BASKET LATE Trial Basel Stent Costeffectiveness TrialLate Thrombotic

BASKET LATE Trial Basel Stent Cost-effectiveness Trial-Late Thrombotic Events (BASKET LATE) Trial Presented at The American College of Cardiology Scientific Session 2006 Presented by Dr. Matthias E. Pfisterer

BASKET LATE Trial: Background • In the original BASKET trial, a rather complex patient population was enrolled and randomized to receive one of the following: a bare metal stent, paclitaxeleluting stent, or sirolimus-eluting stent and the cost effectiveness of drug-eluting stents vs. bare metal stents was evaluated. • In the BASKET LATE trial, the 743 patients from the BASKET trial who were MACE free at six months were followed for an additional 12 months after the cessation of clopidogrel treatment. • The BASKET LATE trial pooled the paclitaxel- and sirolimus-eluting stent groups into one drug eluting stent group for analysis. • The goal of BASKET LATE was to evaluate late thrombotic events among patients treated with drug eluting stents vs. bare metal stents after clopidogrel discontinuation. www. Clinical trial results. org Presented at ACC 2006

BASKET LATE Trial: Study Design 743 patients all undergoing PCI irrespective of indication for PCI and without target vessel diameter ≥ 4 mm, restenotic lesions, or an event during the on-clopidogrel phase. Original BASKET study randomized patients in 1: 1: 1 strategy. Present study pooled DES patients into one group. 21% female, 21% ST-elevation MI, 37% unstable angina, 42% stable angina, 67% with multivessel disease, 51% with LAD culprit lesions, average 1. 9 stents per patient, mean age 63 years, mean follow-up 18 months. Concomitant medications: aspirin indefinitely. Drug-eluting stents (DES) (pooled paclitaxel and sirolimus DES groups) n=499 g g Bare metal stents (BMS) n=244 Primary Endpoint: Composite cardiac death or nonfatal MI. Other Endpoints: - “Thrombosis-related events”: - angiographically documented stent thrombosis - cardiac death/ target vessel MI - Target vessel revascularization (TVR) www. Clinical trial results. org Presented at ACC 2006

BASKET LATE Trial: Primary Composite Endpoint Composite of Cardiac death or nonfatal MI (%) % patients p=0. 01 www. Clinical trial results. org • In the year following clopidogrel discontinuation, the primary composite endpoint of cardiac death or MI occurred significantly more frequently in the DES group than in the BMS group (4. 9% vs. 1. 3%, p=0. 01). Presented at ACC 2006
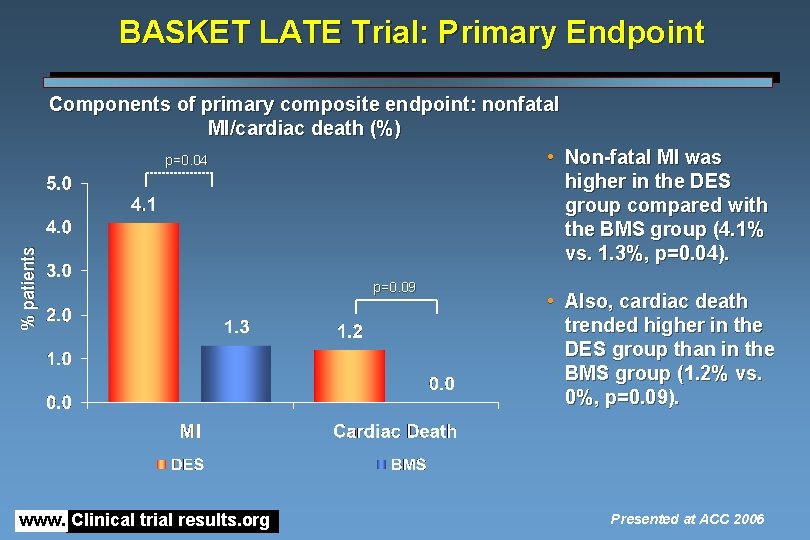
% patients BASKET LATE Trial: Primary Endpoint Components of primary composite endpoint: nonfatal MI/cardiac death (%) • Non-fatal MI was p=0. 04 higher in the DES group compared with the BMS group (4. 1% vs. 1. 3%, p=0. 04). www. Clinical trial results. org p=0. 09 • Also, cardiac death trended higher in the DES group than in the BMS group (1. 2% vs. 0%, p=0. 09). Presented at ACC 2006
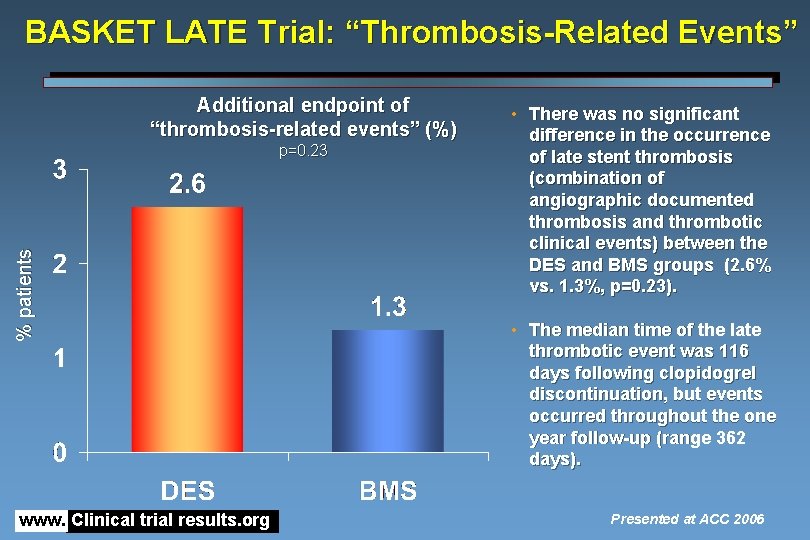
BASKET LATE Trial: “Thrombosis-Related Events” Additional endpoint of “thrombosis-related events” (%) % patients p=0. 23 www. Clinical trial results. org • There was no significant difference in the occurrence of late stent thrombosis (combination of angiographic documented thrombosis and thrombotic clinical events) between the DES and BMS groups (2. 6% vs. 1. 3%, p=0. 23). • The median time of the late thrombotic event was 116 days following clopidogrel discontinuation, but events occurred throughout the one year follow-up (range 362 days). Presented at ACC 2006

BASKET LATE Trial: TVR Restenosis driven TVR (%) % patients p=0. 21 www. Clinical trial results. org • Also, there was no difference in restenosis driven TVR among the two groups (4. 5% vs. 6. 7%, p=0. 21). Presented at ACC 2006

BASKET LATE: Safety MACE (%) p=0. 53 % patients • MACE rates were no different between DES and BMS (9. 3% vs. 7. 9%, p=0. 53). www. Clinical trial results. org Presented at ACC 2006

BASKET LATE Trial: Limitations • The present study could not identify major differences in thrombosis-related events because it was underpowered. www. Clinical trial results. org Presented at ACC 2006

BASKET LATE Trial: Summary • Among patients with coronary artery disease treated with PCI, use of a drug-eluting stent was associated with significantly higher rates of cardiac death or MI compared with a bare metal stent in the year following clopidogrel discontinuation. • Many trials have demonstrated a reduction in target lesion revascularization with DES compared with BMS in recent years, but none has ever demonstrated an effect on the hard endpoints of death or MI. • Restenosis, while not desirable, is not an independent correlate of subsequent mortality. The present study showed a more than threefold increase in death or MI with DES in the year after clopidogrel discontinuation. Late stent thrombosis is a serious and often fatal complication. www. Clinical trial results. org Presented at ACC 2006

BASKET LATE Trial: Summary cont. • Clopidogrel is generally prescribed for the first 6 months following stent placement, along with aspirin indefinitely. These data suggest the 6 month anti-platelet regimen of clopidogrel may not be long enough to provide adequate protection from late thrombosis with DES. • It is unknown if a better strategy would be a longer duration of clopidogrel, a more potent anti-platelet drug, or a different drugelution kinetic pattern. • The authors noted that for every 100 patients treated with a DES, 3. 3 cases of cardiac death or MI are induced for a reduction of 5 cases of target lesion revascularization. Further study on late thrombotic events with DES is strongly warranted given these findings. www. Clinical trial results. org Presented at ACC 2006
- Slides: 11