BCH 342 Transamination of Amino acid Transamination is

BCH 342

Transamination of Amino acid • Transamination is a chemical reaction that transfers an amino group to a ketoacid to form new amino acids. • This pathway is responsible for the deamination of most amino acids. • This is one of the major degradation pathways which convert essential amino acids to nonessential amino acids (amino acids that can be synthesized de novo by the organism).

• Transamination is accomplished by enzymes called transaminases or aminotransferases. α -ketoglutarate acts as the predominant aminogroup acceptor and produces glutamate as the new amino acid. Aminoacid + α-ketoglutarate ↔ α-keto acid + Glutamate • Glutamate's amino group, in turn, is transferred to oxaloacetate in a second transamination reaction yielding aspartate. Glutamate + oxaloacetate ↔ α-ketoglutarate + aspartate
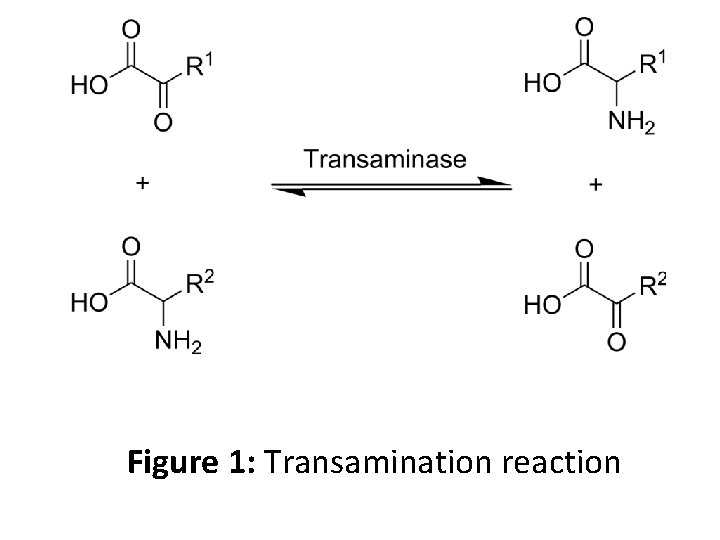
Figure 1: Transamination reaction

Mechanism of Action • Transamination catalyzed by aminotransferase occurs in two stages. • In the first step, the α amino group of an amino acid is transferred to the enzyme, producing the corresponding α-keto acid and the aminated enzyme. • During the second stage, the amino group is transferred to the keto acid acceptor, forming the amino acid product while regenerating the enzyme. • The chirality of an amino acid is determined during transamination.

• For the reaction to complete, aminotransferases require participation of aldehyde containing coenzyme, pyridoxal-5'-phosphate (PLP), a derivative of Pyridoxine (Vitamin B 6). • The amino group is accommodated by conversion of this coenzyme to pyridoxamine-5'-phosphate (PMP). Pyridoxal -5’-phosphate is covalently attached to the enzyme via a Schiff Base linkage formed by the condensation of its aldehyde group with the ε-amino (epsilon (5 th) amino) group of an enzymatic Lys residue. • The Schiff base, which is conjugated to the enzymes pyridinium ring is the focus of the coenzyme activity.
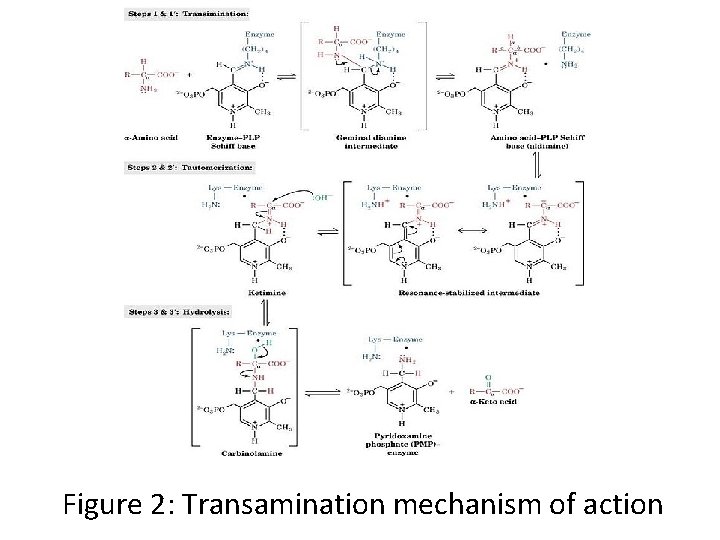
Figure 2: Transamination mechanism of action

• The product of transamination reactions depend on the availability of α-keto acids. • The products usually are either alanine, aspartate or glutamate, since their corresponding alpha-keto acids are produced through metabolism of fuels. • Being a major degradative aminoacid pathway, lysine, proline and threonine are the only three amino acids that do not always undergo transamination and rather use respective dehydrogenase.

Types of Transferases • Transamination is mediated by several different aminotransferase enzymes. • These may be specific for individual amino acids, or they may be able to process a group of chemically similar ones. • The latter applies to the group of the branched-chain amino acids, which comprises leucine, isoleucine, and valine. • The two common types of aminotransferases are Alanine aminotransferase (ALT) and Aspartate aminotransferase (AST).

Oxidative Deamination • Oxidative deamination is a form of deamination that generates α-keto acids and other oxidized products from aminecontaining compounds, and occurs only in the liver. • Oxidative deamination is an important step in the catabolism of amino acids, generating a more metabolizable form of the amino acid, and also generating ammonia as a toxic byproduct. The ammonia generated in this process can then be neutralized into urea via the urea cycle.

• Much of the oxidative deamination occurring in cells involves the amino acid glutamate, which can be oxidatively deaminated by the enzyme glutamate dehydrogenase (GDH), using NAD or NADP as a coenzyme. • This reaction generates α-ketoglutarate (α-KG) and ammonia. Glutamate can then be regenerated from α-ketoglutarate via the action of transaminases or aminotransferase, which catalyze the transfer of an amino group from an amino acid to an α-keto acid. • In this manner, an amino acid can transfer its amine group to glutamate, after which glutamate dehydrogenase can then liberate ammonia via oxidative deamination. This is a common pathway during amino acid catabolism.

• Another enzyme responsible for oxidative deamination is monoamine oxidase, which catalyzes the deamination of monoamines via addition of oxygen. This generates the corresponding ketone- or aldehyde-containing form of the molecule, and generates ammonia.
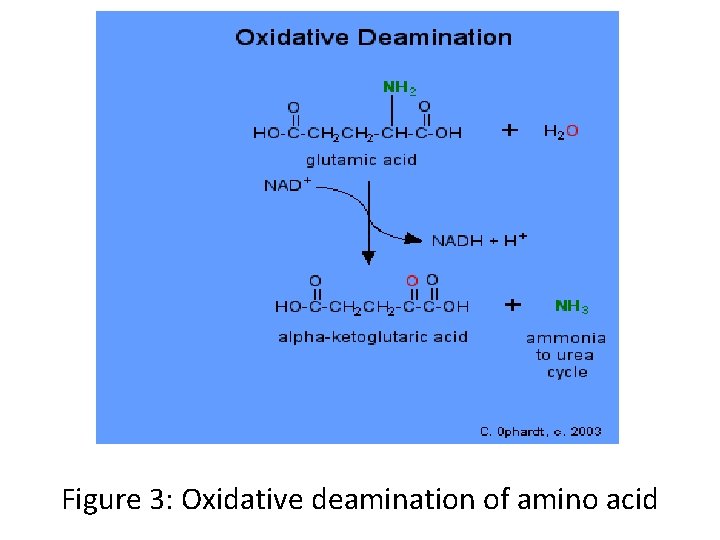
Figure 3: Oxidative deamination of amino acid
- Slides: 13