Basics of Pharmacokinetics Level Intermediate Version No 1

Basics of Pharmacokinetics Level : Intermediate Version No: 1 Version Date: December 2013 Prepared by: Balram Chowbay, Ph. D Principal Pharmacologist Clinical Pharmacology Lab National Cancer Center Narrated by: Natalia Sutiman

Disclaimer/Liability • The information provided in the VAP is made available in good faith and is derived from sources believed to be reliable and accurate at the time of release. • The materials presented on the VAP may include links to external Internet sites. These external information sources are outside the control of Duke-NUS. The user of the Internet links is responsible for making his or her own decision about the accuracy, reliability and correctness of the information found. • In no event shall Duke-NUS be liable for any indirect, special, incidental, or consequential damages arising out of any use of reliance of any information contained in the VAP. Nor does Duke-NUS assume any responsibility for failure or delay in updating or removing the information contained in the VAP. • Moreover, information provided on the VAP does not constitute medical advice or treatment nor should it be considered as a replacement of the patient/physician relationship or a physician’s professional judgment. Duke. NUS expressly disclaims all liability for treatment, diagnosis, decisions and actions taken or not taken in reliance upon information contained in the VAP. This work is licensed under a Creative Commons Attribution-Non. Commercial-No. Derivs 3. 0 Unported License To view a copy of this license, visit [http: //creativecommons. org/licenses/by-nc-nd/3. 0/]

Financial Disclosures (past 3 years) • No Disclosures
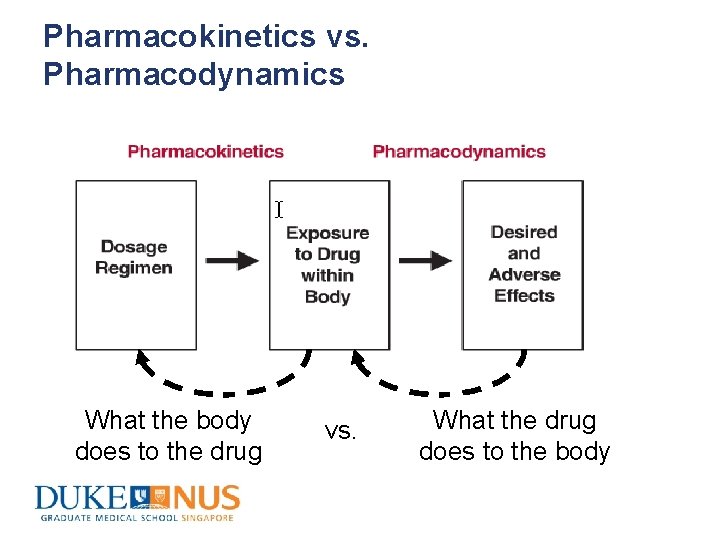
Pharmacokinetics vs. Pharmacodynamics What the body does to the drug vs. What the drug does to the body
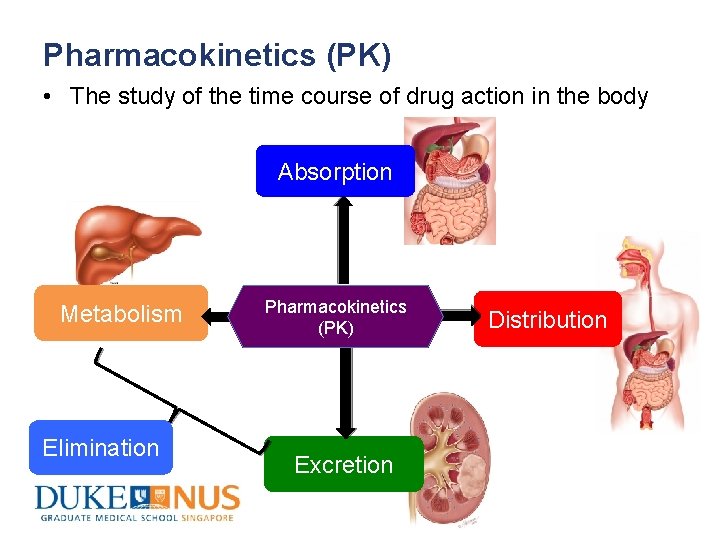
Pharmacokinetics (PK) • The study of the time course of drug action in the body Absorption Metabolism Elimination Pharmacokinetics (PK) Excretion Distribution
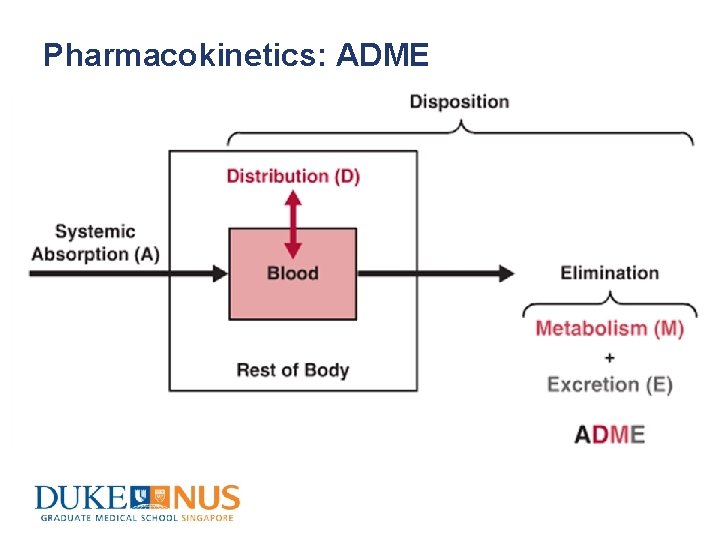
Pharmacokinetics: ADME
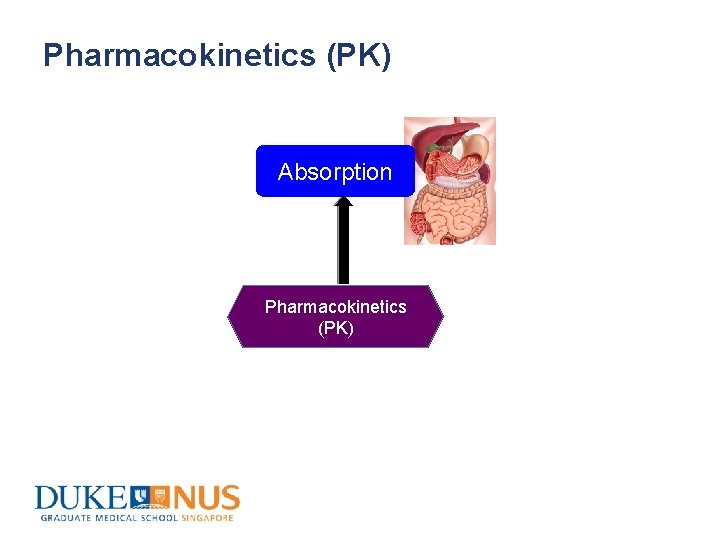
Pharmacokinetics (PK) Absorption Pharmacokinetics (PK)

Routes of Drug Administration Via alimentary canal Intravenous Oral Buccal Rectal Sublingual Other routes Intranasal Inhalation Transdermal Subcutaneous Intramuscular
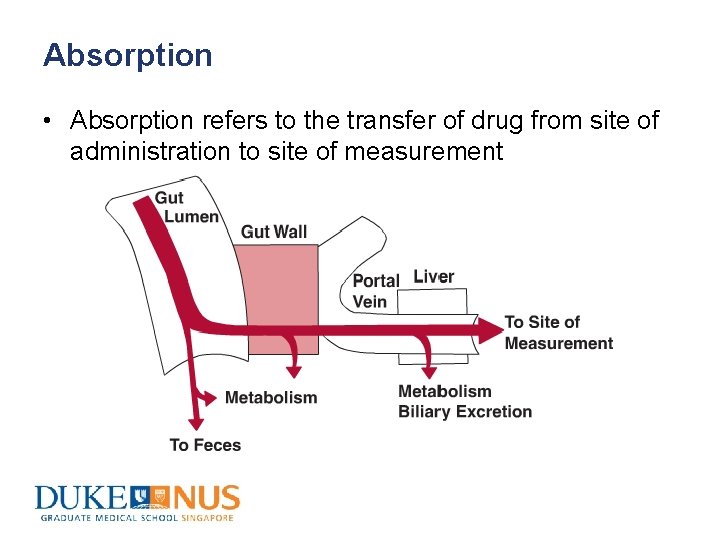
Absorption • Absorption refers to the transfer of drug from site of administration to site of measurement
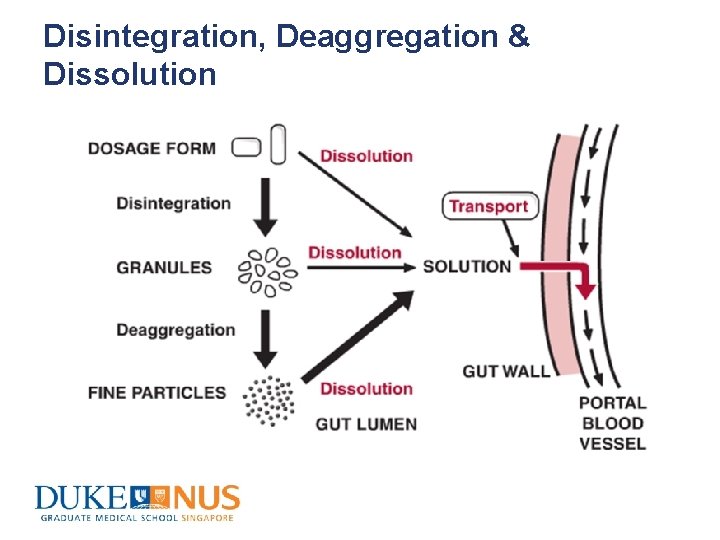
Disintegration, Deaggregation & Dissolution

Factors influencing absorption 1. Drug properties – Drug formulation and dosage form – Molecular size of a drug – Lipophilicity and polarity of drug compound 2. Properties of absorption site – – – Blood flow to absorption site p. H of the absorption site Area of absorption site Permeability of the membrane (e. g. BBB, gut wall) Presence of drug transporters

Factors influencing absorption 3. Other factors – – Food intake Presence of disease states and other abnormalities Co-administration with other drugs First-pass loss/ pro-drugs
Pharmacokinetics (PK) Absorption Pharmacokinetics (PK) Distribution
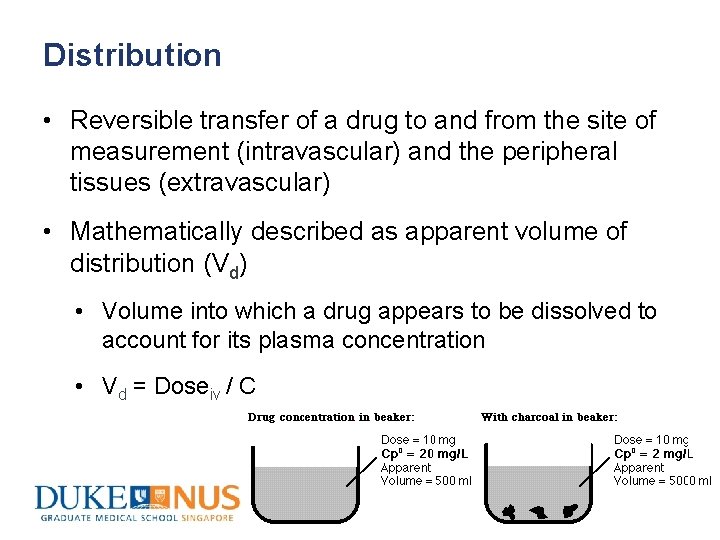
Distribution • Reversible transfer of a drug to and from the site of measurement (intravascular) and the peripheral tissues (extravascular) • Mathematically described as apparent volume of distribution (Vd) • Volume into which a drug appears to be dissolved to account for its plasma concentration • Vd = Doseiv / C

Volume of Distribution (Vd) • Vd does not refer to actual physiological volume – Vd of digoxin = 420 L – Vd of chloroquine = 13000 L • Large Vd indicates that the drug distributes extensively into body tissues and fluids while a small Vd indicates limited drug distribution, mostly only in plasma

Factors influencing distribution • Blood perfusion • Presence of influx and efflux transporters on membrane surface • Lipid solubility (lipid: water partition coefficient) • p. Ka of the drug and regional p. H • Plasma protein binding • Tissue protein binding • Disease states
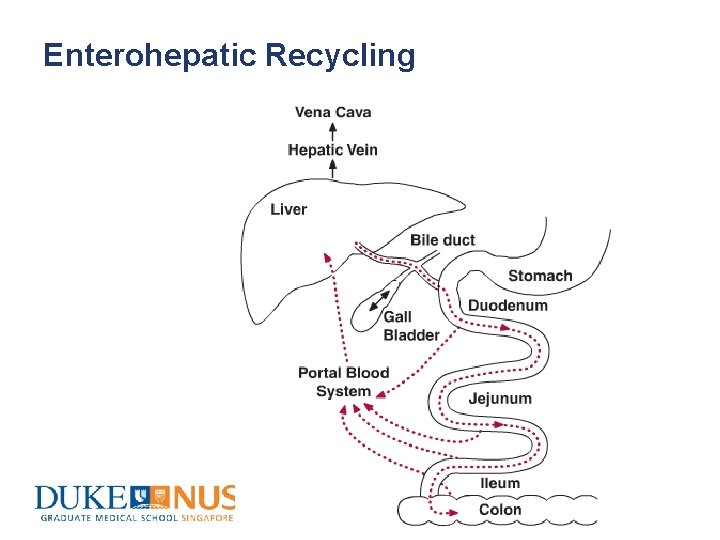
Enterohepatic Recycling
Pharmacokinetics (PK) Absorption Metabolism Elimination Pharmacokinetics (PK) Excretion Distribution

Elimination • Refers to the irreversible loss of drug compound from the site of measurement • Includes both metabolism and excretion Metabolism Metabolites Drug in the body Excretion Unchanged drug in urine

Routes of Elimination • Organs involved in drug elimination: liver, kidney, lung, skin, walls of the gut • Terms to describe elimination – Clearance: Volume of fluid presented to the eliminating organ that is completely cleared of drug each minute. – Elimination half-life: time taken for the concentration of the drug to fall by one half.
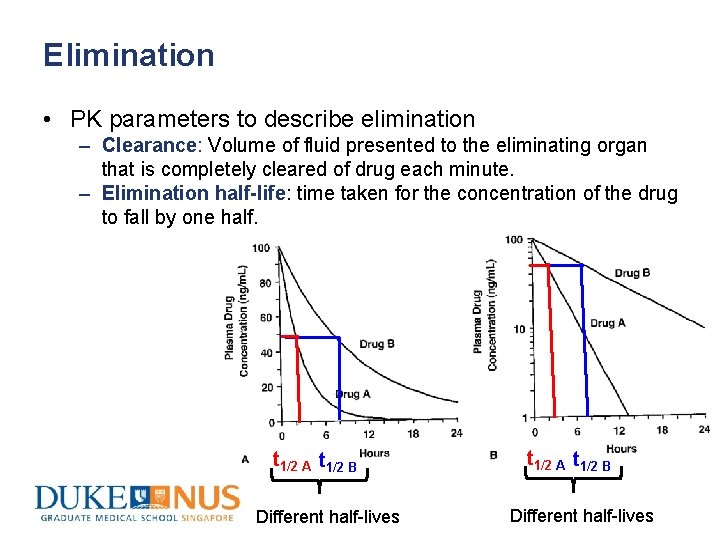
Elimination • PK parameters to describe elimination – Clearance: Volume of fluid presented to the eliminating organ that is completely cleared of drug each minute. – Elimination half-life: time taken for the concentration of the drug to fall by one half. t 1/2 A t 1/2 B Different half-lives
Pharmacokinetics (PK) Absorption Metabolism Pharmacokinetics (PK) Distribution

Metabolism • Refers to the conversion of one chemical species to another • Aims to convert less polar (more lipid soluble) parent drugs to more polar entities – Most hydrophilic (polar) drugs are excreted unchanged e. g. streptomycin, neostigmine • Also includes the bio-activation of pro-drugs into their active metabolites • Organs involved: Liver, gastrointestinal tract, kidney, lungs

Phases of Drug Metabolism • Phase I reactions – Convert parent drug into more polar metabolite(s) by adding or unmasking functional groups (-OH, -NH 2, COOH, etc) – Usually precede phase II reactions • Phase II reactions – Conjugate parent drug or metabolite(s) with endogenous substrate (glucuronic acid, sulfate, acetate, etc) to further increase aqueous solubility – Resulting metabolites are pharmacologically inactive
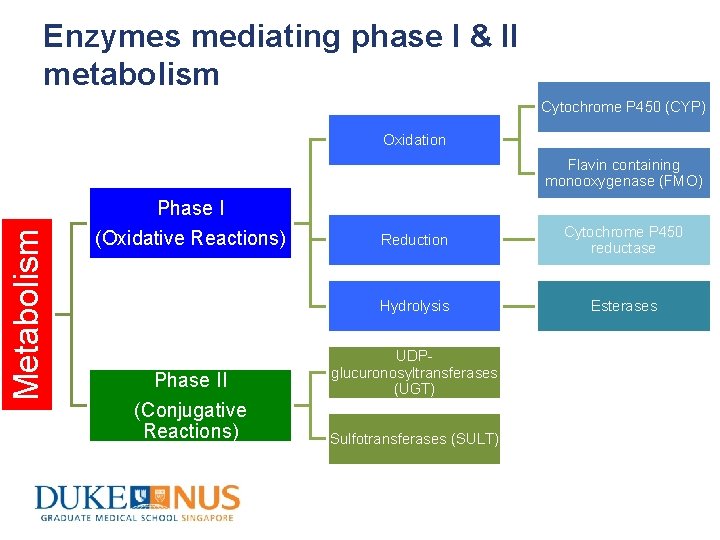
Enzymes mediating phase I & II metabolism Cytochrome P 450 (CYP) Oxidation Metabolism Flavin containing monooxygenase (FMO) Phase I (Oxidative Reactions) Phase II (Conjugative Reactions) Reduction Cytochrome P 450 reductase Hydrolysis Esterases UDPglucuronosyltransferases (UGT) Sulfotransferases (SULT)
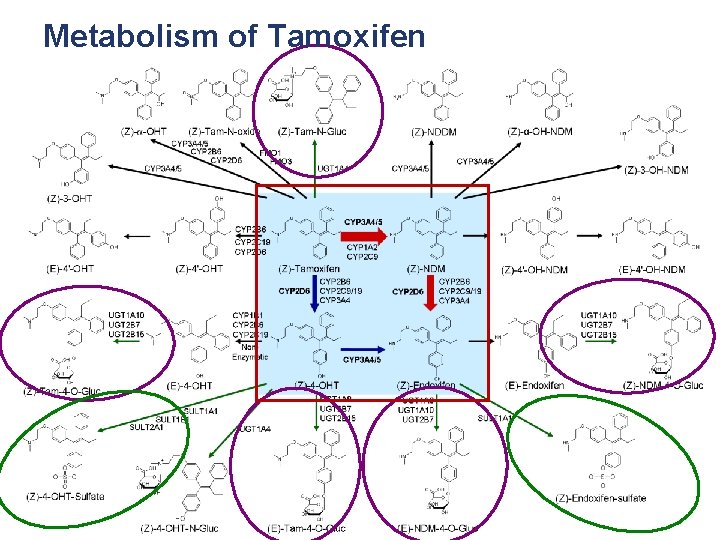
Metabolism of Tamoxifen

Factors influencing metabolism • Liver function • Age • Gender • Smoking status & alcohol consumption • Genetics • Extent of protein binding • Concurrent drugs (Enzyme inhibition & induction)
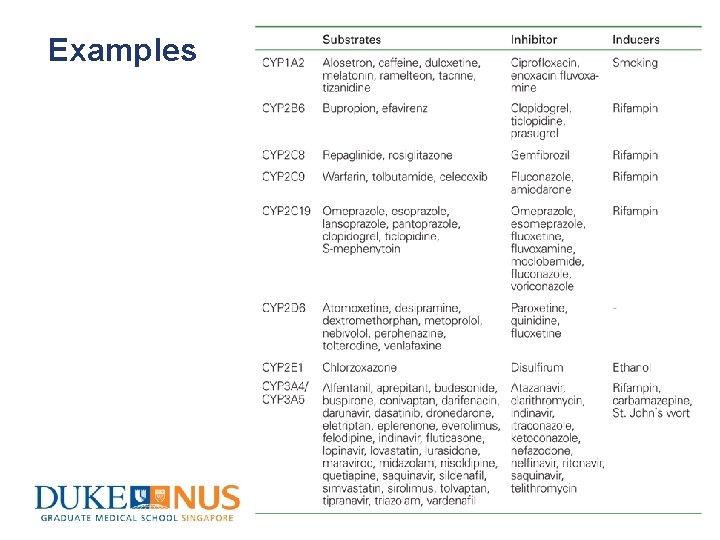
Examples
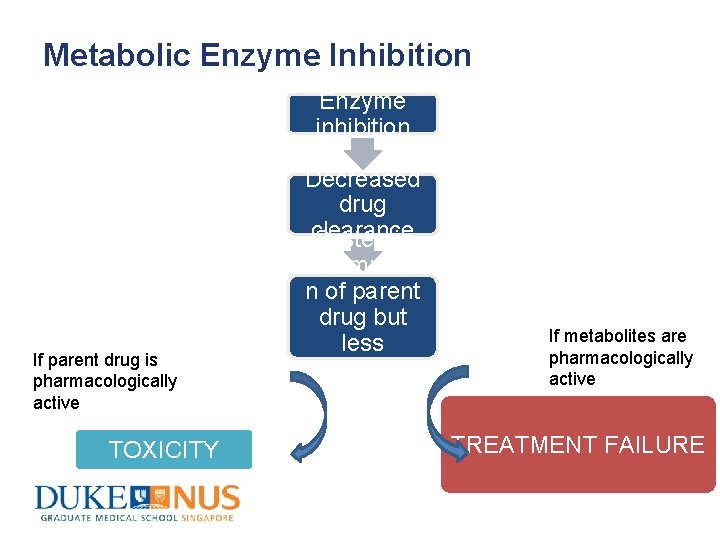
Metabolic Enzyme Inhibition Enzyme inhibition If parent drug is pharmacologically active TOXICITY Decreased drug clearance Systemic accumulatio n of parent drug but less metabolites are formed If metabolites are pharmacologically active TREATMENT FAILURE
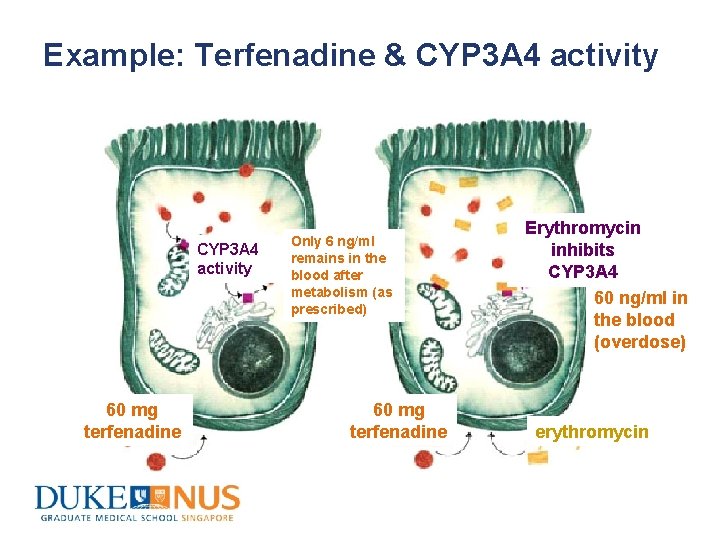
Example: Terfenadine & CYP 3 A 4 activity 60 mg terfenadine Only 6 ng/ml remains in the blood after metabolism (as prescribed) 60 mg terfenadine Erythromycin inhibits CYP 3 A 4 60 ng/ml in the blood (overdose) erythromycin
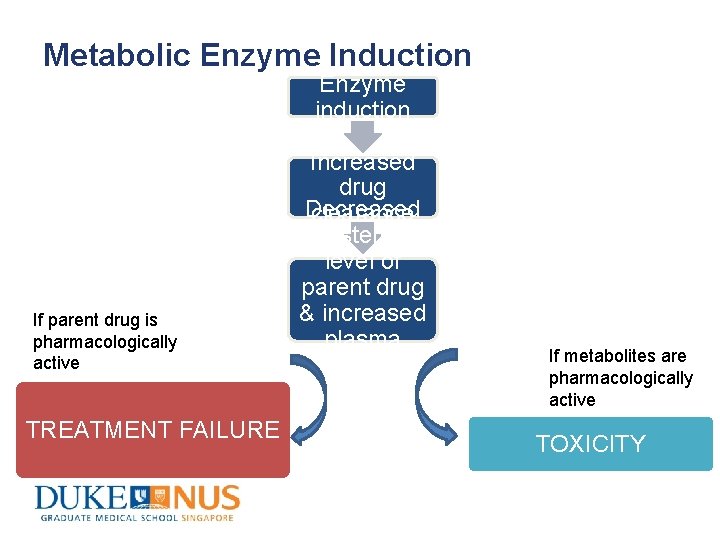
Metabolic Enzyme Induction Enzyme induction If parent drug is pharmacologically active TREATMENT FAILURE Increased drug Decreased clearance systemic level of parent drug & increased plasma level of metabolites If metabolites are pharmacologically active TOXICITY
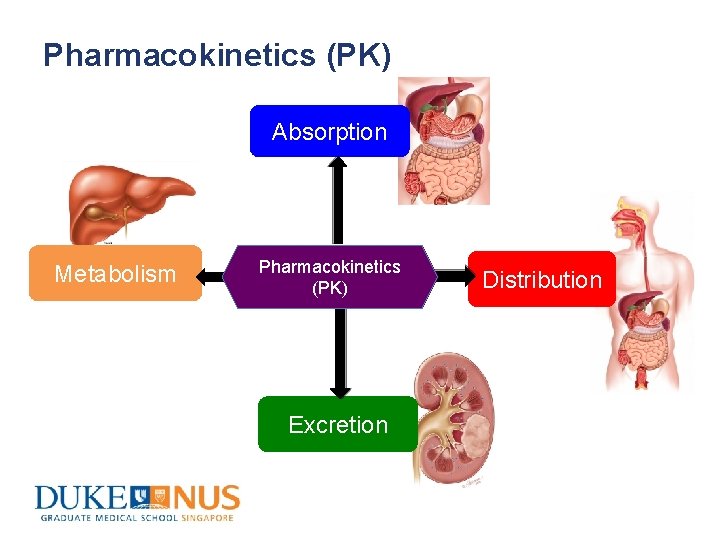
Pharmacokinetics (PK) Absorption Metabolism Pharmacokinetics (PK) Excretion Distribution

Excretion • Irreversible loss of chemically unchanged compound (i. e. parent drug) – Usually hydrophilic or polar compounds • Organs involved: Kidney, lungs, skin, liver
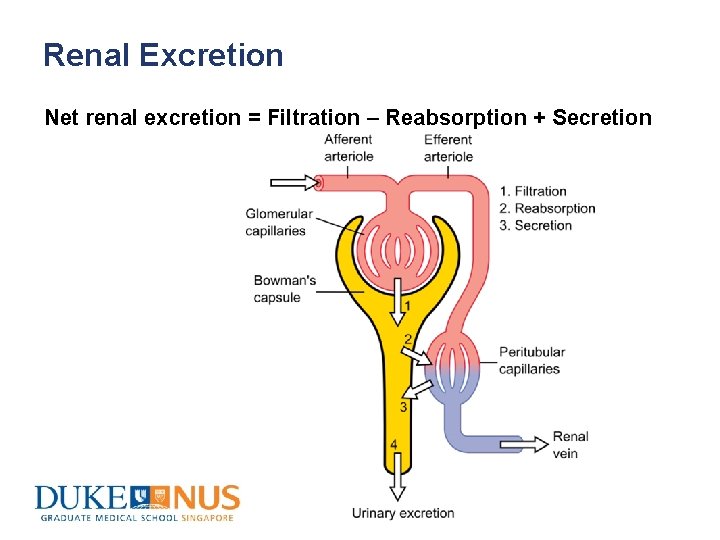
Renal Excretion Net renal excretion = Filtration – Reabsorption + Secretion

Renal Excretion • Glomerular filtration – 20 to 25% of cardiac output, or 1. 1 L of blood per minute, goes to kidneys – Of this volume, 10 % is filtered at the glomerulus by the hydraulic pressure exerted by the arterial blood – Only unbound drug in plasma (concentration Cu) is filtered • Secretion – Active transport of drug from blood to lumen of nephron – Occurs predominantly in proximal tubule – Substances transported by the same system can compete with each other, which may affect their renal clearance

Renal Excretion • Tubular reabsorption – Usually an active process for many vital endogenous compounds e. g. vitamins, electrolytes, etc – Occurs by a passive process for most exogenous compounds including xenobiotics – Degree of reabsorption depends on drug properties, urine flow and urine p. H Renal clearance: volume of plasma passing through the kidney that is cleared of drug per unit time

Factors influencing renal excretion • Renal function • p. H of urine • Concurrent drugs • Genetics • Extent of protein binding

Summary • Pharmacokinetics describes how the body handles the drug. • It is quantitation of the time course of four process: absorption, distribution, metabolism and excretion (ADME) for a drug and its metabolites in the body. • Absorption refers to the transfer of drug from site of administration to site of measurement. – Various sites of administration may be classified as either intravascular or extravascular. – The plasma concentrations achieved can be controlled by the rate of drug administration (i. e. dose of drug and frequency of administration). • Distribution is the process of reversible transfer of a drug to and from the site of measurement, usually plasma, and the peripheral tissues. – It is mathematically described as volume of distribution (Vd) which refers to the volume of bodily fluid into which a drug dose appears to be distributed to account for its plasma concentration.

Summary • Elimination is the irreversible loss of drug from the site of measurement. – It involves two processes, excretion and metabolism. – Metabolism refers to the conversion of one chemical species to another. It generally refers to the conversion of drugs to more polar entities. – Excretion is the irreversible loss of chemically unchanged compound. – PK parameters to describe elimination include clearance (Cl) and elimination half-life (t 1/2). • Clearance (Cl) refers to the volume of fluid presented to the eliminating organ that is completely cleared of drug per unit time. • Elimination half-life (t 1/2) refers to the time taken for the concentration of the drug to fall by one half.

References • Clinical Pharmacokinetics and Pharmacodynamics: Concepts and Applications Malcolm Rowland, Thomas N Tozer • Concepts in Clinical Pharmacokinetics: A Self. Instructional Course Joseph T. Dipiro • Applied Biopharmaceutics & Pharmacokinetics (Fifth Edition) Leon Shargel, Susanna Wu-Pong, Andrew BC Yu

Thank you www. duke-nus. edu. sg
- Slides: 41