Batteries the Chemistry Batteries use a chemical reaction
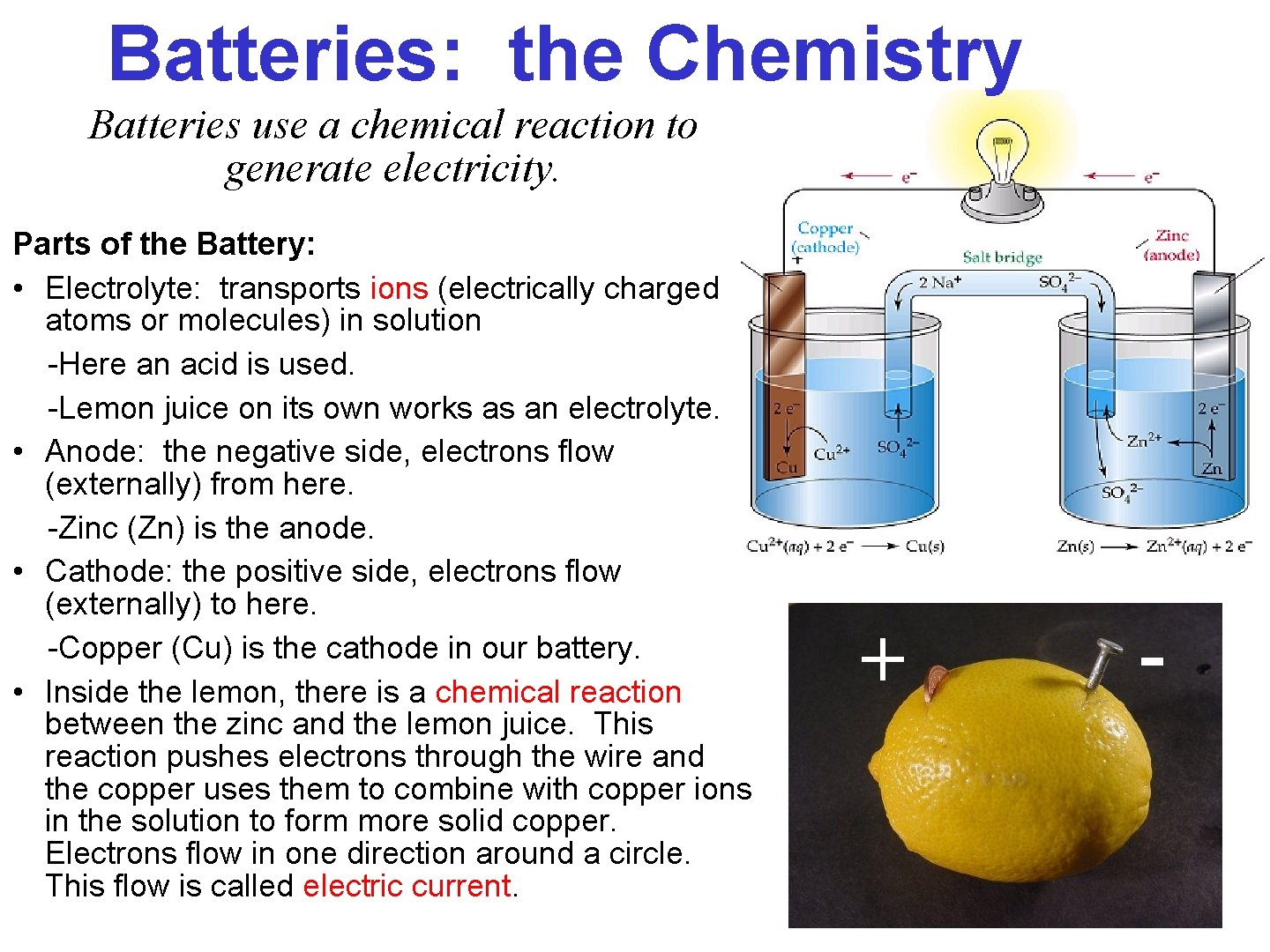
Batteries: the Chemistry Batteries use a chemical reaction to generate electricity. Parts of the Battery: • Electrolyte: transports ions (electrically charged atoms or molecules) in solution -Here an acid is used. -Lemon juice on its own works as an electrolyte. • Anode: the negative side, electrons flow (externally) from here. -Zinc (Zn) is the anode. • Cathode: the positive side, electrons flow (externally) to here. -Copper (Cu) is the cathode in our battery. • Inside the lemon, there is a chemical reaction between the zinc and the lemon juice. This reaction pushes electrons through the wire and the copper uses them to combine with copper ions in the solution to form more solid copper. Electrons flow in one direction around a circle. This flow is called electric current.

Serial -large voltage Parallel – large current Batteries: their Applications Batteries can be run in parallel or series. • Parallel batteries operates longer with more current capacity. • Devices that use smaller amounts of electricity for a long period of time may run batteries in parallel. • Series batteries provide higher voltage. • Devices that need more voltage use batteries in series. The above picture shows how batteries produce a stronger current when connected in parallel. • Some batteries can reverse the current to undo the reaction that causes the electrons to flow. • This makes the chemicals in the battery go back to their original states, so the battery can be used again. These rechargeable batteries • Rechargeable batteries are used in cell phones, can be used multiple times. cameras, cars, and other electrical appliances.
- Slides: 2