BASICS OF HEAT TRANSFER Difference between heat and

BASICS OF HEAT TRANSFER Difference between heat and temperature Temperature: is a measure of the amount of energy possessed by the molecules of a substance. It is a measure of degree of hotness or coldness and helps in identifying the direction of heat transfer. The usual symbol for temperature is T. Heat: it is the form of energy in transit. Heat always flows from high temp to low temp. The symbol for heat is Q and its unit is Joule or Kilo Joule.

Difference between thermodynamics and heat transfer From Thermodynamics we can calculate • how much heat is transferred (d. Q) • how much work is done (d. W) • final state of the system With the knowledge of Heat transfer , we can calculate • Mode of d. Q is transferred • at what rate d. Q is transferred • temperature distribution inside the body

Modes of Heat Transfer 1. Conduction 2. Convection 3. Radiation
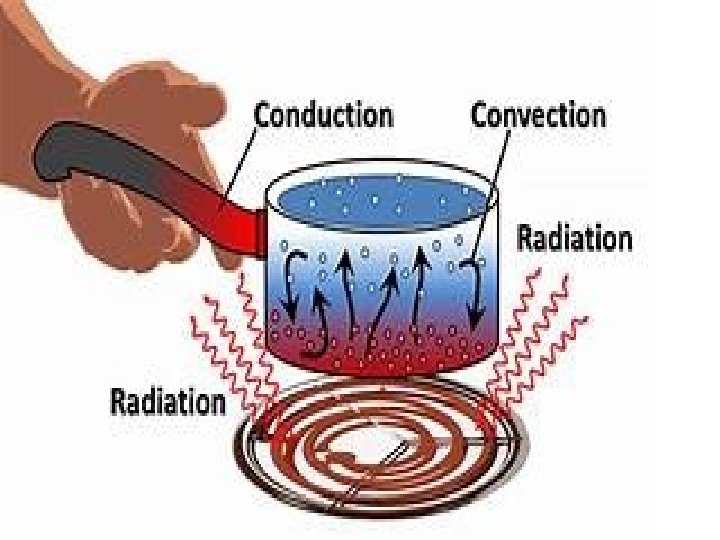

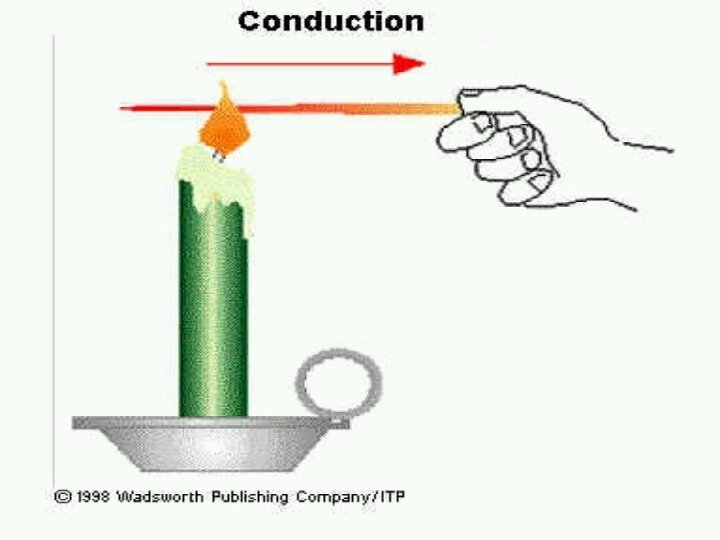
Conduction: An energy transfer from one part of a system to another part of the same system OR from one system to another which are in physical contact with each other. Conduction rate equation is described by the Fourier Law: q = - K A d. T/dx where: q = heat flow , (W) K = thermal conductivity (W/m-K) A = Cross sectional area in direction of heat flow. (m 2 ) d. T/dx = Temperature gradient in x-direction (K/m) Thermal Conductivity: It is the ability of the material to conduct the heat. Higher the value of thermal conductivity , higher the rate of heat transfer.
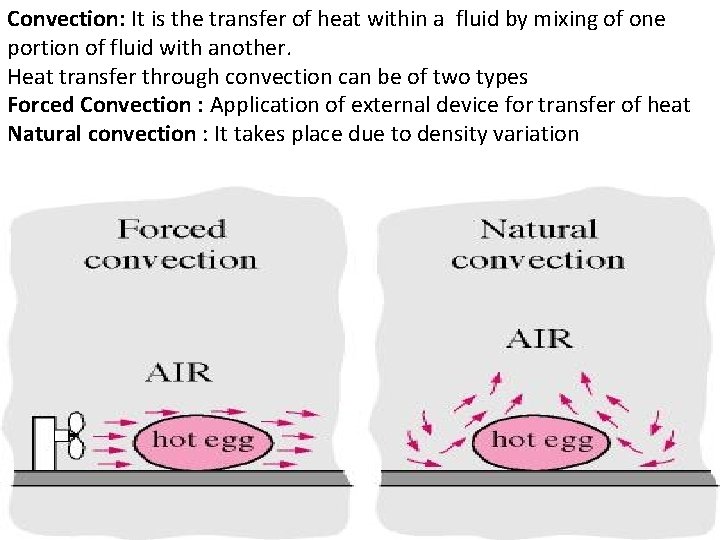
Convection: It is the transfer of heat within a fluid by mixing of one portion of fluid with another. Heat transfer through convection can be of two types Forced Convection : Application of external device for transfer of heat Natural convection : It takes place due to density variation

Heat Transfer through Convection can be given as Q= h. A(Ts-Ta) where: Q = heat flow , (W) h = Convective heat transfer coefficient (W/m 2 -K) A = surface area (m 2 ) Ts= Surface Temperature Ta=Ambient Temperature
- Slides: 8