Batch Control Process Control Development Ltd Batch Control

Batch. Control Process Control Development Ltd Batch. Control Ltd Hungary, 1043 Budapest Dugonics u. 11. www. batchcontrol. hu Dr. Lajos Böröcz lborocz@batchcontrol. hu Founder member of Hungarian Batch Forum F. Molnar 1

About Batch. Control Ltd Mission: „Competence in batch control”, It means: our company has specialized in the problems of batch control area, and has become the specialist to solve them. F. Molnar 2

Batch. Control Ltd • Foundation: 1991 • Origin: CHINOIN Pharmaceutical Factory (now Sanofi-Aventis) • Employees : 20 • Engineers: 17 • Capital: 480 000 Є • Sales volume: 1 000 Є F. Molnar 3

Possible Partners We look for: Pharmaceutical and chemical production companies applying batch control processes for reactors Food processing companies (e. g. beer), building material production companies (e. g. cement) applying similar technologies Engineering companies working for the pharma industry ( Like Jacobs from Italy, Tecnimont from Belgium, Novo Nordisk Engineering from Denmark- we have no contact to them ) F. Molnar 4

Our strengths • • Batch (control) analysis Batch control strategies – – – • Control Systems Validation – – – • Batch algorithms Recipe control Operations PLC programing DCS programing Protocols Test practices including software tests Installation Qualifications Operational Qualifications 21 CFR Part 11 Project Execution Procedure – Based on Good Automated Manufacturing Practice (GAMP) – Based on Baseline recommendations – Based on validation requirements • Integration of package units F. Molnar 5

Activity • Desing • Construction and building • Control System programme design • Quality assurance • Development • Explosion proof practice • Maintenance F. Molnar 6

Design • • Studies, Basic instrumentation design, Construction plans, Execution plans. F. Molnar 7

Construction and building • Production of control panels. • Installation of instruments both in the production area and in the instrument rooms. • Putting in operation of instrumentalisation elements. • Tests • Configuration of process control systems (DCS/PLC/SCADA) F. Molnar 8

Validation • • • Validation before installation. Installation validation. Validation of the operation. Software validations. GAP analysis Risk analysis F. Molnar 9

Development • Solution of unique process control problems • Development of special SWs • Simulations, application of models • Training F. Molnar 10

Explosion proof activities • 7 certified experts F. Molnar 11

Main customers – Sanofi-Aventis CHINOIN, – Richter Gedeon Chemical Works, – EGIS Pharmaceutical Works (major shareholder is the French Servier Group), – Glaxo. Smith. Kline Biologicals, – VALEANT (ICN), – Biogal, F. Molnar 12

References – CHINOIN • • • Control systems of CHEMIFLEX reactors (validated) Process control systems of active ingredient (TD) production (validated) Process control system of multipurpose plant (validated) Enlarging of process control system of multipurpose plant (validated) Process control system of active ingredient (SR) production (validated) Process control system of pilot plant (validated) Control system of climate cabinets of PW systems. Instrumentation of Prostaglandin plant Process control system of active ingredient (Kemia 2) production (21 CFR) Process control system of active ingredient (Kemia 1) production (21 CFR) Process control system of Guedu dryer (Kemia 2) (21 CFR) Above projects are based on Emerson’s Delta. V DCS (Distributed Control System). F. Molnar 13

References – RICHTER • Process control system of WFI (old water system)(validated) • Measurement of TOC of PW/WFI water • Planning of process control system of new WFI system (validated) • Process control system of active ingredient (Kemia 3) production (validated) • Process control system of dissolving equipment (validated) • CHEMIFLEX system (Kemia 3) F. Molnar 14

References – others • Pharma. Park - Validation activity of building management system (21 CFR Part 11). • Glaxo. Smith. Kline Biologicals – Enviroment monitoring system • Glaxo. Smith. Kline Biologicals – Vaccine Plant • EGIS – – Producing of active ingredients under pressure. – Low volume active ingredient production instrumentation plans – Active ingredients production (resolving plant) – Glatt dryer – Lenticular homogenizator F. Molnar 15
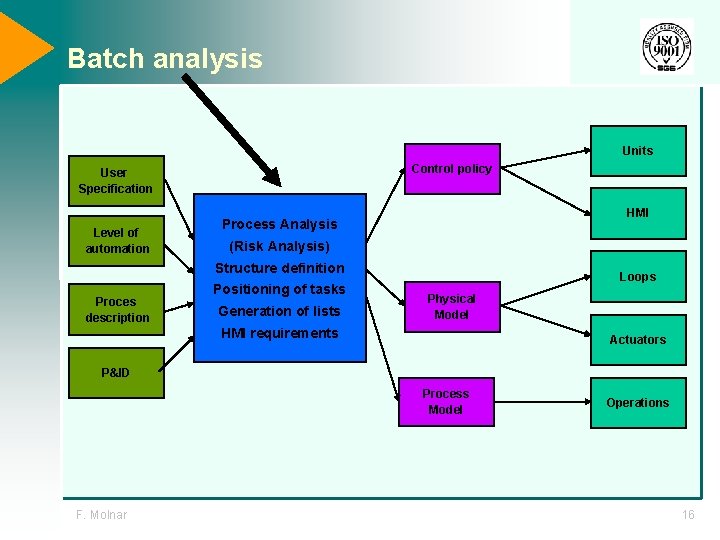
Batch analysis Units Control policy User Specification Level of automation HMI Process Analysis (Risk Analysis) Structure definition Proces description Positioning of tasks Generation of lists Loops Physical Model HMI requirements Actuators P&ID Process Model F. Molnar Operations 16

Integration of package units based on S 88. 01 models Control tasks can be allocated to: • central control system (e. g. DCS) • control device of Package Unit (PLC) F. Molnar 17

Specialities • High accuracy of heat control • Compact solution of thermo block (newly invented, under registration) F. Molnar 18
Trend 1 F. Molnar 19

Highest publications success Lipták, B. G. and Molnár, F. Instrument Engineers' Handbook, Process Control Volume, Reactor Control, 3 rd Edition, Chilton Book Company, 1995. F. Molnar 20
- Slides: 20