Battery Basics A guide to battery use in

Battery Basics A guide to battery use in engineering projects Thomas G. Cleaver University of Louisville Department of Electrical and Computer Engineering Nov. 14, 2016

References This presentation was developed using the following sources: • T. E. Bell, “Choosing the Best Battery for Portable Equipment, ” IEEE Spectrum, March, 1988, pp 30 -35. • Walt Kester, Joe Buxton, “SECTION 5, BATTERY CHARGERS, ” available at http: //www. analog. com/Uploaded. Files/Associated_Docs/43241673 Power_s ect 5. PDF#search=%22 batteries%20%22 discharge%20 profiles%22%22 • Custom Power Solutions, available at http: //www. mpoweruk. com/performance. htm • New Technology Batteries Guide (1998), available at http: //www. nlectc. org/rwscripts/rwisapi. dll/@JUSTNET. env? CQ_SESSION_ KEY=CUANXLBUKMIR&CQ_TPT_VIEW_DOC=YES&CQDOC_NUM=2 • Green Batteries, available at http: //www. greenbatteries. com/libafa. html • Steve Garland, Kyle Jamieson, “Battery Overview, ” available at: http: //nms. csail. mit. edu/fun/battery. ppt#268 • Harding energy Inc, available at http: //www. hardingenergy. com/ • Battery. University. com, available at http: //www. batteryuniversity. com/

Battery Terms 1 • Capacity: The charge a battery can hold in ampere-hours (Ah) or milliampere-hours (m. Ah) or the energy the battery can hold in watthours. • C: Charge or discharge rate. Battery capacity in Ah or m. Ah divided by 1 hour. Also know as C rate. • Charge life: The total capacity over the life of the battery (capacity x cycles). • Discharge rate: The maximum allowable load or discharge current. • End voltage: The voltage below which a battery will not operate satisfactorily. Also know as “final voltage. ” • Energy density: The energy storage capacity of a battery compared to its mass or volume. The higher the energy density, the better. • Memory effect: The tendency of some rechargeable batteries to lose capacity when not periodically totally drained – a particular problem in Ni. Cd batteries.

Battery Terms 2 • Primary battery: A disposable battery. • Polarity reversal: The reversal of the polarity of an overdischarged cell of a rechargeable battery in a series connection. If one cell in a series string discharges before the others, the discharged cell may reverse polarity. If the current is maintained, the reversed cell may be permanently damaged. • Secondary battery: A rechargeable (storage) battery. • Self-discharge: The loss of charge over time of a battery when it is unused. • Service life: The length of time a battery is expected to be usable. • Shelf life: The length of time a battery will retain useful charge when stored.

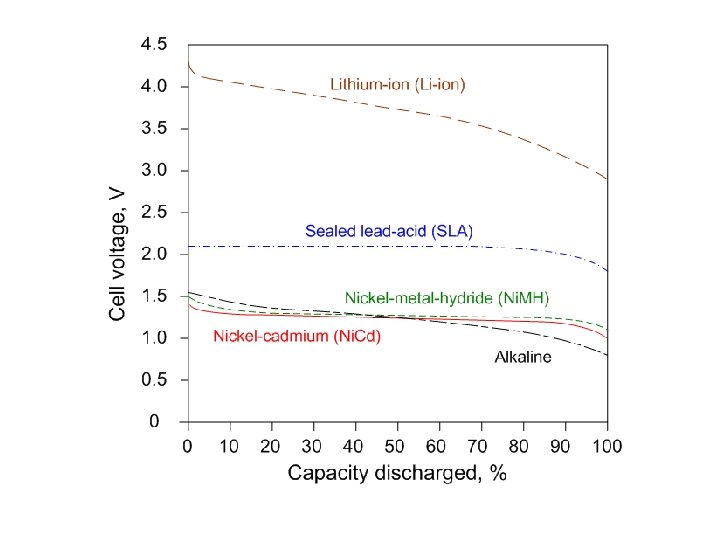
Primary (Disposable) Battery Types • Zinc-carbon: – “Ordinary” battery – Voltage decreases steadily during discharge • Zinc-alkaline: – “Alkaline” battery – Better than zinc-carbon – Voltage decreases steadily during discharge • Zinc-air: – Button cell hearing aid batteries – Voltage almost constant over useful life • Lithium ion: – High energy density – Voltage almost constant over useful life

Secondary (rechargeable) Battery Types • Sealed Lead-Acid (SLA): – Automobile batteries – Low cost – Lead is toxic; sulfuric acid is corrosive. • Nickel-Cadmium (Ni. Cd): – Inexpensive – Memory effect – Cadmium is toxic. • Nickel-metal-hydride (Ni. MH): – Moderately expensive – Voltage almost constant over useful life • Lithium ion (Li-ion): – – Expensive High energy density Voltage almost constant over useful life Dangerous if overcharged

Standard Sizes • Button – used in hearing aids and in other applications that require small size • Cylindrical – like AAA, C, D – all usually 1. 2 to 1. 5 V • Prismatic – like 9 V batteries • Rechargeable Li-ion does not typically come in standard cylindrical sizes.

Discharge and Voltage • The voltage of some batteries doesn’t change much as the battery is discharged, for example, Ni. Cd and Ni. MH. • The voltage of others drops off as the battery is discharged, for example, zinccarbon, and alkaline.

Discharge and Current • Battery capacity, usually expressed in m. Ah, is measured under specific conditions. • The higher the current, the less the effective capacity. • Example: A battery rated at 1500 m. Ah may be able to deliver 150 m. A for 10 hours, but it may not be able to actually deliver 1500 m. A for 1 hour.
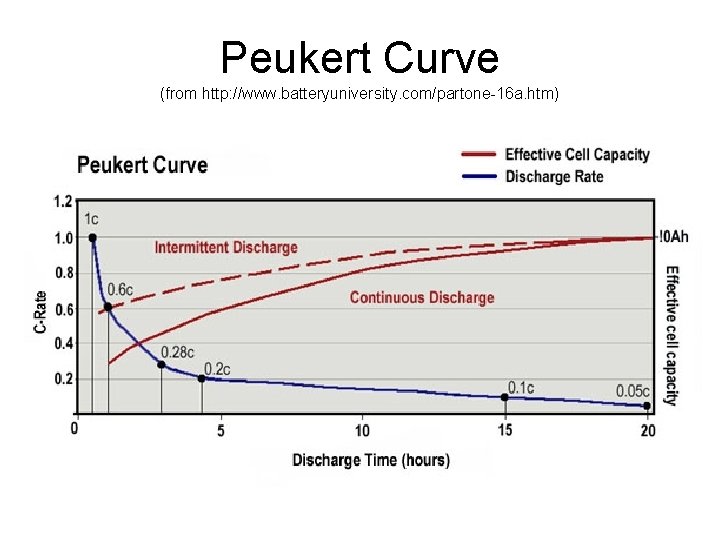
Peukert Curve (from http: //www. batteryuniversity. com/partone-16 a. htm)

C Rate Calculations • C = Rated capacity/ 1 hour • Example: A 2800 m. Ah Ni. MH battery has a C of 2800 m. A. • Batteries can be tested at various multiples of C. • Example: For the 2800 m. Ah battery, C/4 would be 700 m. A; 3 C would be 8400 m. A.

Voltage Dependence on Current • Batteries are not ideal devices – They have internal resistance. • Vloss = IRinternal Battery Type Ni. Cd 1. 2 V AA Typical Internal Resistance (milliohms) 30 Ni. MH 1. 2 V AA 150 Li-ion 3. 6 V 320 Alkaline 1. 5 V AA 150

Maximum and Suggested Drain Battery Type Max Drain Alkaline SLA . 5 C. 2 -5 C Suggested Drain <. 2 C Ni. Cd Ni. MH Li-ion 2 -20 C. 5 -5 C 1 -2 C <. 5 C <1 C

Batteries in Series • Batteries should be identical. • Total voltage = Voltage of each cell x number of cells • When using rechargeable batteries in series, beware of deep discharge because of polarity reversal.

Batteries in Parallel • Batteries should be identical. • Total current = Current of each cell x number of cells • Usually a bad idea • Good batteries may discharge through bad battery.

Illumination Economics Incandescent, Compact Fluorescent (CFL), and LED lighting characteristics Cost of bulb Lumens Efficiency Lifetime $1 840 2% 1 K hours (~ 1 Month) 13 W CFL $2 825 9% 10 k hours (~ 1 year) 10 W LED $6 810 12 % 50 k hours (~ 5 years) Type 60 W Incandescent

Total Cost by Bulb Type Cost for purchase of bulb(s) and for electrical energy @ 10 ₵/k. Wh. Type 1 month 1 year 5 years 60 W Incandescent $5 $62 $307 13 W CFL $3 $13 $67 10 W LED $7 $15 $50 But this assumes you turn the light on and never turn it off until it blows out and you replace it.

What’s so Bad about CFLs? • • • On/off cycling shortens lifetime. They are sensitive to physical shock and breakage. Most CFLs are not dimmable. Some people don’t like the quality of the light (too harsh). Some CFLs take time (~ 30 seconds) to achieve maximum light output – not a problem for newer CFLs. • CFLs contain a small amount of mercury (a disposal issue). • Low temperature reduces CFL light output (an outdoor use issue). • High temperature shortens CFL lifetime (a luminaire issue).
- Slides: 19