BASICS OF BONDING Types of Bonding Bonds how

BASICS OF BONDING

Types of Bonding ■ Bonds: how atoms are held together ■ 3 main bonding types 1. Ionic (strong) 2. Covalent (weak) 3. Metallic (strong)

Ionic ■ Between metals and nonmetals – Both form ions ■ Ions are attracted to each other – By nuclear charge – Metals form cations ■ lose electrons – Nonmetals form anions ■ gain electrons
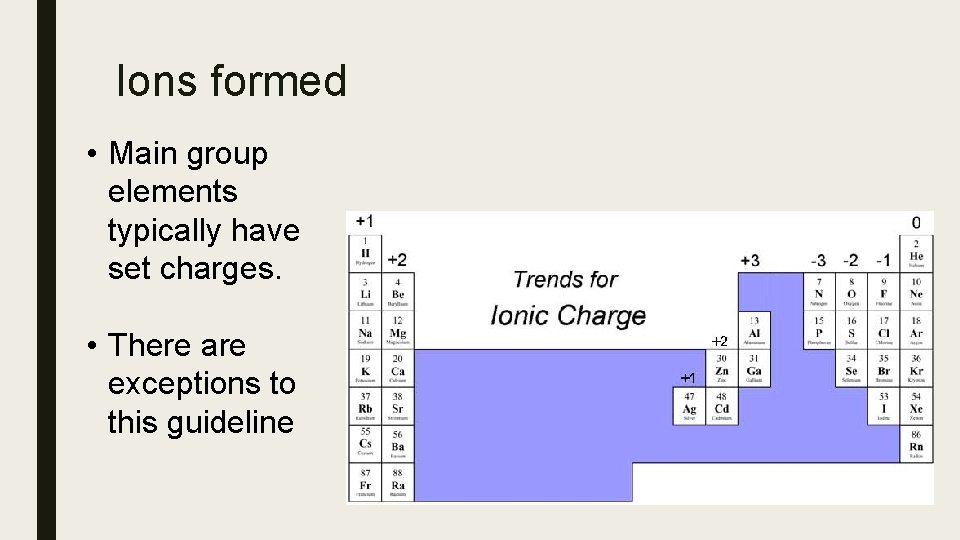
Ions formed • Main group elements typically have set charges. • There are exceptions to this guideline

Transition Metal ■

Polyatomic Ions ■ Nonmetals, covalently, bonded together that have an overall charge – Poly: many – Atomic: atoms – Ions: charged ■ they form both cations and anions – Must memorized grouping and charge ■ Have a different naming system ■ DON’T FORGET THE QUIZLET

Why do compounds form ? ■ STABILITY!!!! – All ions either lose or gain electrons ■ Octet Rule: To have a FULL (2 or 8) outer shell – Nonmetals gain electrons – Metals lose electrons ■ Oppositely charged ions attract to form bonds

Creating Ionic Compounds 1. Find the metal on the periodic table 2. Determine type of atom 1. Metal, nonmetal, transition metal 3. Determine charge of atom 1. How many electrons does it gain/lose 1. Metals lose (cation) 2. Nonmetals gain(anion) 4. Equalize the charge 1. By using subscripts which tell number

Let's form some “normal” ionic compounds! Calcium Oxide Ca 2+ O 2(2+)+(2 -) = 0 Ca. O Sodium Fluoride Na 1+ F 1(1+)+(1 -) = 0 Na. F Na 1+ O 2 - Calcium Fluoride Ca 2+ F 1(2+)+(1 -) = 1+ (1+) + (2 -) = 12 (1+) + (2 -) = 0 ___ 2 (1 -) = 0 (2+)+ ___ Na 2 O Ca. F 2 Sodium Oxide

How to determine a Transition Metal Charge From the formula ■ Determine the NONMETAL’S or Polyatomic ions’ charge ■ Equalize the compound to neutral – Make charges equal and opposite – Use subscripts From the Name • The roman numeral is the charge of the transition metal

Lets Determine Charge! Fe. O Fe: ? O: 22 _____ + 2 - = 0 Fe 2 S 3 Fe: ? S: 2 S: (2 -) (3) = 63 (2) = 6+ Fe: (__) Manganese (II) Oxide 2 Mn: O: 2 -

Naming Ionic Compounds 1. The cation keeps its full name 2. Anion take the root of the word 1. Change the ending to “ide” 1. Exception: polyatomic ions always keep their full name 3. Put them together

Lets name some compounds! 1. Cu. Cl 2 1. copper (II) chloride 2. Li(C 2 H 3 O 2) 2. lithium acetate 3. V 2 Se 3 3. vanadium (III) selenide 4. Mn 3 N 4 5. Be. O 6. Na 2(SO 4) 4. manganese (IV) nitride 5. beryllium oxide 6. sodium sulfate

Properties of Ionic Bonds ■ Strong ■ Form Crystalline solids – Network solids ■ Can conduct electricity when – molten – Aqueous ■ High melting points ■ Hard ■ Brittle ■ Electronegativity Difference > 1. 9

Covalent ■ When atoms share electrons – Between non-metals ■ Not all sharing is equal

Why do the form ? ■ Valence shell/ orbitals over lap to share electrons – Covalent compounds can share ■ One pair ■ Two pairs ■ Three pairs ■ Stability – Share electrons so valence shell is full ■ Duet (Hydrogen and Helium only) or Octet – Lowest possible energy

Steps to making covalent compounds ■ NO CHARGES ARE INVOVOLED – Amount is indicated by number prefix in the name

Naming Covalent Compounds ■ First element keeps it full name – If there is more then one of the element ■ Gets a number prefix ■ Second element keeps the root of the name – Ends in “ide” ■ Always gets a number prefix

Lets make some Covalent Compounds 1. carbon tetrafluoride 2. silicon dioxide 3. dinitrogen trisulfide 4. phosphorus mononitride 5. hydrogen gas 6. carbon disulfide 1. 2. 3. 4. 5. 6. CF 4 Si. O 2 N 2 S 3 PN H 2 CS 2

Lets Name Covalent Compounds 1. CO 2 3. N 2 O 3 4. N 2 5. NP 6. SCl 2 1. Carbon Monoxide 2. Carbon Dioxide 3. Dinitrogen Trioxide 4. Nitrogen gas 5. Nitrogen Mono. Phosphide 6. Sulfide Dichloride

Properties of Covalent Bonds ■ ■ ■ ■ Weak Liquids Gases Nonconductive Low melting points Flows easily Electronegativity difference of 0. 3 < EN < 1. 9

Metallic Bonding ■ Force that keeps metal atoms together – Delocalization of electrons ■ Sea of electrons flowing between metal cations – Leads to metallic properties ■ Ductility – Pulled into wires ■ Malleability – Hammered into sheets ■ Conductivity – Can move electricity

Properties of Metallic Bonds ■ ■ ■ Strong Solids Extremely conductive High melting points Hard – Malleable – Ductile ■ Electronegativity Difference < 0. 3
- Slides: 23