BCH 342 Metabolism of Carbohydrate glycolysis krebs cycle

BCH 342 Metabolism of Carbohydrate: glycolysis, kreb’s cycle, hexose monophosphate shunt, gluconeogenesis, glycogenolysis. Transamination and oxidative deamination of amino acids: the urea cycle

Glycolysis • Nearly every living cell carries out a catabolic process known as glycolysis, the stepwise degradation of glucose (and other simple sugars). • Glycolysis is a paradigm of metabolic pathways. Carried out in the cytosol of cells, it is basically an anaerobic process; its principal steps occur with no requirement for oxygen.

• Living things first appeared in an environment lacking O 2, and glycolysis was an early and important pathway for extracting energy from nutrient molecules. Modern organisms still employ glycolysis to provide precursor molecules for aerobic catabolic pathways (such as the tricarboxylic acid cycle) and as a short-term energy source when oxygen is limiting.
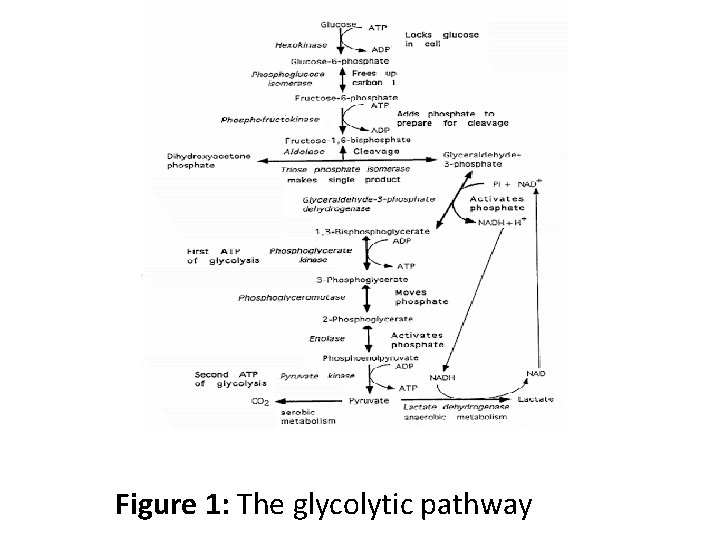
Figure 1: The glycolytic pathway

• The initial reaction of the glycolysis pathway involves phosphorylation of glucose at carbon atom 6 by either hexokinase or glucokinase. • The second step in glycolysis is a common type of metabolic reaction; the isomerization of a sugar. In this particular case, the carbonyl oxygen of glucose-6 -phosphate is shifted from C-1 to C-2. This amounts to isomerization of an aldose (glucose-6 -phosphate) to a ketose—fructose-6 phosphate. The enzyme responsible for this isomerization is phosphoglucoisomerase, also known as glucose phosphate isomerase.

• In the third step of glycolysis, the action of phosphoglucoisomerase, moving the carbonyl group from C-1 to C-2, creates a new primary alcohol function at C-1, which is the conversion of fructose-6 -phosphate to fructose-1, 6 -bisphoshate by the enzyme phosphofructokinase. • In the fourth step of glycolysis, the enzyme Fructose bisphosphate aldolase cleaves fructose-1, 6 bisphosphate between the C-3 and C-4 carbons to yield two triose phosphates. The products are dihydroxyacetone phosphate (DHAP) and glyceraldehyde-3 -phosphate (G-3 -P).

• In the fifth step of glycolysis; of the two products formed in the forth step which are dihydroxyacetone phosphate (DHAP) and glyceraldehydes-3 -phosphate (G-3 -P), only glyceraldehyde-3 -phosphate (G-3 -P) goes directly into the second phase of glycolysis. • The other triose phosphate, dihydroxyacetone phosphate, must be converted to glyceraldehyde-3 phosphate by the enzyme triose phosphate isomerase. • This reaction thus permits both products of the aldolase reaction to continue in the glycolytic pathway, and in essence makes the C-1, C-2, and C-3 carbons of the starting glucose molecule equivalent to the C-6, C-5, and C-4 carbons, respectively.

• In the sixth step of glycolysis, the reaction involves oxidation–reduction, glyceraldehyde 3 -phosphate is oxidized to 1, 3 bisphoglycerate by the enzyme glyceraldehyde-3 -phosphate dehydrogenase (sixth step of glycolysis). • In the seventh step of glycolysis, the enzyme phosphoglycerate kinase transfers a phosphoryl group from 1, 3 bisphoglycerate to ADP to form an ATP and eventually produces 3 -phosphoglycerate.

• In the eight step of glycolysis, the remaining steps in the glycolytic pathway prepare for synthesis of the second ATP equivalent. This begins with the enzyme phosphoglycerate mutase reaction, in which the phosphoryl group of 3 -phosphoglycerate is moved from C 3 to C-2. to form 2 -phosphoglycerate. • In the ninth step of glycolysis, the enzyme enolase catalyzes the formation of phosphoenolpyruvate from 2 phosphoglycerate.

• In the tenth step of glycolysis, the reaction of glycolysis is catalyzed by the enzyme pyruvate kinase, which converts phosphoenol pyruvate to Pyruvate is converted to acetyl co -A by the enzyme pyruvate dehydrogenase which plays a vital role in further production of energies including the Kreb’s cycle

Regulation and Rate limiting step of enzymes in Glycolysis • Hexokinase or Glucokinase • Phosphofructokinase • Pyruvate kinase

Post-glycolytic processes • Anaerobic conditions - Lactic acid fermentation - Ethanolic fermentation • Aerobic condition - acetly co-A production leading to the citric acid cycle

The Citric acid cycle • The citric acid cycle (CAC) – also known as the TCA cycle (tricarboxylic acid cycle) or the Krebs cycle is a series of chemical reactions used by all aerobic organisms to release stored energy through the oxidation of acetyl-Co. A derived from carbohydrates, fats, and proteins, into adenosine triphosphate (ATP) and carbon dioxide. • In addition, the cycle provides precursors of certain amino acids, as well as the reducing agent NADH, that are used in numerous other reactions.

• In eukaryotic cells, the citric acid cycle occurs in the matrix of the mitochondrion. • In prokaryotic cells, such as bacteria, which lack mitochondria, the citric acid cycle reaction sequence is performed in the cytosol with the proton gradient for ATP production being across the cell's surface (plasma membrane) rather than the inner membrane of the mitochondrion. • The overall yield of energy-containing compounds from the TCA cycle is three NADH, one FADH 2, and one GTP.
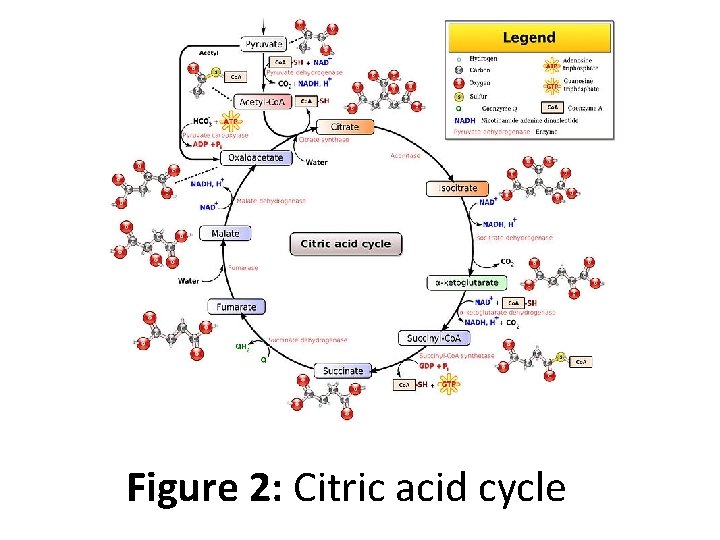
Figure 2: Citric acid cycle

• There are ten basic steps in the citric acid cycle. • Two carbon atoms are oxidized to CO 2, the energy from these reactions is transferred to other metabolic processes through GTP (or ATP), and as electrons in NADH and QH 2 (ubiquitol). • The NADH generated in the citric acid cycle may later be oxidized (donate its electrons) to drive ATP synthesis in a type of process called oxidative phosphorylation. • FADH 2 is covalently attached to succinate dehydrogenase, an enzyme which functions both in the citric acid cycle and the mitochondrial electron transport chain in oxidative phosphorylation.

• FADH 2, therefore, facilitates transfer of electrons to coenzyme Q (ubiquitone), which is the final electron acceptor of the reaction catalyzed by the succinate: ubiquinone oxidoreductase complex, also acting as an intermediate in the electron transport chain • Mitochondria in animals, including humans, possess two succinyl-Co. A synthetases: one that produces GTP from GDP, and another that produces ATP from ADP. • Plants have the type that produces ATP (ADP-forming succinyl-Co. A synthetase).

• Several of the enzymes in the cycle may be loosely associated in a multienzyme protein complex within the mitochondrial matrix. • The GTP that is formed by GDP-forming succinyl-Co. A synthetase may be utilized by nucleoside-diphosphate kinase to form ATP (the catalyzed reaction is GTP + ADP → GDP + ATP).
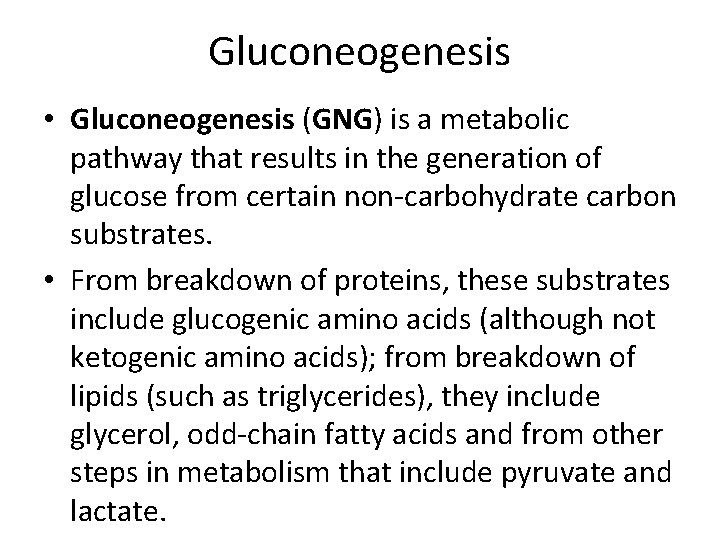
Gluconeogenesis • Gluconeogenesis (GNG) is a metabolic pathway that results in the generation of glucose from certain non-carbohydrate carbon substrates. • From breakdown of proteins, these substrates include glucogenic amino acids (although not ketogenic amino acids); from breakdown of lipids (such as triglycerides), they include glycerol, odd-chain fatty acids and from other steps in metabolism that include pyruvate and lactate.

• Although most gluconeogenesis occurs in the liver, the relative contribution of gluconeogenesis by the kidney is increased in diabetes and prolonged fasting. • Gluconeogenesis is one of several main mechanisms used by humans and many other animals to maintain blood glucose levels, avoiding low levels (hypoglycemia).
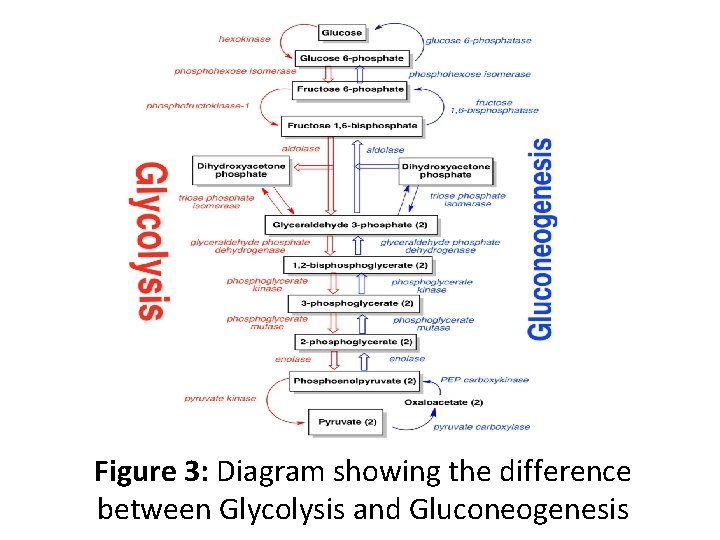
Figure 3: Diagram showing the difference between Glycolysis and Gluconeogenesis

Gluconeogenesis Pathway • Gluconeogenesis is a pathway consisting of a series of eleven enzyme-catalyzed reactions. • Gluconeogenesis begins in the mitochondria with the formation of oxaloacetate by the carboxylation of pyruvate. This reaction also requires one molecule of ATP, and is catalyzed by pyruvate carboxylase • .

• Oxaloacetate is reduced to malate using NADH, a step required for its transportation out of the mitochondria. • Malate is oxidized to oxaloacetate using NAD+ in the cytosol, where the remaining steps of gluconeogenesis take place. • Oxaloacetate is decarboxylated and then phosphorylated to form phosphoenolpyruvate using the enzyme phosphoenol pyruvate decarboxylase. • A molecule of GTP is hydrolyzed to GDP during this reaction.

• The next steps in the reaction are the same as reversed glycolysis. • However, fructose 1, 6 -bisphosphatase converts fructose 1, 6 -bisphosphate to fructose 6 -phosphate, using one water molecule and releasing one phosphate. This is also the rate-limiting step of gluconeogenesis. • Glucose-6 -phosphate is formed from fructose 6 phosphate by phosphoglucoisomerase(the reverse of step 2 in glycolysis). Glucose-6 -phosphate can be used in other metabolic pathways or dephosphorylated to free glucose.

• Whereas free glucose can easily diffuse in and out of the cell, the phosphorylated form (glucose-6 phosphate) is locked in the cell, a mechanism by which intracellular glucose levels are controlled by cells. • The final gluconeogenesis, the formation of glucose, occurs in the lumen of the endoplasmic reticulum, where glucose-6 -phosphate is hydrolyzed by glucose 6 -phosphatase to produce glucose and release an inorganic phosphate.

• Like two steps prior, this step is not a simple reversal of glycolysis, in which hexokinase catalyzes the conversion of glucose and ATP into G 6 P and ADP. Glucose is shuttled into the cytoplasm by glucose transporters located in the endoplasmic reticulum's membrane.

Pentose Phosphate Pathway • The pentose phosphate pathway (also called the phosphogluconate pathway and the hexose monophosphate shunt) is a metabolic pathway parallel to glycolysis. • It generates NADPH and pentoses (5 -carbon sugars) as well as ribose 5 -phosphate, a precursor for the synthesis of Nucleotides. • While the pentose phosphate pathway does involve oxidation of glucose, its primary role is anabolic rather than catabolic. • The pathway is especially important in red blood cells (erythrocytes).

• There are two distinct phases in the pathway. • The first is the oxidative phase, in which NADPH is generated, and the second is the non-oxidative synthesis of 5 -carbon sugars. For most organisms, the pentose phosphate pathway takes place in the cytosol; in plants, most steps take place in plastids.
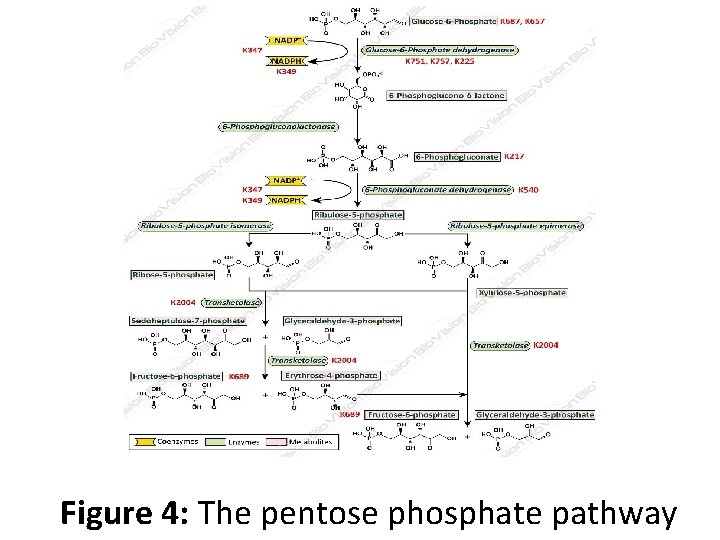
Figure 4: The pentose phosphate pathway

• In mammals, the pentose phosphate pathway occurs exclusively in the cytoplasm. • In humans, it is found to be most active in the liver, mammary glands, and adrenal cortex. The PPP is one of the three main ways the body creates molecules with reducing power, accounting for approximately 60% of NADPH production in humans. • One of the uses of NADPH in the cell is to prevent oxidative stress. • It reduces glutathione via glutathione reductase, which converts reactive H 2 O 2 into H 2 O by glutathione peroxidase. If absent, the H 2 O 2 would be converted to hydroxyl free radicals by Fenton chemistry, which can attack the cell.

• Erythrocytes, for example, generate a large amount of NADPH through the pentose phosphate pathway to use in the reduction of glutathione. • Hydrogen peroxide is also generated for phagocytes in a process often referred to as a respiratory burst

Glycogenolysis • Glycogenolysis is the breakdown of glycogen to glucose-1 -phosphate and glycogen. • Glycogen branches are catabolized by the sequential removal of glucose monomers via phosphorolysis, by the enzyme glycogen phosphorylase

Mechanism of Action • The overall reaction for the breakdown of glycogen to glucose-1 -phosphate is: • glycogen(n residues) + Pi ⇌ glycogen(n-1 residues) + glucose-1 -phosphate • Here, glycogen phosphorylase cleaves the bond linking a terminal glucose residue to a glycogen branch by substitution of a phosphoryl group for the α[1→ 4] linkage.

• Glucose-1 -phosphate is converted to glucose-6 phosphate (which often ends up in glycolysis) by the enzyme phosphoglucomutase. • Glucose residues are phosphorolysed from branches of glycogen until four residues before a glucose that is branched with a α[1→ 6] linkage. • Glycogen debranching enzyme then transfers three of the remaining four glucose units to the end of another glycogen branch. This exposes the α[1→ 6] branching point, which is hydrolysed by α[1→ 6] glucosidase, removing the final glucose residue of the branch as a molecule of glucose and eliminating the branch.

• This is the only case in which a glycogen metabolite is not glucose-1 -phosphate. The glucose is subsequently phosphorylated to glucose-6 -phosphate by hexokinase.
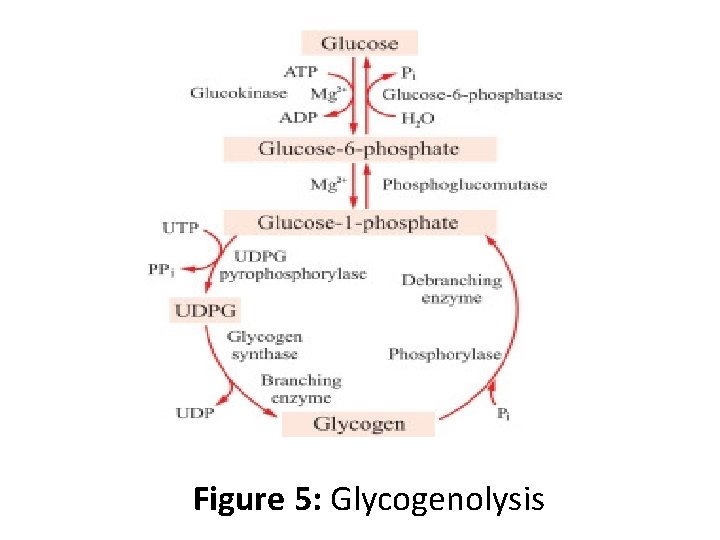
Figure 5: Glycogenolysis

Function of Glycogenolysis • Glycogenolysis takes place in the cells of the muscle and liver tissues in response to hormonal and neural signals. In particular, glycogenolysis plays an important role in the fight-or-flight response and the regulation of glucose levels in the blood. • In myocytes (muscle cells), glycogen degradation serves to provide an immediate source of glucose-6 -phosphate for glycolysis, to provide energy for muscle contraction.

• In hepatocytes (liver cells), the main purpose of the breakdown of glycogen is for the release of glucose into the bloodstream for uptake by other cells. • The phosphate group of glucose-6 -phosphate is removed by the enzyme glucose-6 phosphatase, which is not present in myocytes, and the free glucose exits the cell via GLUT 2 facilitated diffusion channels in the hepatocyte cell membrane.

- Slides: 39