with a water molecule to form the hydronium ion H 3 O + • High ionic mobility due to proton jumping
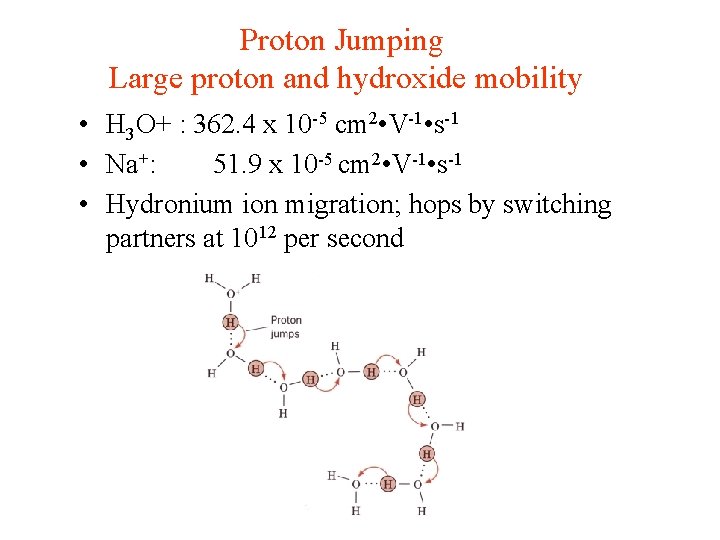
Proton Jumping Large proton and hydroxide mobility • H 3 O+ : 362. 4 x 10 -5 cm 2 • V-1 • s-1 • Na+: 51. 9 x 10 -5 cm 2 • V-1 • s-1 • Hydronium ion migration; hops by switching partners at 1012 per second
![Equilibrium expression • Described by: K = [H+][OH-] [H 2 O] • Where K Equilibrium expression • Described by: K = [H+][OH-] [H 2 O] • Where K](http://slidetodoc.com/presentation_image/3c11af433901be506b7c3ebc769c9cbc/image-6.jpg)
Equilibrium expression • Described by: K = [H+][OH-] [H 2 O] • Where K is the dissociation constant • Considering [H 2 O] constant yields Kw = [H+][OH-]
![Kw Kw = [H+][OH-] • Where Kw is the ionization constant of water • Kw Kw = [H+][OH-] • Where Kw is the ionization constant of water •](http://slidetodoc.com/presentation_image/3c11af433901be506b7c3ebc769c9cbc/image-7.jpg)
Kw Kw = [H+][OH-] • Where Kw is the ionization constant of water • For pure water ionization constant is 10 -14 M 2 at 25º • For pure water [H+] = [OH-] = (Kw)1/2 = 10 -7 M
![Acids and bases • For pure water (neutral) [H+] = [OH-] = (Kw)1/2 = Acids and bases • For pure water (neutral) [H+] = [OH-] = (Kw)1/2 =](http://slidetodoc.com/presentation_image/3c11af433901be506b7c3ebc769c9cbc/image-8.jpg)
Acids and bases • For pure water (neutral) [H+] = [OH-] = (Kw)1/2 = 10 -7 M • Acidic if [H+] > 10 -7 M • Basic if [H+] < 10 -7 M
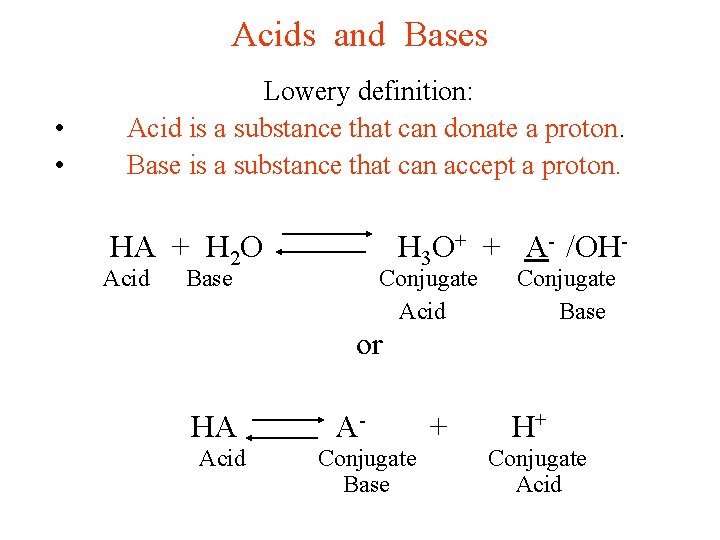
Acids and Bases • • Lowery definition: Acid is a substance that can donate a proton. Base is a substance that can accept a proton. HA + H 2 O Acid Base H 3 O+ + A- /OH- Conjugate Acid Base or HA A- + H+ Acid Conjugate Base Acid
![If you establish equilibrium, changes in [H+] will shift the ratio of HA and If you establish equilibrium, changes in [H+] will shift the ratio of HA and](http://slidetodoc.com/presentation_image/3c11af433901be506b7c3ebc769c9cbc/image-10.jpg)
If you establish equilibrium, changes in [H+] will shift the ratio of HA and A-. By adding more H+ , A- will be consumed forming HA. If there is sufficient [A-], the extra H+ will also be consumed and the [H+] will not change.
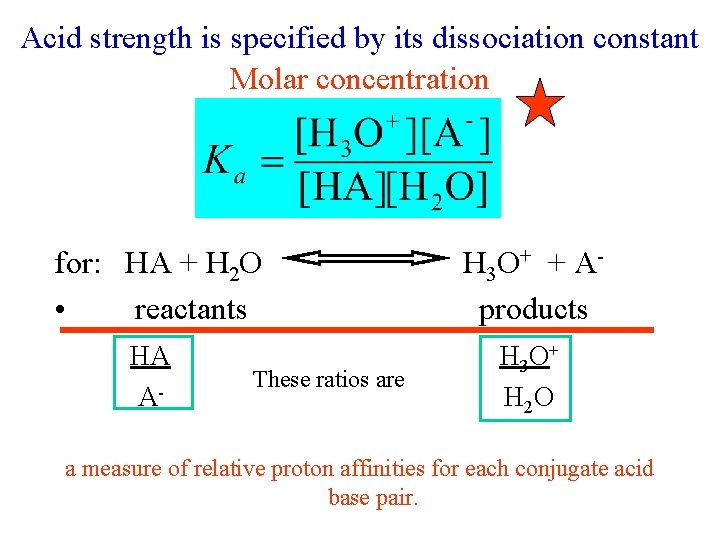
Acid strength is specified by its dissociation constant Molar concentration for: HA + H 2 O H 3 O+ + A • reactants products HA H 3 O+ A- These ratios are H 2 O a measure of relative proton affinities for each conjugate acid base pair.

What to do about the water! The concentration of H 2 O remains almost unchanged especially in dilute acid solutions. What is the concentration of H 2 O? Remember the definition: Moles per liter 1 mole of H 2 O = 18 g = 18 ml 1000 g/liter
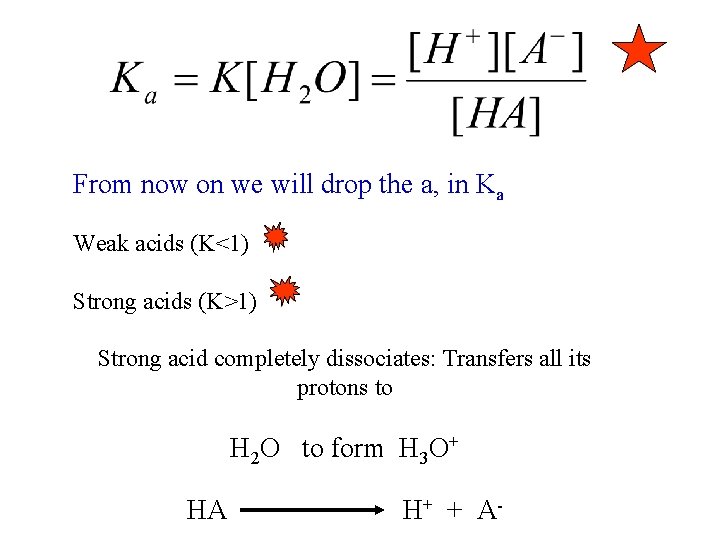
From now on we will drop the a, in Ka Weak acids (K<1) Strong acids (K>1) Strong acid completely dissociates: Transfers all its protons to H 2 O to form H 3 O+ HA H+ + A-

Weak Acids Weak acids do not completely dissociate: They form an equilibrium: If we ADD more H+, the equilibrium shifts to form more HA using up A- that is present.
![Dissociation of H 2 O Water also dissociates [H 2 O] = 55. 5 Dissociation of H 2 O Water also dissociates [H 2 O] = 55. 5](http://slidetodoc.com/presentation_image/3c11af433901be506b7c3ebc769c9cbc/image-15.jpg)
Dissociation of H 2 O Water also dissociates [H 2 O] = 55. 5 Ionization constant for water
![Since there is equal amounts of [H+] and [OH-] This is neutral At [H+] Since there is equal amounts of [H+] and [OH-] This is neutral At [H+]](http://slidetodoc.com/presentation_image/3c11af433901be506b7c3ebc769c9cbc/image-16.jpg)
Since there is equal amounts of [H+] and [OH-] This is neutral At [H+] above this concentration the solution is ACIDIC At [H+] below this concentration the solution is BASIC
![[H+] p. H 10 -7 = 7 10 -3 = 3 10 -2 = [H+] p. H 10 -7 = 7 10 -3 = 3 10 -2 =](http://slidetodoc.com/presentation_image/3c11af433901be506b7c3ebc769c9cbc/image-17.jpg)
[H+] p. H 10 -7 = 7 10 -3 = 3 10 -2 = 2 10 -10 = 10 5 x 10 -4 = 3. 3 7 x 10 -6 = 5. 15 3. 3 x 10 -8 = 7. 48 p. H = -Log[H+] It is easier to think in log of concentrations but it takes practice!!
![Relationship between p. H and [H+] / [OH-] concentration Relationship between p. H and [H+] / [OH-] concentration](http://slidetodoc.com/presentation_image/3c11af433901be506b7c3ebc769c9cbc/image-18.jpg)
Relationship between p. H and [H+] / [OH-] concentration

Observation If you add. 01 ml i. e 1/100 ml of 1 M HCl to 1000 ml of water, the p. H of the water drops from 7 to 5!! i. e 100 fold increase in H+ concentration: Log = 2 change. Problem: Biological properties change with small changes in p. H, usually less than 1 p. H unit. How does a system prevent fluctuations in p. H?

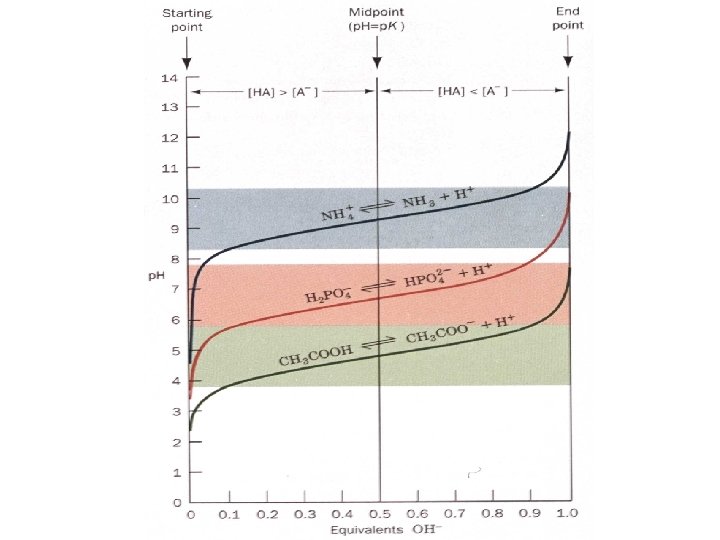
Buffers A buffer can resist p. H changes if the p. H is at or near a weak acid p. K value. Buffer range: the p. H range where maximum resistance to p. H change occurs when adding acid or base. It is = +1 p. H from the weak acid p. K If p. K is 4. 8 the buffering range is 3. 8 Why? 5. 8

Henderson - Hasselbalch equation From Rearrange Take (-)Log of each

Above and below this range there is insufficient amount of conjugate acid or base to combine with the base or acid to prevent the change in p. H.

For weak acids HA A- + H+ This equilibrium depends on concentrations of each component. If [HA] = [A-] or 1/2 dissociated Then : p. H = p. K By definition the p. K is the p. H where [HA] = [A-] : 50% dissociated

The buffer effect can be seen in a titration curve. To a weak acid salt, CH 3 C 00 -, add HC 1 while monitoring p. H vs. the number of equivalents of acid added. or do the opposite with base. Buffer capacity: the molar amount of acid which the buffer can handle without significant changes in p. H. i. e 1 liter of a. 01 M buffer can not buffer 1 liter of a 1 M solution of HCl but 1 liter of a 1 M buffer can buffer 1 liter of a. 01 M solution of HCl
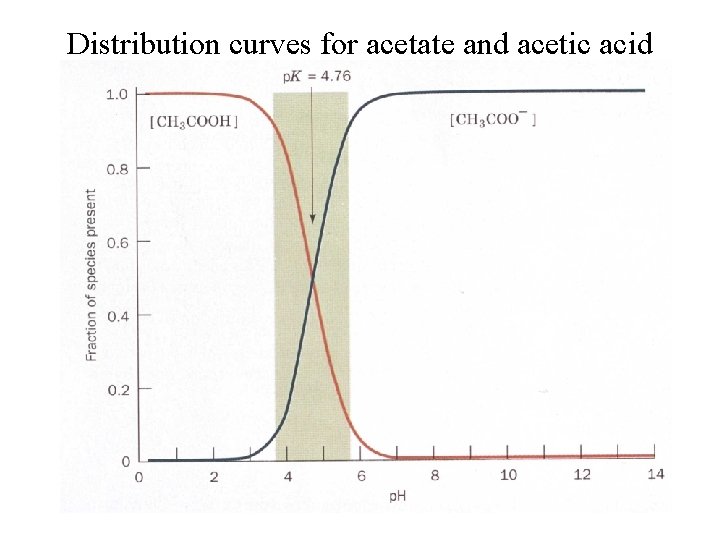
Distribution curves for acetate and acetic acid
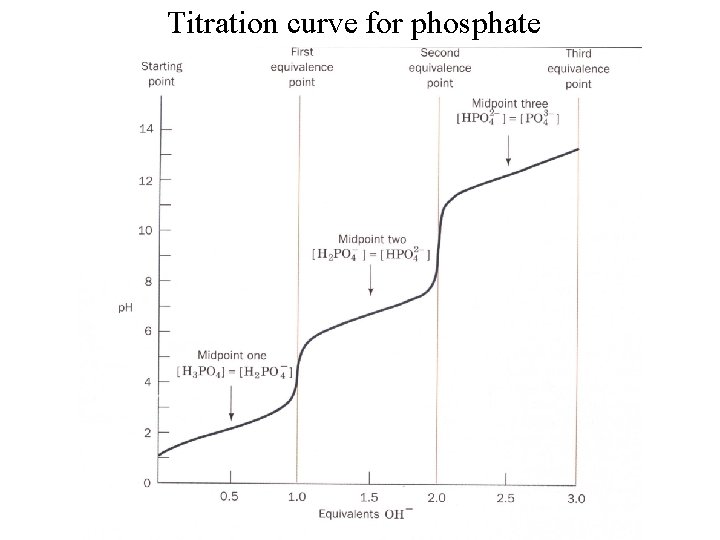
Titration curve for phosphate
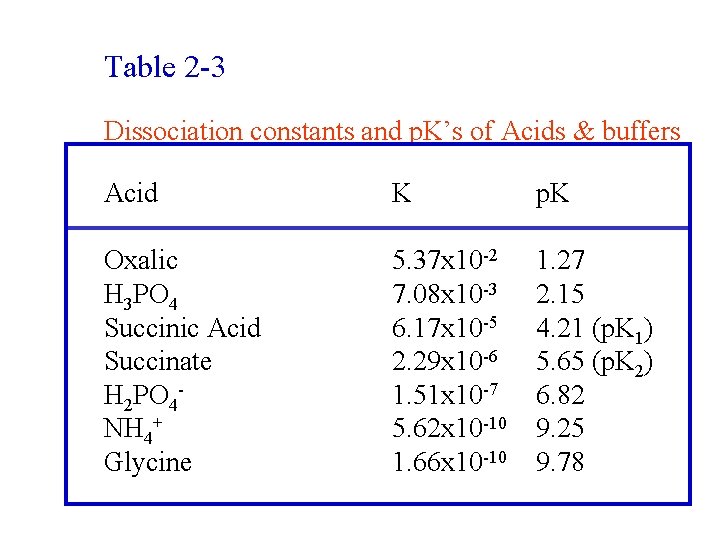
Table 2 -3 Dissociation constants and p. K’s of Acids & buffers Acid K p. K Oxalic H 3 PO 4 Succinic Acid Succinate H 2 PO 4 NH 4+ Glycine 5. 37 x 10 -2 7. 08 x 10 -3 6. 17 x 10 -5 2. 29 x 10 -6 1. 51 x 10 -7 5. 62 x 10 -10 1. 66 x 10 -10 1. 27 2. 15 4. 21 (p. K 1) 5. 65 (p. K 2) 6. 82 9. 25 9. 78
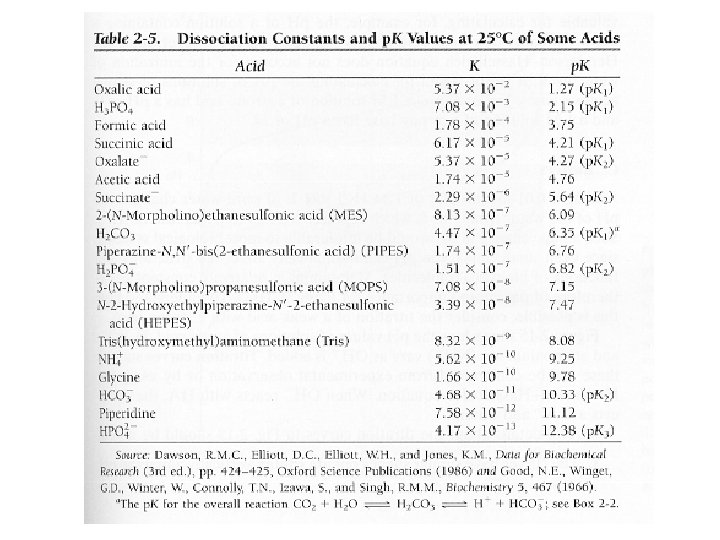

Exams Problems You will be asked to solve Henderson - Hasselbalch type problems: You may be asked the p. H, p. K, the ratio of acid or base or solve for the final concentrations of each.

The 6 step approach 1. Write the Henderson + Hasselbalch equation. 2. Write the acid base equation 3. Make sure either an H+ or OH- is in the equation. 3. Find out what you are solving for 4. Write down all given values. 5. Set up equilibrium conditions. 6. Plug in H + H equation and solve.

What is the p. H of a solution of that contains 0. 1 M CH 3 C 00 - and 0. 9 M CH 3 C 00 H? 1) p. H = p. K + Log [A-] [HA] 2) CH 3 C 00 H CH 3 C 00 - + H+ 3) Find p. H 4) p. K = 4. 76 A- = 0. 1 M HA = 0. 9 M 5) Already at equilibrium 6) X = 4. 76 + Log 0. 1 0. 9 Log 0. 111 = -. 95 X = 4. 76 + (-. 95) X = 3. 81

What would the concentration of CH 3 C 00 - be at p. H 5. 3 if 0. 1 M CH 3 C 00 H was adjusted to that p. H. 1) 2) 3) 4) 5) p. H = p. K + Log [A-] [HA] CH 3 C 00 H CH 3 C 00 - + H+ Find equilibrium value of [A-] i. e [CH 3 C 00 -] p. H = 5. 3; p. K = 4. 76 Let X = amount of CH 3 C 00 H dissociated at equilibrium [A-] = [X] [HA] = [0. 1 - X] 6) 5. 3 = 4. 76 + Log [X] [0. 1 - X] Now solve.

Blood Buffering System • Bicarbonate most significant buffer • Formed from gaseous CO 2 + H 2 O <-> H 2 CO 3 <-> H+ + HCO 3 • Normal value blood p. H 7. 4 • Deviations from normal p. H value lead to acidosis
- Slides: 34