Bayesian Brain Dynamic Causal Modelling DCM This material

Bayesian Brain: Dynamic Causal Modelling (DCM) This material was modified from Uta Noppeney et al. (Functional Imaging Lab, Wellcome Dept. of Imaging Neuroscience, Institute of Neurology, University College London)

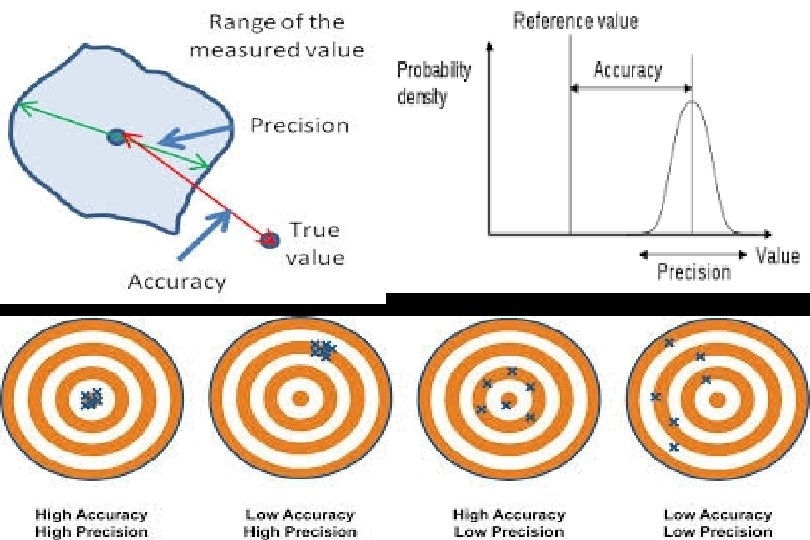
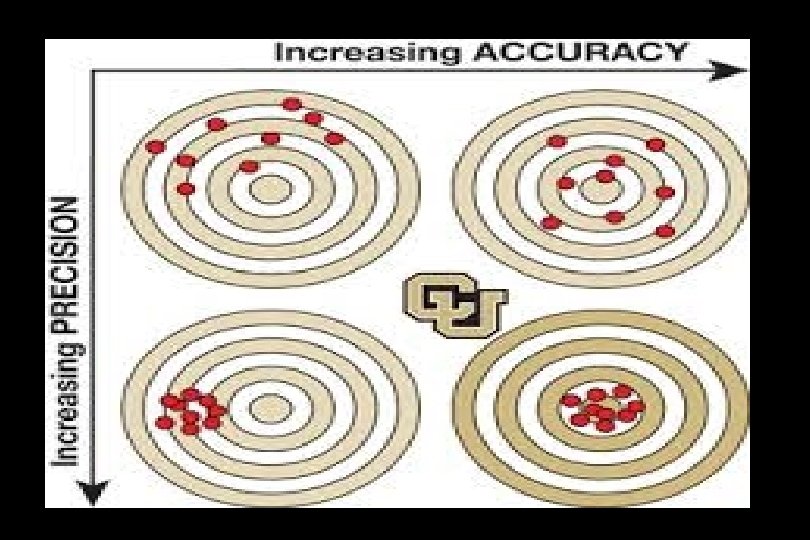
Bounded rationality

Bounded rationality System 1 § Fast § Intuitive, associative § heuristics & biases System 2 § Slow (lazy) § Deliberate, ‘reasoning’ § Rational
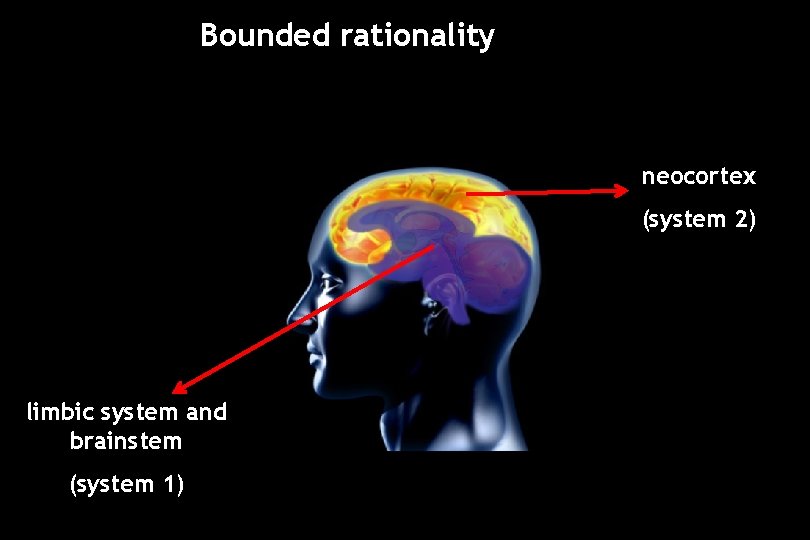
Bounded rationality neocortex (system 2) limbic system and brainstem (system 1)

System 1: very prone to biases § § § § § Seeing order in randomness Mental corner cutting Misinterpretation of incomplete data Halo effect False consensus effect Group think § Self serving bias § Sunk cost fallacy § Cognitive dissonance reduction § § § Confirmation bias Authority bias Small numbers fallacy In-group bias Recall bias Anchoring bias Inaccurate covariation detection Distortions due to plausibility

Bounded Rationality Critical Thinking The Small Numbers Problem of Individual Experience Decision Supports Research Prone to See Patterns Even in Random Data The “Human” Problem • Large Ns > individual experience • Controls reduce bias Evidence-Based Practice

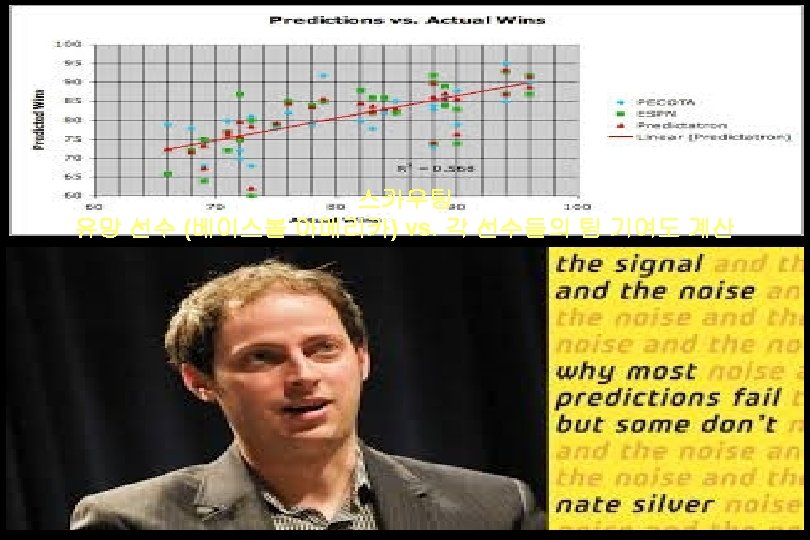
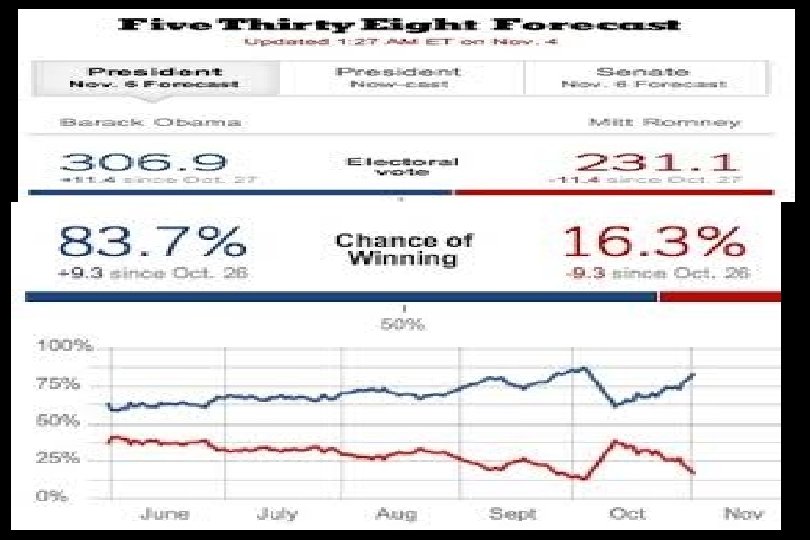
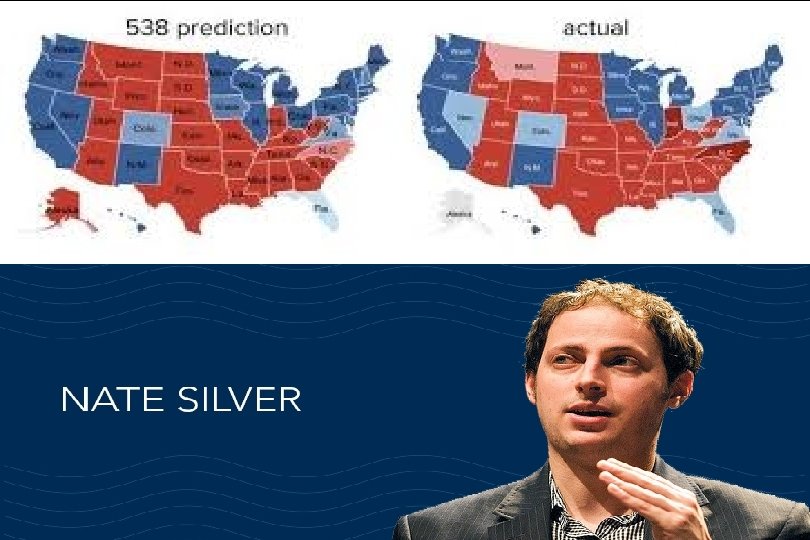
Evidence-based deciison-making “It’s hard to tell the signal from the noise. The story the data tell us is often the one we’d like to hear, and we usually make sure it has a happy ending. It is when we deny our role in the process that the odds of failure rise. ”



We don’t like uncertainty!

Nate Silver









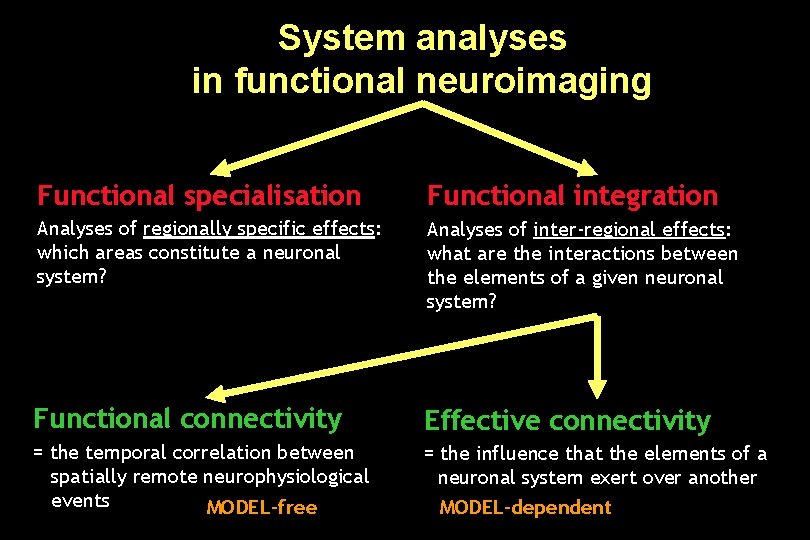
System analyses in functional neuroimaging Functional specialisation Functional integration Analyses of regionally specific effects: which areas constitute a neuronal system? Analyses of inter-regional effects: what are the interactions between the elements of a given neuronal system? Functional connectivity Effective connectivity = the temporal correlation between spatially remote neurophysiological events MODEL-free = the influence that the elements of a neuronal system exert over another MODEL-dependent

Approaches to functional integration • Functional Connectivity Eigenimage analysis and PCA Nonlinear PCA ICA • Effective Connectivity Psychophysiological Interactions MAR and State space Models Structure Equation Models Volterra Models Dynamic Causal Models
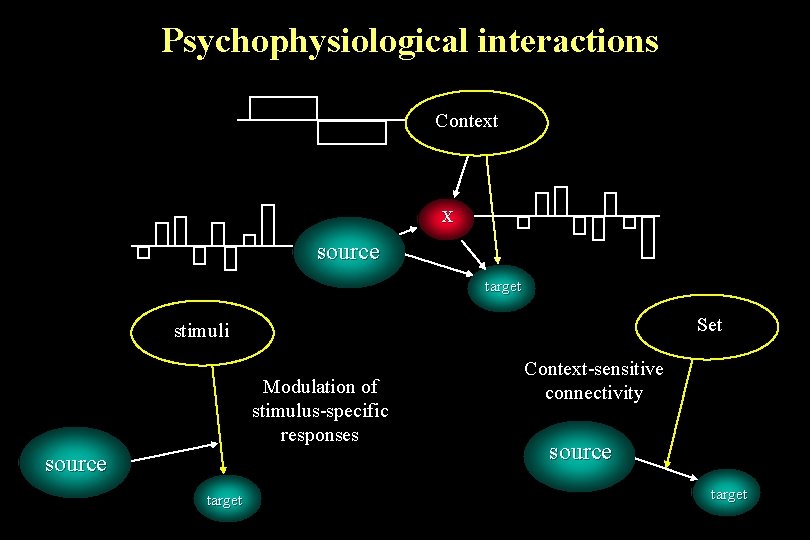
Psychophysiological interactions Context X source target Set stimuli Modulation of stimulus-specific responses source target Context-sensitive connectivity source target
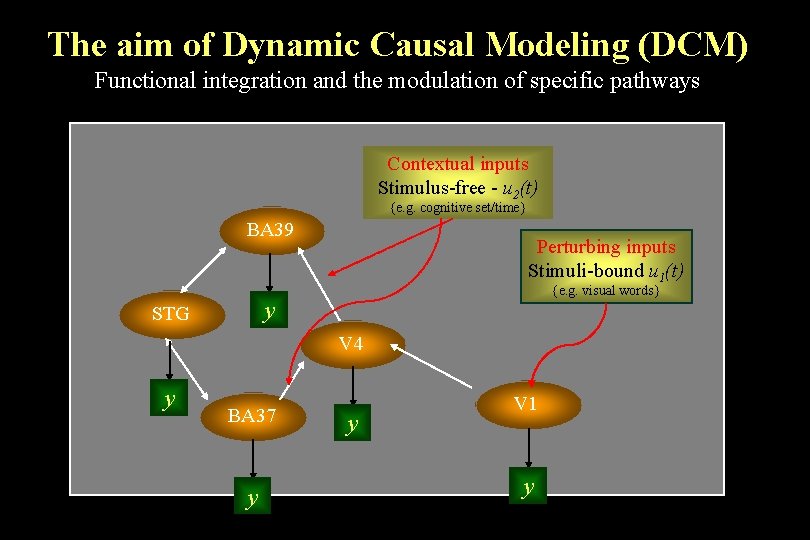
The aim of Dynamic Causal Modeling (DCM) Functional integration and the modulation of specific pathways Contextual inputs Stimulus-free - u 2(t) {e. g. cognitive set/time} BA 39 Perturbing inputs Stimuli-bound u 1(t) {e. g. visual words} y STG V 4 y BA 37 y y V 1 y
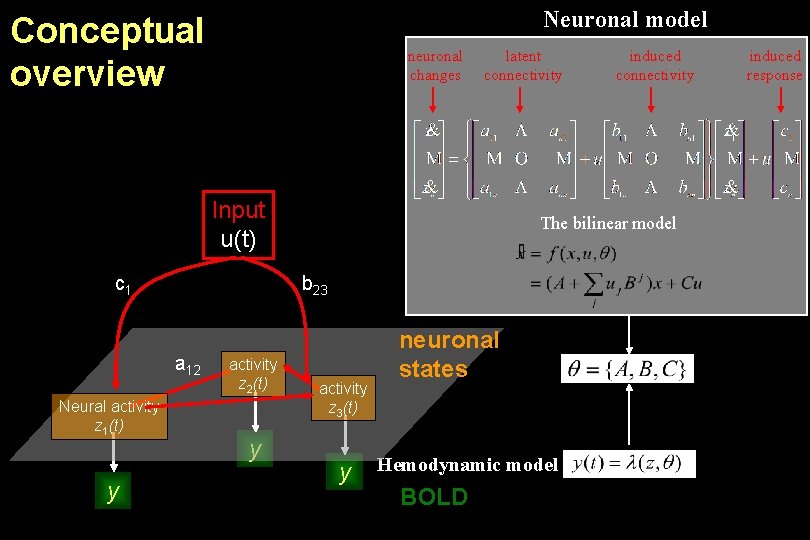
Neuronal model Conceptual overview neuronal changes latent connectivity Input u(t) c 1 Neural activity z 1(t) y The bilinear model b 23 a 12 activity z 2(t) y induced connectivity activity z 3(t) y neuronal states Hemodynamic model BOLD induced response

Rational statistical inference (Bayes, Laplace) Posterior probability Likelihood Prior probability Sum over space of hypotheses

Conceptual overview Models of Constraints on • Responses in a single region • Neuronal interactions • Connections • Biophysical parameters Bayesian estimation posterior likelihood ∙ prior
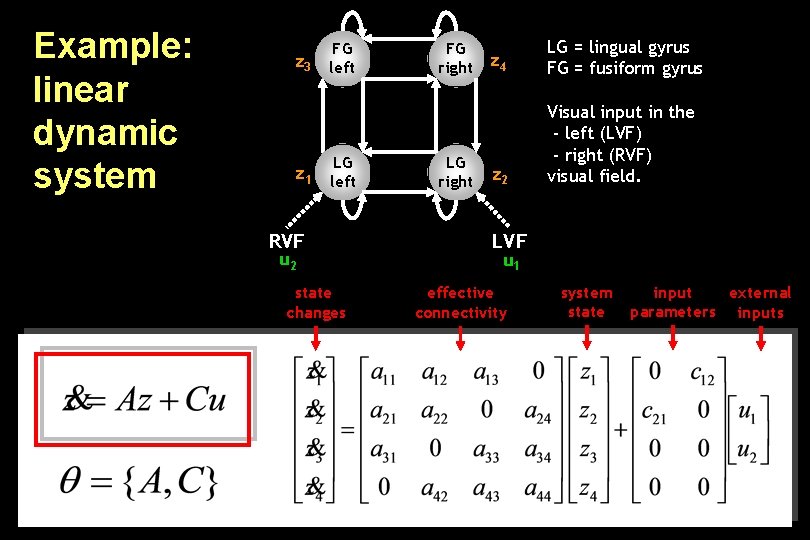
Example: linear dynamic system FG z 3 left z 1 LG left RVF u 2 state changes FG right LG right z 4 LG = lingual gyrus FG = fusiform gyrus z 2 Visual input in the - left (LVF) - right (RVF) visual field. LVF u 1 effective connectivity system state input external parameters inputs
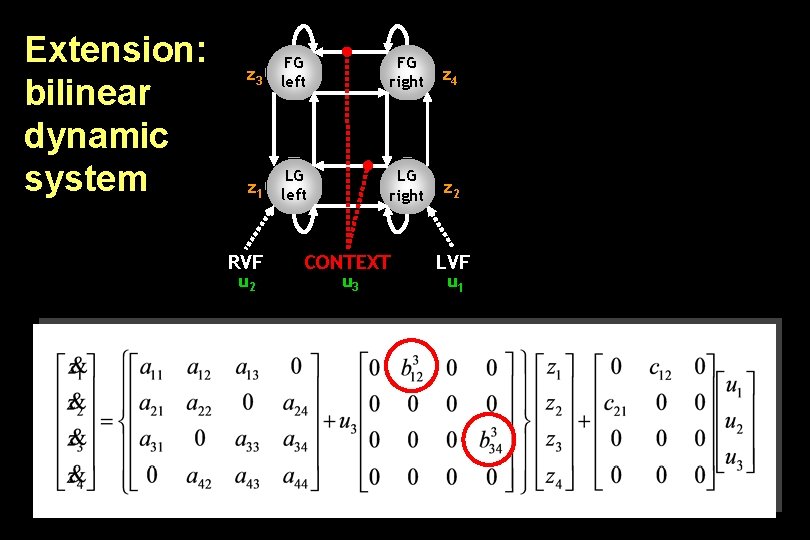
Extension: bilinear dynamic system z 3 FG left FG right z 4 z 1 LG left LG right z 2 RVF u 2 CONTEXT u 3 LVF u 1

Bilinear state equation in DCM state changes intrinsic connectivity modulation of system connectivity state direct inputs m external inputs
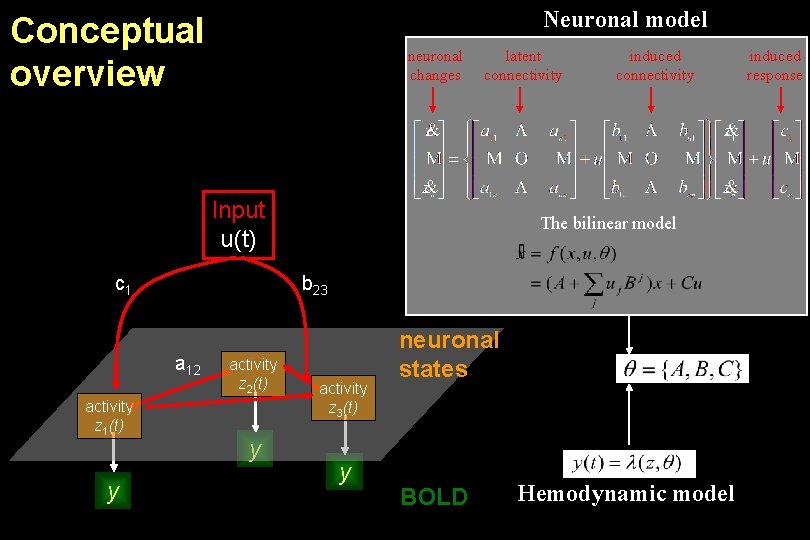
Neuronal model Conceptual overview neuronal changes latent connectivity Input u(t) c 1 y The bilinear model b 23 a 12 activity z 1(t) induced connectivity activity z 2(t) y activity z 3(t) y neuronal states BOLD Hemodynamic model induced response
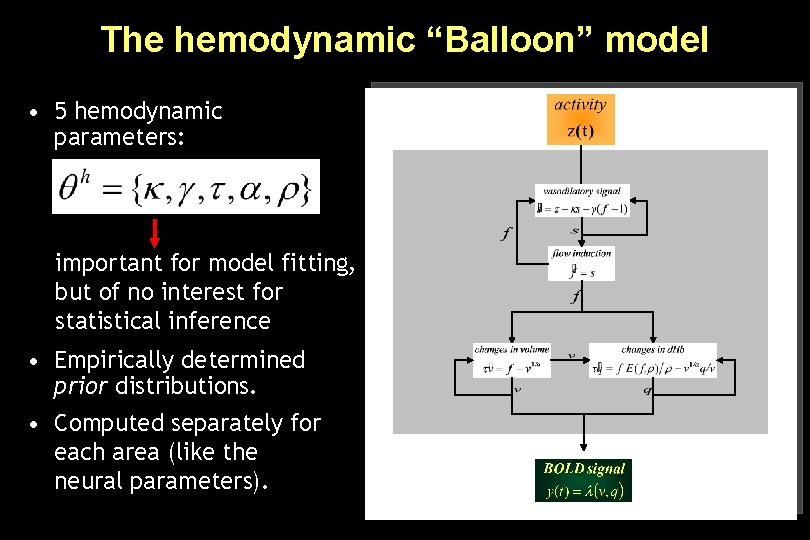
The hemodynamic “Balloon” model • 5 hemodynamic parameters: important for model fitting, but of no interest for statistical inference • Empirically determined prior distributions. • Computed separately for each area (like the neural parameters).
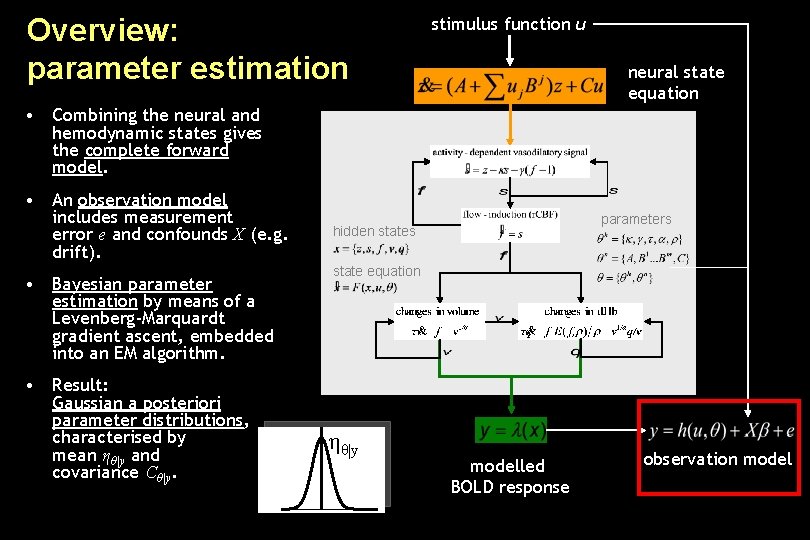
Overview: parameter estimation stimulus function u neural state equation • Combining the neural and hemodynamic states gives the complete forward model. • An observation model includes measurement error e and confounds X (e. g. drift). • Bayesian parameter estimation by means of a Levenberg-Marquardt gradient ascent, embedded into an EM algorithm. • Result: Gaussian a posteriori parameter distributions, characterised by mean ηθ|y and covariance Cθ|y. parameters hidden states state equation ηθ|y modelled BOLD response observation model

Overview: parameter estimation Models of Constraints on • Responses in a single region • Neuronal interactions • Connections • Biophysical parameters posterior likelihood ∙ prior Bayesian estimation
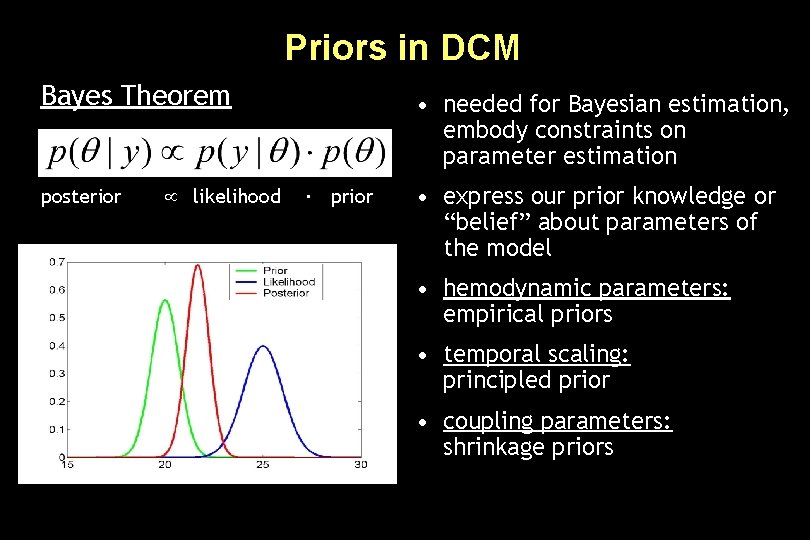
Priors in DCM Bayes Theorem posterior likelihood • needed for Bayesian estimation, embody constraints on parameter estimation ∙ prior • express our prior knowledge or “belief” about parameters of the model • hemodynamic parameters: empirical priors • temporal scaling: principled prior • coupling parameters: shrinkage priors

Priors in DCM • Principled priors: – System stability: in the absence of input, the neuronal states must return to a stable mode • Shrinkage priors for coupling parameters (η=0) → conservative estimates! – Constraints on prior variance of intrinsic connections (A): Probability <0. 001 of obtaining a non-negative Lyapunov exponent (largest real eigenvalue of the intrinsic coupling matrix) – Self-inhibition: Priors on the decay rate constant σ (ησ=1, Cσ=0. 105); these allow for neural transients with a half life in the range of 300 ms to 2 seconds • Temporal scaling: Identical in all areas by factorising A and B with σ (a single rate constant for all regions) : all connection strengths are relative to the self-connections.
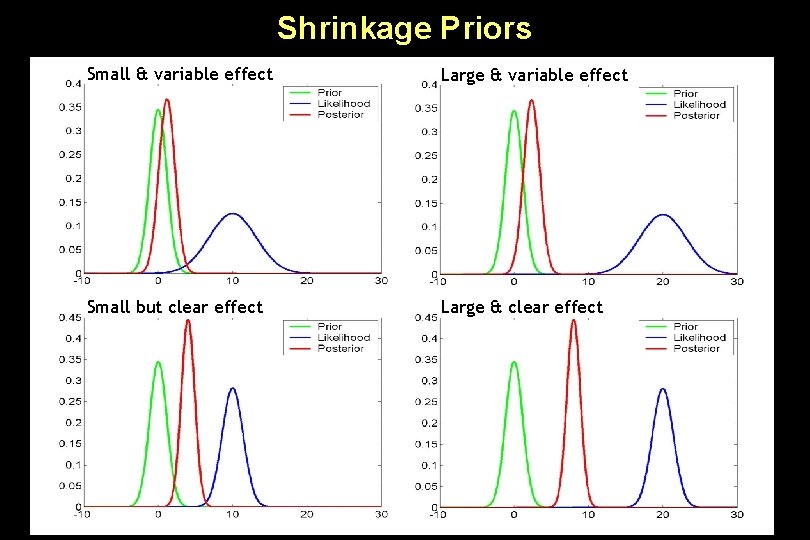
Shrinkage Priors Small & variable effect Large & variable effect Small but clear effect Large & clear effect

EM and gradient ascent • Bayesian parameter estimation by means of expectation maximisation (EM) – E-step: gradient ascent (Fisher scoring & Levenberg-Marquardt regularisation) to compute • (i) the conditional mean ηθ|y (= expansion point of gradient ascent), • (ii) the conditional covariance Cθ|y – M-step: Estimation of hyperparameters i for error covariance components Qi: • Note: Gaussian assumptions about the posterior (Laplace approximation)

Parameter estimation in DCM • Combining the neural and hemodynamic states gives the complete forward model: • Bayesian parameter estimation under Gaussian assumptions by means of EM and gradient ascent. • Result: Gaussian a posteriori parameter distributions with mean ηθ|y and covariance Cθ|y. • The observation model includes measurement error and confounds X (e. g. drift): ηθ|y
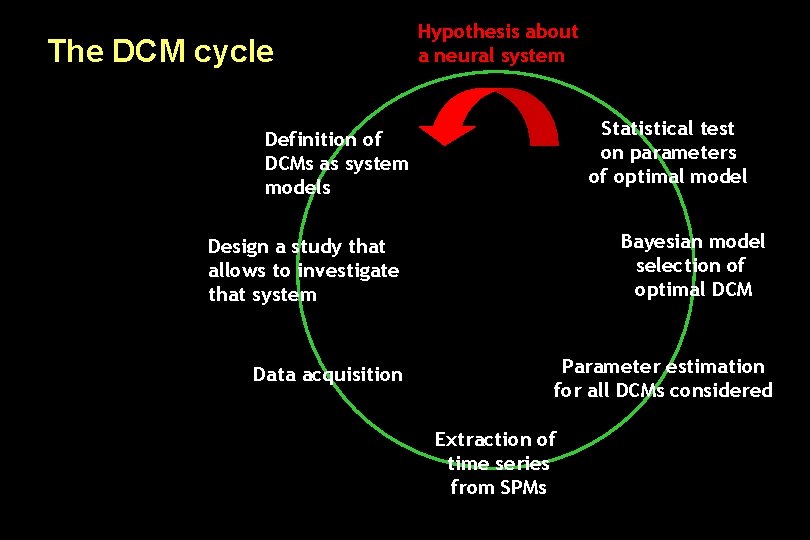
The DCM cycle Hypothesis about a neural system Statistical test on parameters of optimal model Definition of DCMs as system models Bayesian model selection of optimal DCM Design a study that allows to investigate that system Data acquisition Parameter estimation for all DCMs considered Extraction of time series from SPMs

Planning a DCM-compatible study • Suitable experimental design: – preferably multi-factorial (e. g. 2 x 2) – e. g. one factor that varies the driving (sensory) input – and one factor that varies the contextual input • Hypothesis and model: – define specific a priori hypothesis – Which alternative models? – which parameters are relevant to test this hypothesis? • TR: – as short as possible (optimal: < 2 s)
- Slides: 48