Basics of Pharmacodynamics Level Intermediate Version No 1

Basics of Pharmacodynamics Level : Intermediate Version No: 1 Version Date: November 2013 Prepared by: Balram Chowbay, Ph. D Principal Pharmacologist Clinical Pharmacology Lab National Cancer Center Narrated by: Natalia Sutiman

Disclaimer/Liability • The information provided in the VAP is made available in good faith and is derived from sources believed to be reliable and accurate at the time of release. • The materials presented on the VAP may include links to external Internet sites. These external information sources are outside the control of Duke-NUS. The user of the Internet links is responsible for making his or her own decision about the accuracy, reliability and correctness of the information found. • In no event shall Duke-NUS be liable for any indirect, special, incidental, or consequential damages arising out of any use of reliance of any information contained in the VAP. Nor does Duke-NUS assume any responsibility for failure or delay in updating or removing the information contained in the VAP. • Moreover, information provided on the VAP does not constitute medical advice or treatment nor should it be considered as a replacement of the patient/physician relationship or a physician’s professional judgment. Duke. NUS expressly disclaims all liability for treatment, diagnosis, decisions and actions taken or not taken in reliance upon information contained in the VAP. This work is licensed under a Creative Commons Attribution-Non. Commercial-No. Derivs 3. 0 Unported License To view a copy of this license, visit [http: //creativecommons. org/licenses/by-nc-nd/3. 0/]

Financial Disclosures (past 3 years) • No Disclosures
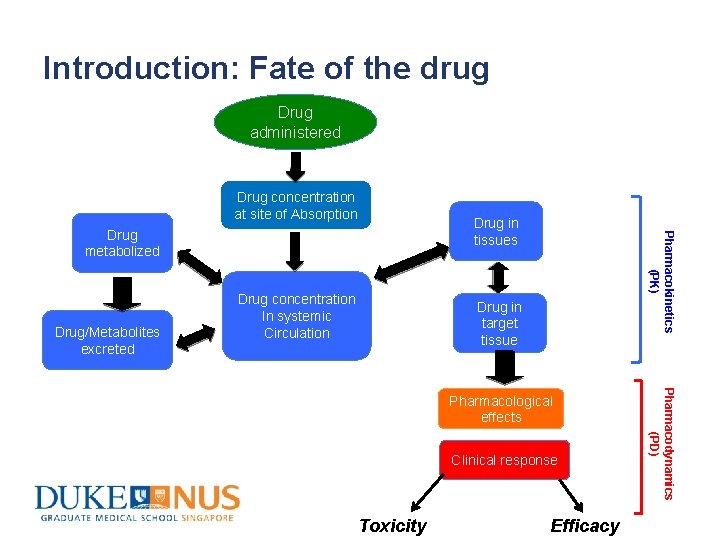
Introduction: Fate of the drug Drug administered Drug concentration at site of Absorption Drug/Metabolites excreted Drug concentration In systemic Circulation Drug in tissues Pharmacokinetics (PK) Drug metabolized Drug in target tissue Clinical response Toxicity Efficacy Pharmacodynamics (PD) Pharmacological effects

Introduction: Concepts of PD • Pharmacodynamics (PD): the study of how a drug acts on a living organism – Type of pharmacological response – Duration of response – Magnitude of response • Drugs will produce response(s) when it is present at high enough concentrations at the target site. • [Drug]target sites ≡ [Drug]plasma
![Drug targets PDTD [Potential Drug Target Database] http: //www. dddc. ac. cn/pdtd/ Drug targets PDTD [Potential Drug Target Database] http: //www. dddc. ac. cn/pdtd/](http://slidetodoc.com/presentation_image_h/97c7434fddccaf87e46fb6f762e750ea/image-6.jpg)
Drug targets PDTD [Potential Drug Target Database] http: //www. dddc. ac. cn/pdtd/
![Concentration-response Relationship Linear Scale Semi-Log Scale Relationship of concentration and effect n Emax [C] Concentration-response Relationship Linear Scale Semi-Log Scale Relationship of concentration and effect n Emax [C]](http://slidetodoc.com/presentation_image_h/97c7434fddccaf87e46fb6f762e750ea/image-7.jpg)
Concentration-response Relationship Linear Scale Semi-Log Scale Relationship of concentration and effect n Emax [C] n E = Kd + [C] E is the effect observed at concentration C Emax is the maximum effect of the drug EC 50 is the concentration required to produce 50% of the maximum effect Interactive Clinical Pharmacology. (http: //www. icp. org. nz/)

Concentration-response Relationship • Terms used to describe drug properties and compare different drugs with the same pharmacological effects: – Potency – Efficacy – Therapeutic Index (TI)
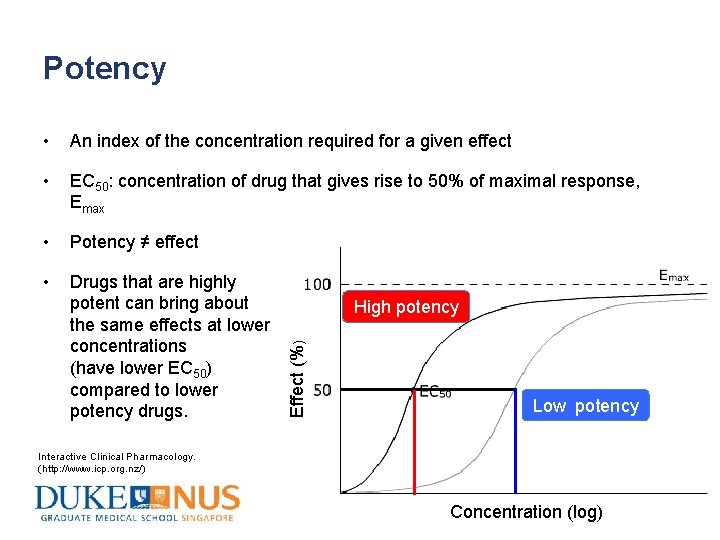
Potency • An index of the concentration required for a given effect • EC 50: concentration of drug that gives rise to 50% of maximal response, Emax • Potency ≠ effect • Drugs that are highly potent can bring about the same effects at lower concentrations (have lower EC 50) compared to lower potency drugs. Effect (%) High potency Low potency Interactive Clinical Pharmacology. (http: //www. icp. org. nz/) Concentration (log)
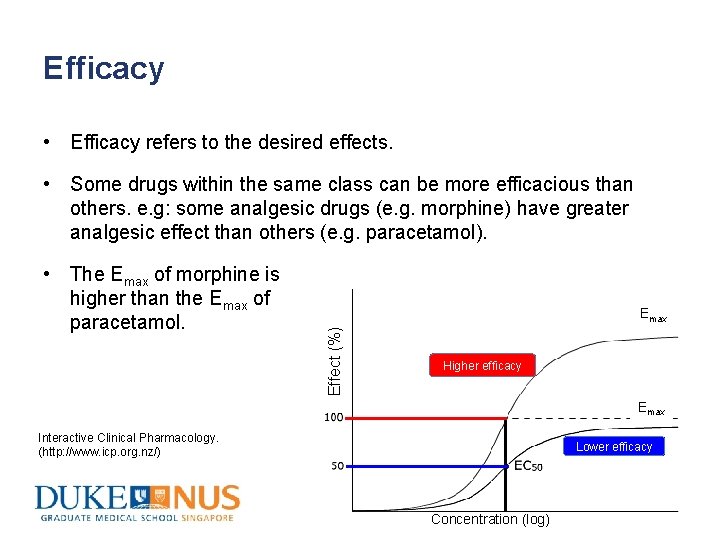
Efficacy • Efficacy refers to the desired effects. • Some drugs within the same class can be more efficacious than others. e. g: some analgesic drugs (e. g. morphine) have greater analgesic effect than others (e. g. paracetamol). Emax Effect (%) • The Emax of morphine is higher than the Emax of paracetamol. Higher efficacy Emax Interactive Clinical Pharmacology. (http: //www. icp. org. nz/) Lower efficacy Concentration (log)
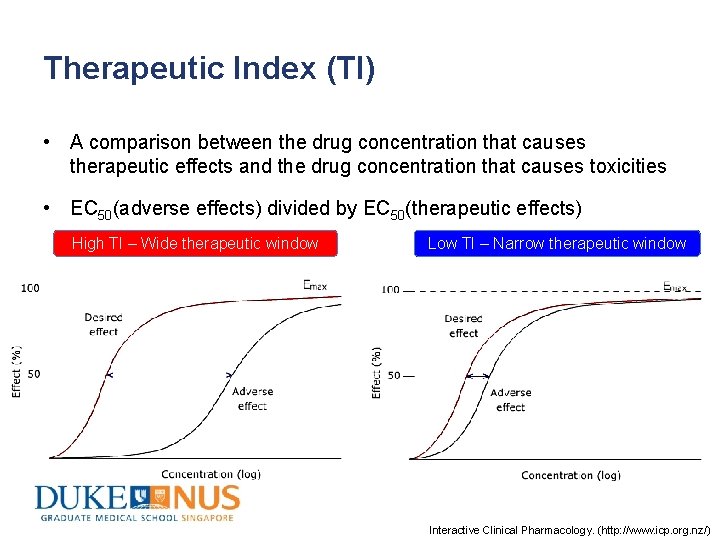
Therapeutic Index (TI) • A comparison between the drug concentration that causes therapeutic effects and the drug concentration that causes toxicities • EC 50(adverse effects) divided by EC 50(therapeutic effects) High TI – Wide therapeutic window Low TI – Narrow therapeutic window Interactive Clinical Pharmacology. (http: //www. icp. org. nz/)
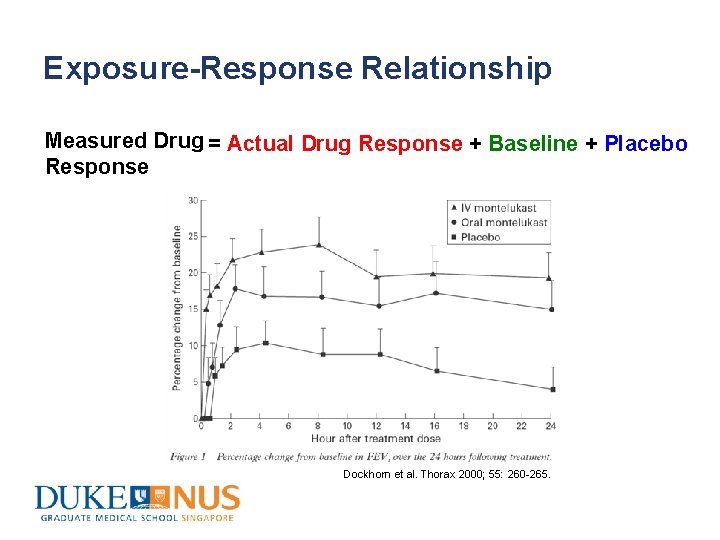
Exposure-Response Relationship Measured Drug = Actual Drug Response + Baseline + Placebo Response Dockhorn et al. Thorax 2000; 55: 260 -265.

Exposure-Response Relationship • Drug response can be classified in various ways: – By clinical impact • Desired: intended, therapeutic effect • Harmful: unwanted, adverse effect – By whether it occurs on a continuous basis • Graded: magnitude of response can be scaled or graded within each individual • Quantal: response do not occur on a continuous basis ie allor-none
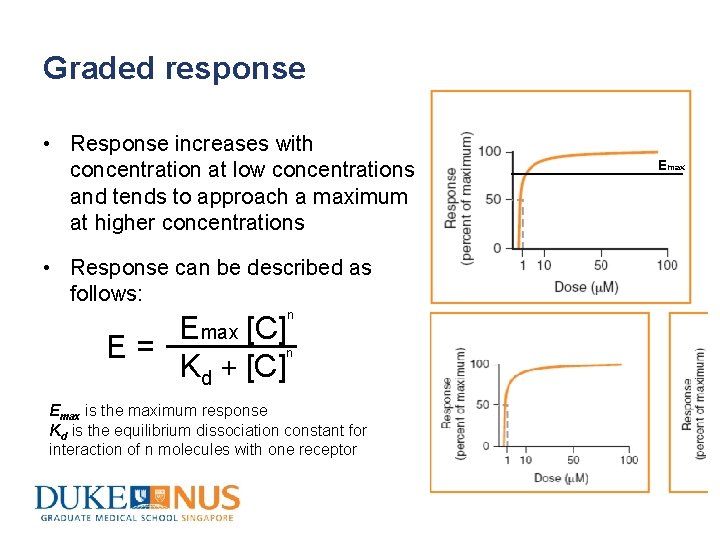
Graded response • Response increases with concentration at low concentrations and tends to approach a maximum at higher concentrations • Response can be described as follows: n Emax [C] n E = Kd + [C] Emax is the maximum response Kd is the equilibrium dissociation constant for interaction of n molecules with one receptor Emax
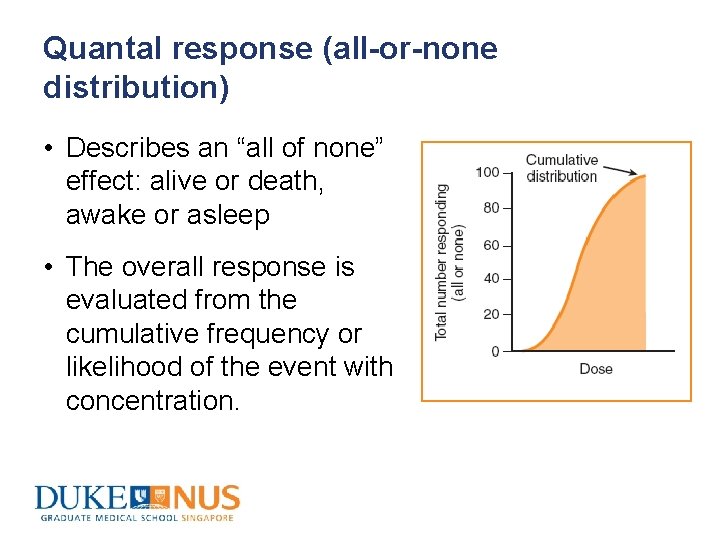
Quantal response (all-or-none distribution) • Describes an “all of none” effect: alive or death, awake or asleep • The overall response is evaluated from the cumulative frequency or likelihood of the event with concentration.
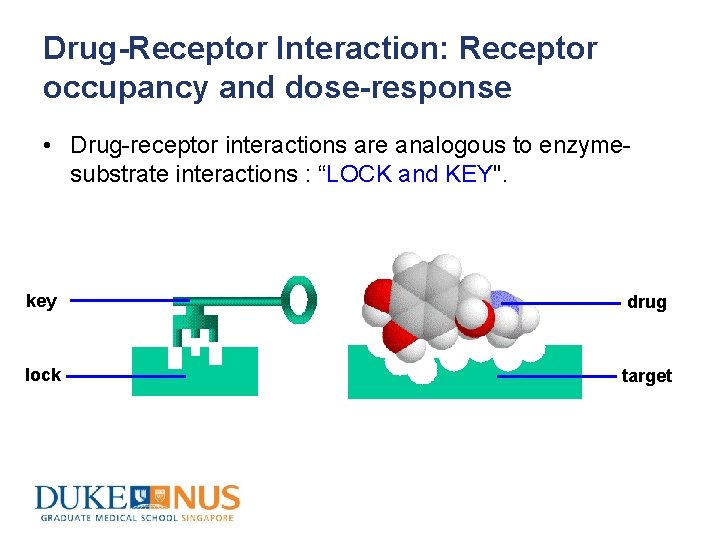
Drug-Receptor Interaction: Receptor occupancy and dose-response • Drug-receptor interactions are analogous to enzymesubstrate interactions : “LOCK and KEY". key drug lock target
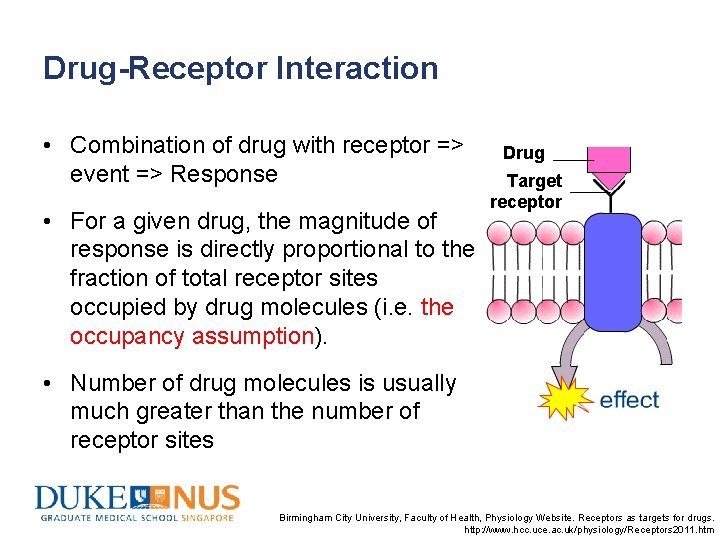
Drug-Receptor Interaction • Combination of drug with receptor => event => Response • For a given drug, the magnitude of response is directly proportional to the fraction of total receptor sites occupied by drug molecules (i. e. the occupancy assumption). Drug Target receptor • Number of drug molecules is usually much greater than the number of receptor sites Birmingham City University, Faculty of Health, Physiology Website. Receptors as targets for drugs. http: //www. hcc. uce. ac. uk/physiology/Receptors 2011. htm
![Drug-Receptor Interaction: Receptor occupancy and dose-response kon Receptor + Drug [RD] = koff Receptor Drug-Receptor Interaction: Receptor occupancy and dose-response kon Receptor + Drug [RD] = koff Receptor](http://slidetodoc.com/presentation_image_h/97c7434fddccaf87e46fb6f762e750ea/image-18.jpg)
Drug-Receptor Interaction: Receptor occupancy and dose-response kon Receptor + Drug [RD] = koff Receptor R + D [R][D] Drug RD koff kon = Kd [RT][D] Kd + [D] R is the concentration of unoccupied receptors D is the concentration of drug RD is the concentration of bound receptors RT is concentration of the total number of receptors kon is the association constant koff is the dissociation constant Kd is the equilibrium dissociation constant http: //www. unpopularmedicine. com/Pharmacology/Basic concepts in pharmacology/Pharmacology Receptor + Drug R + D
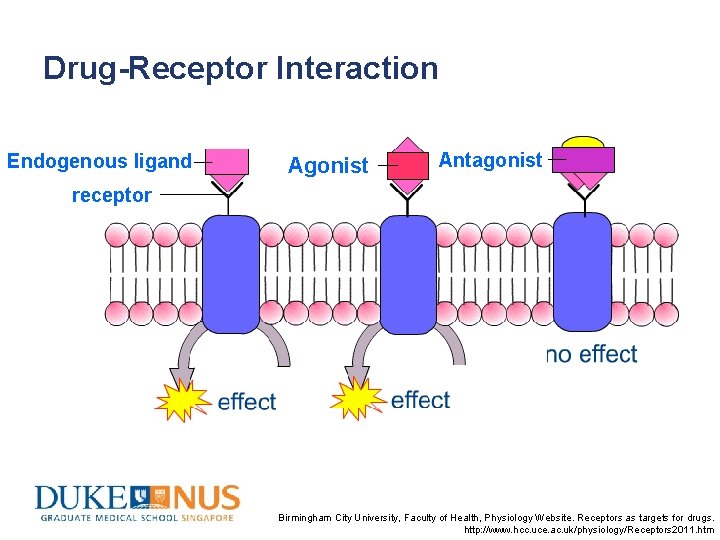
Drug-Receptor Interaction Endogenous ligand Agonist Antagonist receptor Dsf’knfkns’gkas’gk s’b s’ gsggkn’slrgn’sg n’ Birmingham City University, Faculty of Health, Physiology Website. Receptors as targets for drugs. http: //www. hcc. uce. ac. uk/physiology/Receptors 2011. htm
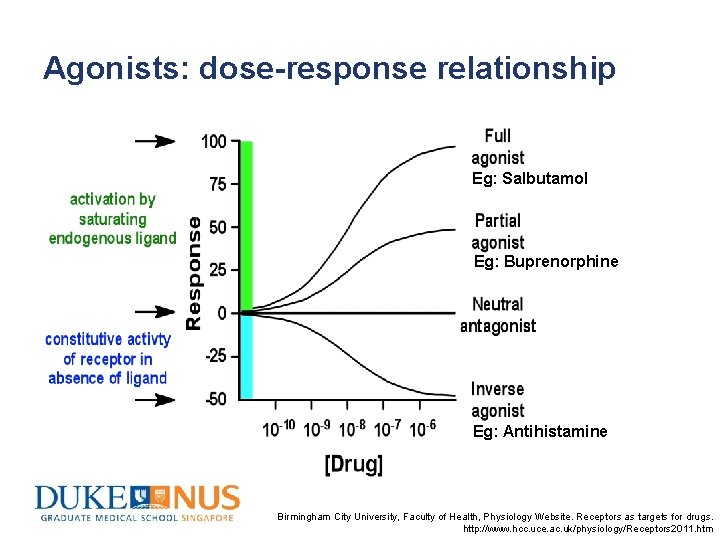
Agonists: dose-response relationship Eg: Salbutamol Eg: Buprenorphine Eg: Antihistamine Birmingham City University, Faculty of Health, Physiology Website. Receptors as targets for drugs. http: //www. hcc. uce. ac. uk/physiology/Receptors 2011. htm
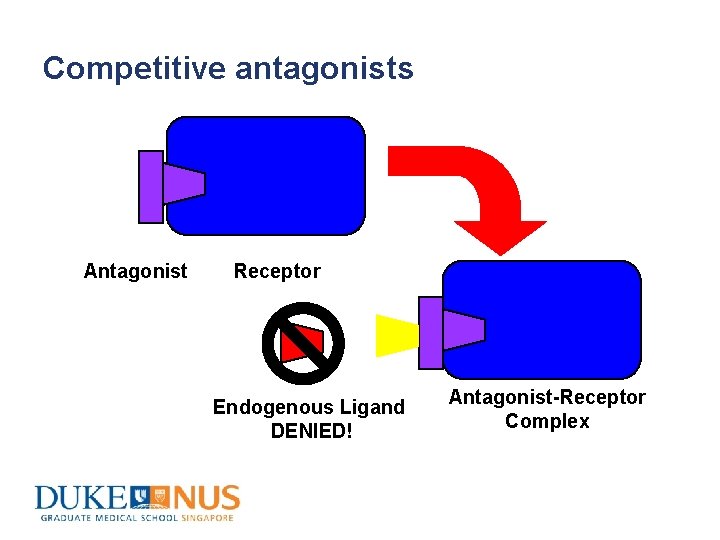
Competitive antagonists Antagonist Receptor Endogenous Ligand DENIED! Antagonist-Receptor Complex
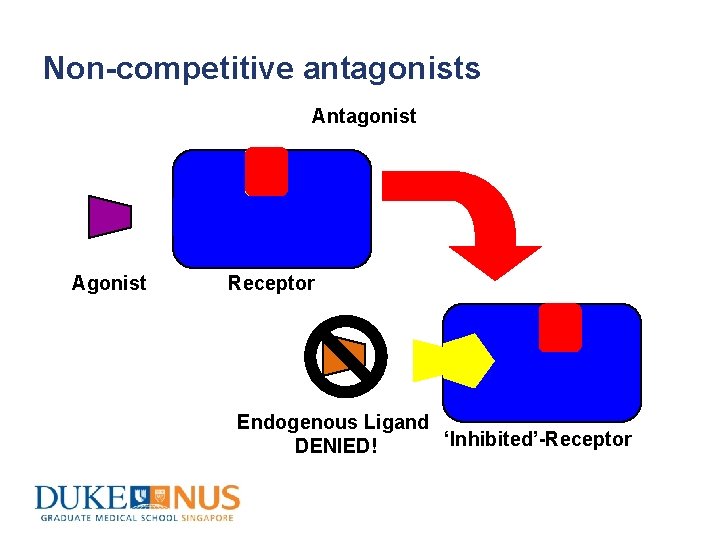
Non-competitive antagonists Antagonist Agonist Receptor Endogenous Ligand ‘Inhibited’-Receptor DENIED!
![Biologic effect Irreversible antagonist [Agonist] Biologic effect Irreversible antagonist [Agonist]](http://slidetodoc.com/presentation_image_h/97c7434fddccaf87e46fb6f762e750ea/image-23.jpg)
Biologic effect Irreversible antagonist [Agonist]

Effects of Agonists & Antagonists Native ligand or full agonist Partial agonist, equal potency, or non competitive antagonist Full agonist, lower potency, or competitive antagonist Agonists and antagonists. [Adapted from Waldman & Terzic, Pharmacology and Therapeutics: Principles to Practice. 2009 Saunders. p 64]

Pharmacodynamic (PD) Models • Relate drug concentrations at the active site to the pharmacological response • Assumption: drug concentration at site of measurement is at equilibrium with that of the active site • Four PD models : – – Sigmoid Emax model Linear model Log-linear model

Sigmoid Emax Model • Based on the hyperbolic relationship of drugreceptor interaction E approaches plateau at high concentrations Emax [C]n E = EC 50 + [C]n Emax EC 50 n is the maximum response is the plasma concentration required to produce 50% of Emax is the steepness or shape factor Effect (%) Emax Rise in magnitude of response (E) vs concentration occurs at concentration much lower than EC 50 Concentration Interactive Clinical Pharmacology. (http: //www. icp. org. nz/)

Sigmoid Emax Model • Steepness or shape factor – Accommodates the steepness of the curve around EC 50. – Larger value indicates a greater change in response with concentration around EC 50 – The part of the curve (on log scale) between 20% and 80% of maximal response appears approx. linear. [Adapted from Rowland & Tozer, Clinical Pharmacokinetics and Pharmacodynamics: Concepts and Applications. 4 th edn Lippincott Williams & Wilkins. p 37]

Emax Model • Submodel of sigmoid Emax model • Based on the interaction of a single drug molecule with a single receptor site Emax [C] E = EC 50 + [C] % Decrease in Heart Rate • When n=1 Emax is the maximum response EC 50 is the plasma concentration required to produce 50% of Emax Unbound Concentration of Propranolol (ng/ml) [Adapted from Waldman & Terzic, Pharmacology and Therapeutics: Principles to Practice. 2009 Saunders. p 205]
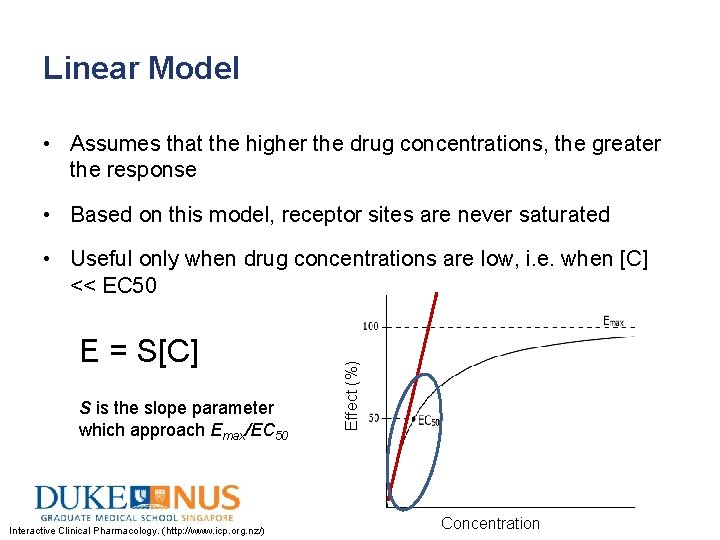
Linear Model • Assumes that the higher the drug concentrations, the greater the response • Based on this model, receptor sites are never saturated E = S[C] S is the slope parameter which approach Emax/EC 50 Interactive Clinical Pharmacology. (http: //www. icp. org. nz/) Effect (%) • Useful only when drug concentrations are low, i. e. when [C] << EC 50 Concentration
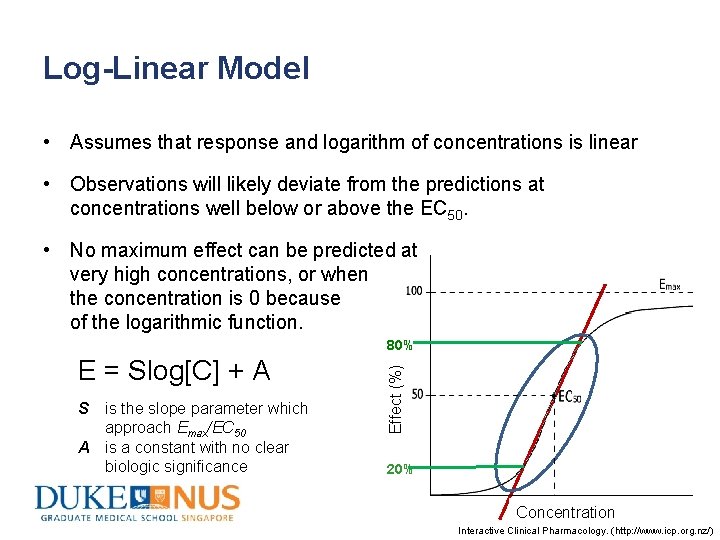
Log-Linear Model • Assumes that response and logarithm of concentrations is linear • Observations will likely deviate from the predictions at concentrations well below or above the EC 50. • No maximum effect can be predicted at very high concentrations, or when the concentration is 0 because of the logarithmic function. E = Slog[C] + A S is the slope parameter which approach Emax/EC 50 A is a constant with no clear biologic significance Effect (%) 80% 20% Concentration Interactive Clinical Pharmacology. (http: //www. icp. org. nz/)

Dose-response relationships • The amount of drug required to produce a response at the site of action is usually a minute fraction of the total amount of drug in the body • The measured response does not directly reflect the actual effect of the drug – Measured response relates to the concentration of endogenous substance in the body – Direct effect of the drug is to ↑ or ↓ rate of formation or elimination of endogenous substance
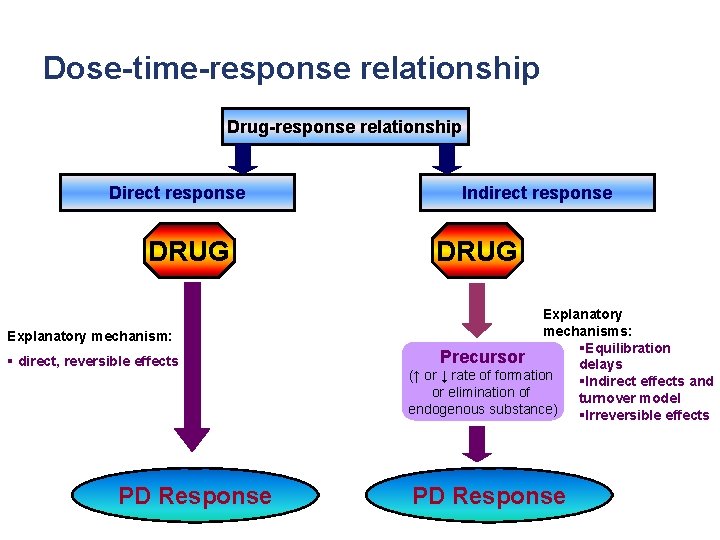
Dose-time-response relationship Drug-response relationship Direct response DRUG Explanatory mechanism: § direct, reversible effects PD Response Indirect response DRUG Explanatory mechanisms: §Equilibration Precursor delays (↑ or ↓ rate of formation §Indirect effects and or elimination of turnover model endogenous substance) §Irreversible effects PD Response
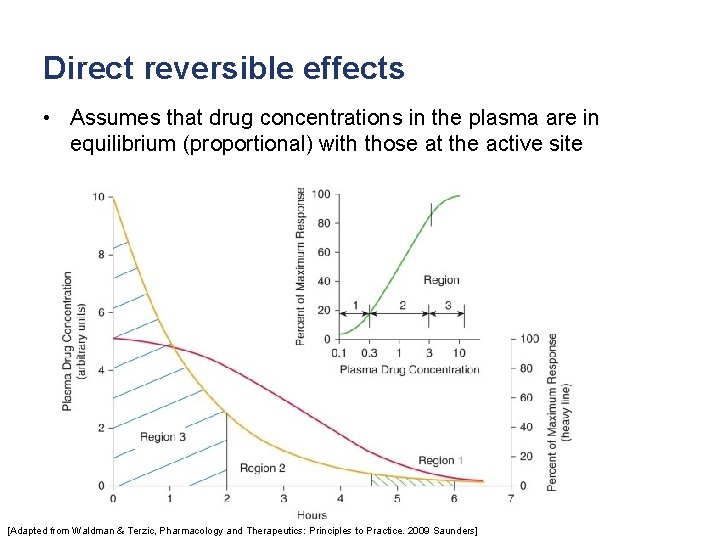
Direct reversible effects • Assumes that drug concentrations in the plasma are in equilibrium (proportional) with those at the active site [Adapted from Waldman & Terzic, Pharmacology and Therapeutics: Principles to Practice. 2009 Saunders]
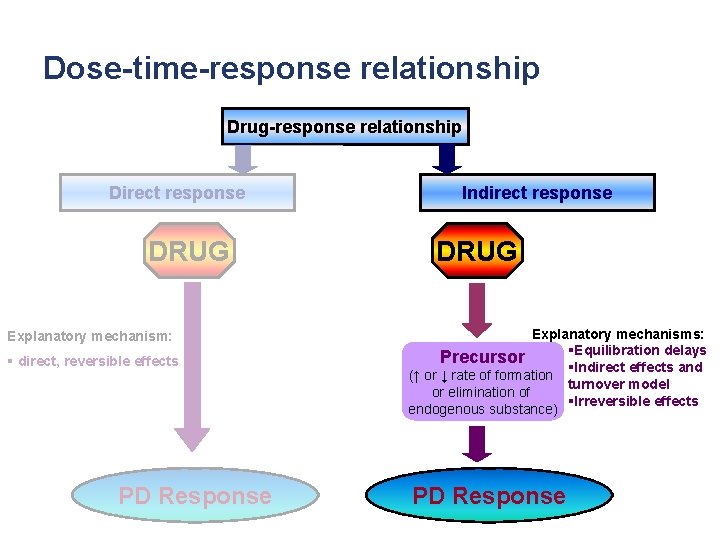
Dose-time-response relationship Drug-response relationship Direct response DRUG Explanatory mechanism: § direct, reversible effects PD Response Indirect response DRUG Explanatory mechanisms: §Equilibration delays Precursor §Indirect effects and (↑ or ↓ rate of formation turnover model or elimination of §Irreversible effects endogenous substance) PD Response
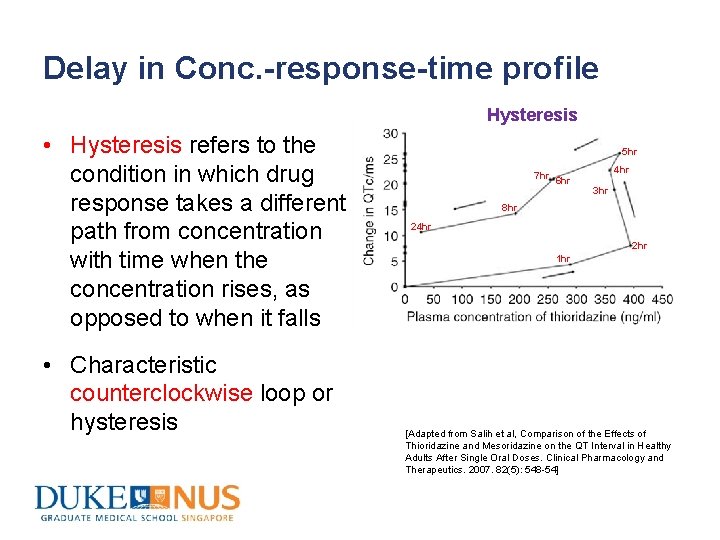
Delay in Conc. -response-time profile Hysteresis • Hysteresis refers to the condition in which drug response takes a different path from concentration with time when the concentration rises, as opposed to when it falls • Characteristic counterclockwise loop or hysteresis 5 hr 7 hr 6 hr 4 hr 3 hr 8 hr 24 hr 2 hr 1 hr [Adapted from Salih et al, Comparison of the Effects of Thioridazine and Mesoridazine on the QT Interval in Healthy Adults After Single Oral Doses. Clinical Pharmacology and Therapeutics. 2007. 82(5): 548 -54]

Delay in Conc. -response-time profile Proteresis • A clockwise hysteresis loop occurs when rapid tolerance (tachyphylaxis) develops. • Examples: cocaine or indirectly acting sympathomimetic drugs such as pseudoephedrine Alprazolam concentration (ng/ml) [Adapted from Barbanoj et al, Different acute tolerance development to EEG, psychomotor performance and subjective assessment effects after two intermittent oral doses of alprazolam in healthy volunteers. Neuropsychobiology. 2007; 55(3 -4): 203 -12. ]
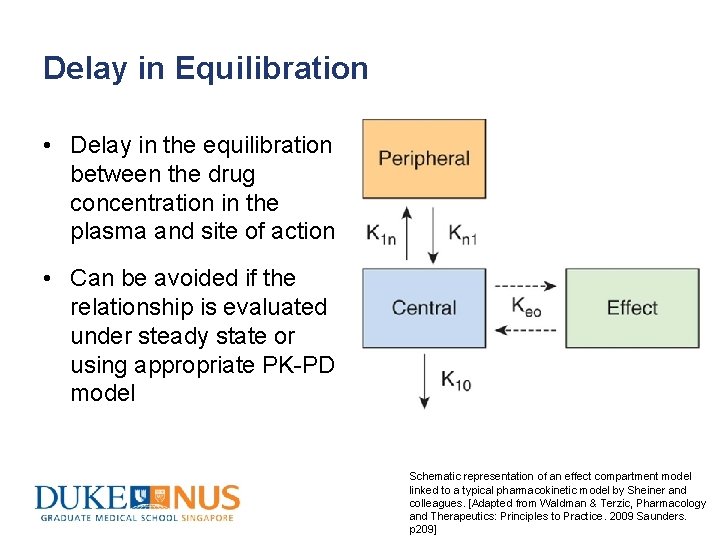
Delay in Equilibration • Delay in the equilibration between the drug concentration in the plasma and site of action • Can be avoided if the relationship is evaluated under steady state or using appropriate PK-PD model Schematic representation of an effect compartment model linked to a typical pharmacokinetic model by Sheiner and colleagues. [Adapted from Waldman & Terzic, Pharmacology and Therapeutics: Principles to Practice. 2009 Saunders. p 209]
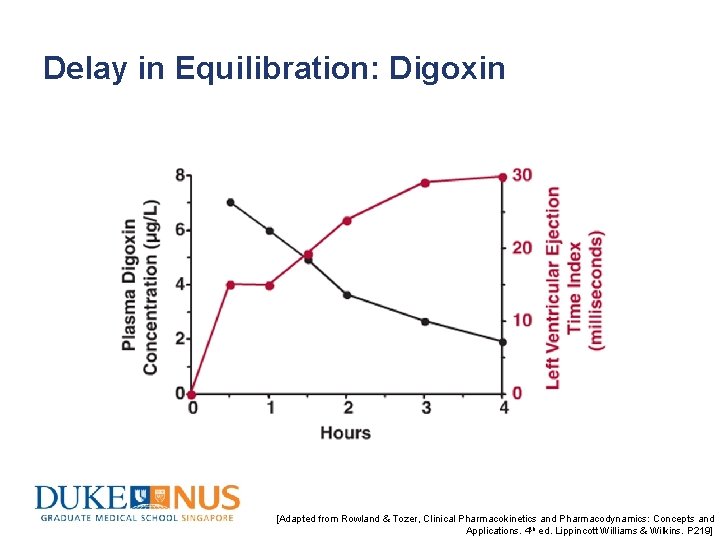
Delay in Equilibration: Digoxin [Adapted from Rowland & Tozer, Clinical Pharmacokinetics and Pharmacodynamics: Concepts and Applications. 4 th ed. Lippincott Williams & Wilkins. P 219]
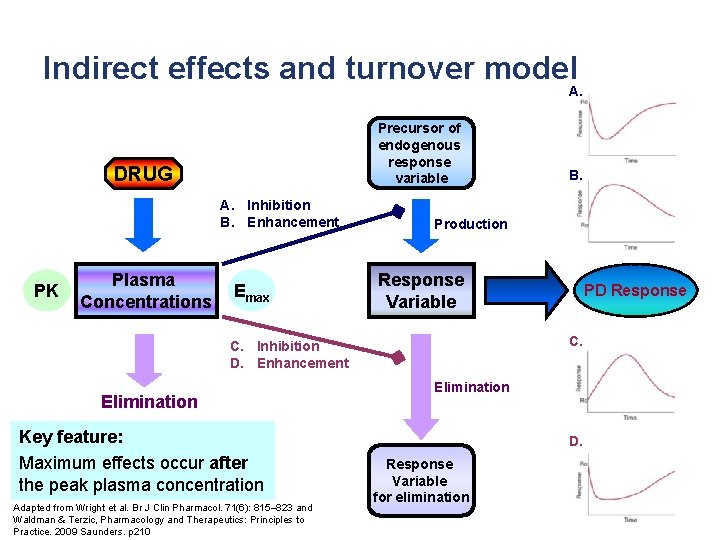
Indirect effects and turnover model A. Precursor of endogenous response variable DRUG A. Inhibition B. Enhancement PK Plasma Concentrations Emax Production Response Variable Key feature: Maximum effects occur after the peak plasma concentration Adapted from Wright et al. Br J Clin Pharmacol. 71(6): 815– 823 and Waldman & Terzic, Pharmacology and Therapeutics: Principles to Practice. 2009 Saunders. p 210 PD Response C. C. Inhibition D. Enhancement Elimination B. Elimination D. Response Variable for elimination
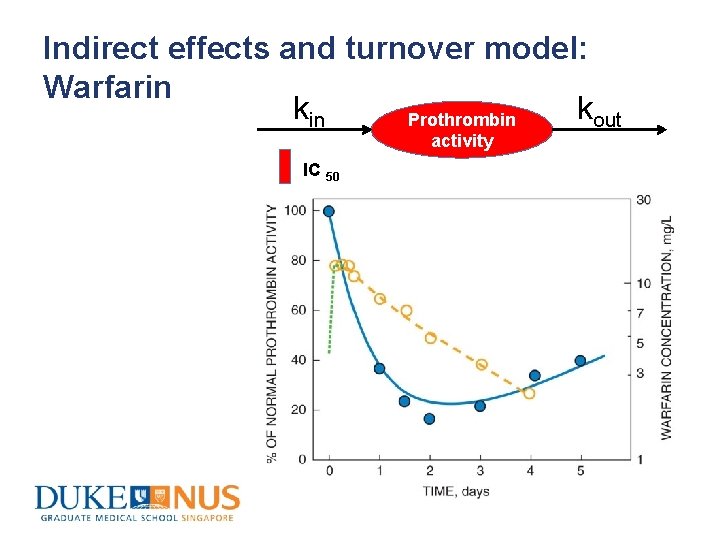
Indirect effects and turnover model: Warfarin kout Prothrombin activity IC 50
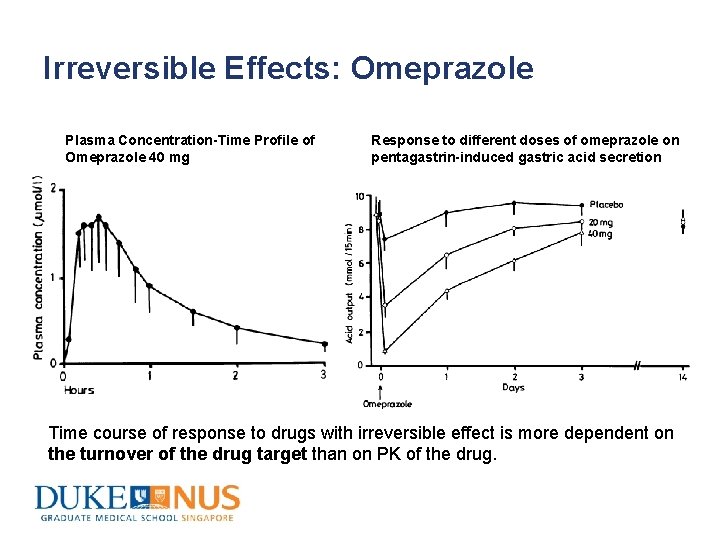
Irreversible Effects: Omeprazole Plasma Concentration-Time Profile of Omeprazole 40 mg Response to different doses of omeprazole on pentagastrin-induced gastric acid secretion Time course of response to drugs with irreversible effect is more dependent on the turnover of the drug target than on PK of the drug.

Summary • Pharmacodynamics is the study of how drug acts on the body of a living organism. • It includes pharmacologic response as well as the duration and magnitude of response observed. • Drugs produce effect when there is adequate interaction with proteins at the target sites. • Drug-receptor interactions are analogous to the way a key fits into a lock. • Based on its effects, drugs can be classified as agonists (mimic the effect of endogenous ligands) or antagonists (conteract the effect of endogenous ligands). • Drug response can be classified in two ways: – Graded or quantal (all-or-none) – Desired (therapeutic effect) or harmful (adverse effect) Measured Response = Drug Response + Placebo Response + Baseline

Summary • Pharmacodynamic (PD) models relate drug concentrations at the active site and pharmacological response – Sigmoid Emax Model – Linear Model – Log-Linear Model • Drug response is often delayed and measured response does not directly reflect the actual drug effects • Drug response can be explained by the following mechanisms: – Direct reversible effects – Equilibration delays – Indirect effects and turnover model – Irreversible effects

Thank you www. duke-nus. edu. sg
- Slides: 44