BASIC VACUUM PRACTICE Why is a Vacuum Needed

BASIC VACUUM PRACTICE

Why is a Vacuum Needed? To move a particle in a (straight) line over a large distance (Page 5 manual)

Why is a Vacuum Needed? Atmosphere Contamination (usually water) (High)Vacuum Clean surface To provide a clean surface

HOW DO WE CREATE A VACUUM?
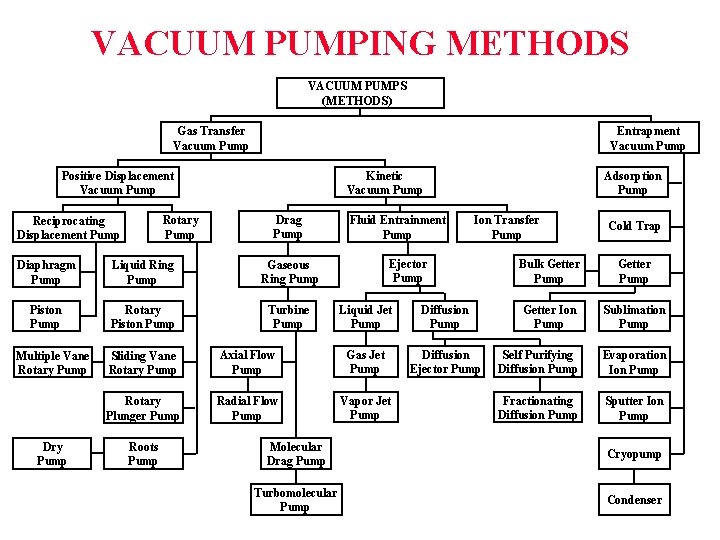
VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Gas Transfer Vacuum Pump Entrapment Vacuum Pump Kinetic Vacuum Pump Positive Displacement Vacuum Pump Rotary Pump Reciprocating Displacement Pump Drag Pump Diaphragm Pump Liquid Ring Pump Gaseous Ring Pump Piston Pump Rotary Piston Pump Turbine Pump Multiple Vane Rotary Pump Dry Pump Fluid Entrainment Pump Ion Transfer Pump Ejector Pump Liquid Jet Pump Diffusion Ejector Pump Sliding Vane Rotary Pump Axial Flow Pump Gas Jet Pump Rotary Plunger Pump Radial Flow Pump Vapor Jet Pump Roots Pump Adsorption Pump Cold Trap Bulk Getter Pump Getter Ion Pump Sublimation Pump Self Purifying Diffusion Pump Evaporation Ion Pump Fractionating Diffusion Pump Sputter Ion Pump Molecular Drag Pump Cryopump Turbomolecular Pump Condenser
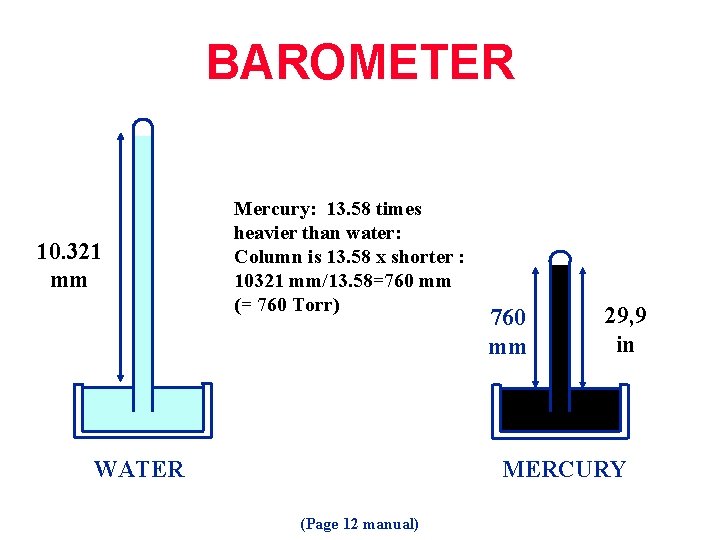
BAROMETER 10. 321 mm Mercury: 13. 58 times heavier than water: Column is 13. 58 x shorter : 10321 mm/13. 58=760 mm (= 760 Torr) WATER 760 mm 29, 9 in MERCURY (Page 12 manual)

PRESSURE OF 1 STANDARD ATMOSPHERE: 760 TORR, 1013 mbar AT SEA LEVEL, 0 O C AND 45 O LATITUDE
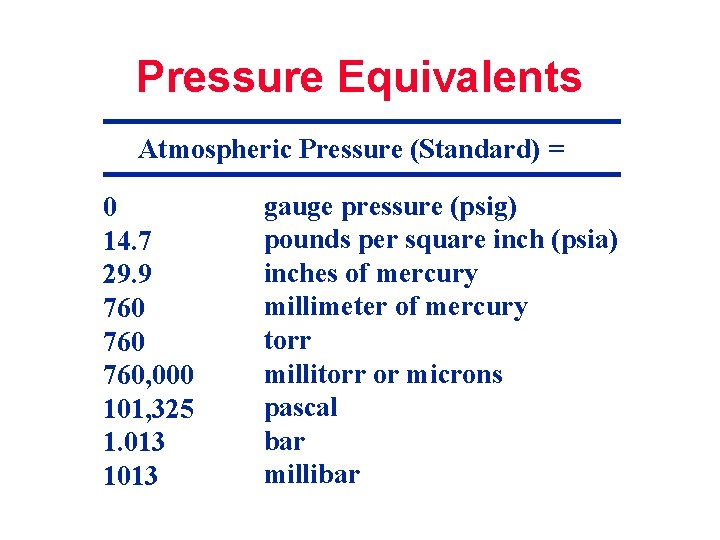
Pressure Equivalents Atmospheric Pressure (Standard) = 0 14. 7 29. 9 760 760, 000 101, 325 1. 013 1013 gauge pressure (psig) pounds per square inch (psia) inches of mercury millimeter of mercury torr millitorr or microns pascal bar millibar
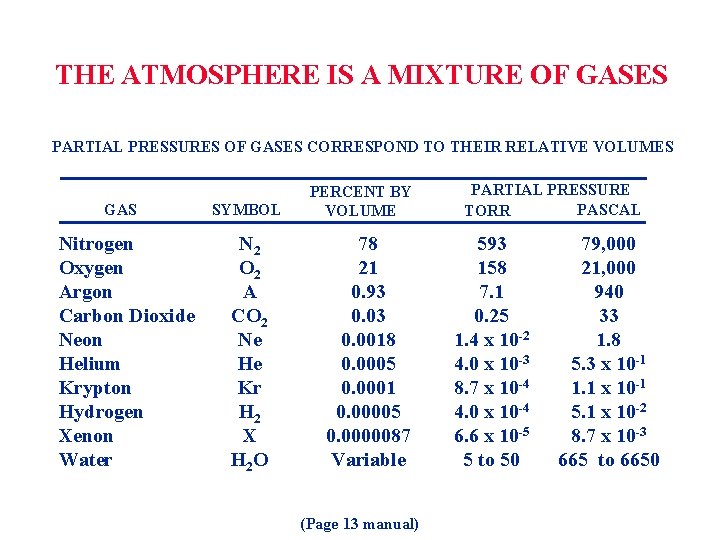
THE ATMOSPHERE IS A MIXTURE OF GASES PARTIAL PRESSURES OF GASES CORRESPOND TO THEIR RELATIVE VOLUMES GAS Nitrogen Oxygen Argon Carbon Dioxide Neon Helium Krypton Hydrogen Xenon Water SYMBOL N 2 O 2 A CO 2 Ne He Kr H 2 X H 2 O PERCENT BY VOLUME 78 21 0. 93 0. 0018 0. 0005 0. 0001 0. 00005 0. 0000087 Variable (Page 13 manual) PARTIAL PRESSURE PASCAL TORR 593 158 7. 1 0. 25 1. 4 x 10 -2 4. 0 x 10 -3 8. 7 x 10 -4 4. 0 x 10 -4 6. 6 x 10 -5 5 to 50 79, 000 21, 000 940 33 1. 8 5. 3 x 10 -1 1. 1 x 10 -1 5. 1 x 10 -2 8. 7 x 10 -3 665 to 6650

VAPOR PRESSURE OF WATER AT VARIOUS TEMPERATURES P (mbar) T (O C) 100 (BOILING) 32 25 0 1013 (FREEZING) 6. 4 0. 13 -40 -78. 5 (DRY ICE) 6. 6 x 10 -4 -196 (LIQUID NITROGEN) 10 -24 (Page 14 manual)
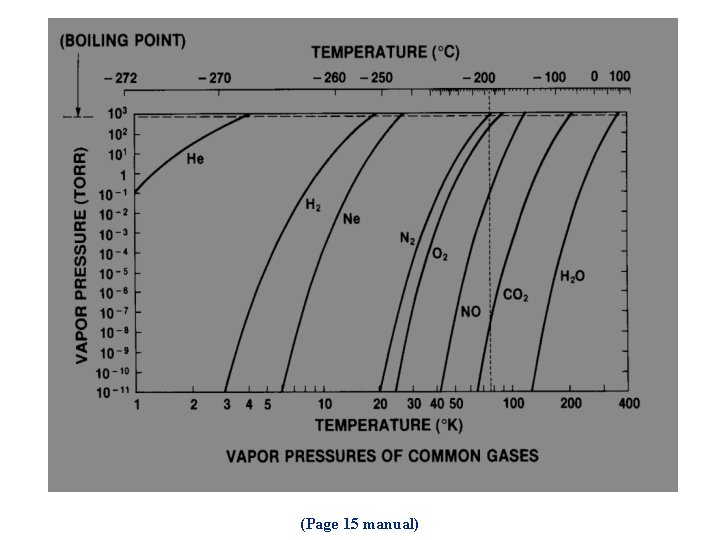
(Page 15 manual)
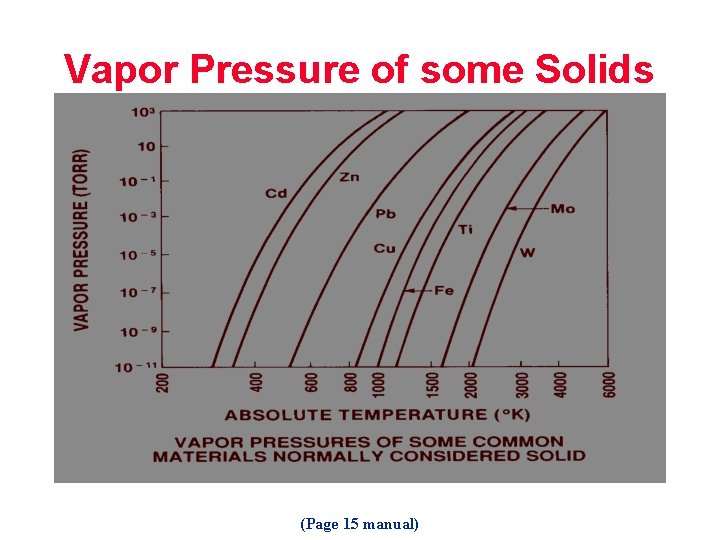
Vapor Pressure of some Solids (Page 15 manual)

PRESSURE RANGES RANGE PRESSURE ROUGH (LOW) VACUUM 759 TO 1 x 10 -3 (mbar) HIGH VACUUM 1 x 10 -3 TO 1 x 10 -8 (mbar) ULTRA HIGH VACUUM LESS THAN 1 x 10 -8 (mbar) (Page 17 manual)

GAS FLOW CONDUCTANCE (Page 24 manual)
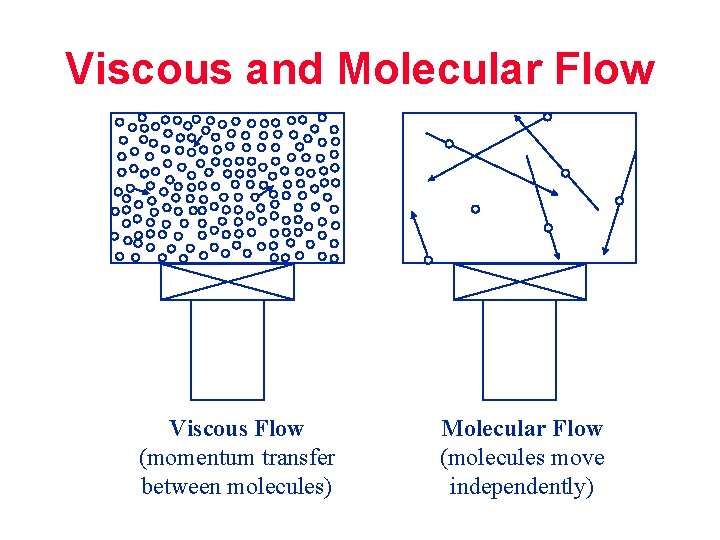
Viscous and Molecular Flow Viscous Flow (momentum transfer between molecules) Molecular Flow (molecules move independently)

FLOW REGIMES Viscous Flow: Distance between molecules is small; collisions between molecules dominate; flow through momentum transfer; generally P greater than 0. 1 mbar Transition Flow: Region between viscous and molecular flow Molecular Flow: Distance between molecules is large; collisions between molecules and wall dominate; flow through random motion; generally P smaller than 10 -3 mbar (Page 25 manual)

MEAN FREE PATH MOLECULAR DENSITY AND MEAN FREE PATH 1013 mbar (atm) 1 x 10 -3 mbar 1 x 10 -9 mbar # mol/cm 3 3 x 10 19 (30 million trillion) 4 x 10 13 (40 trillion) 4 x 10 7 (40 million) MFP 2. 5 x 10 -6 in 6. 4 x 10 -5 mm 2 inches 5. 1 cm 31 miles 50 km
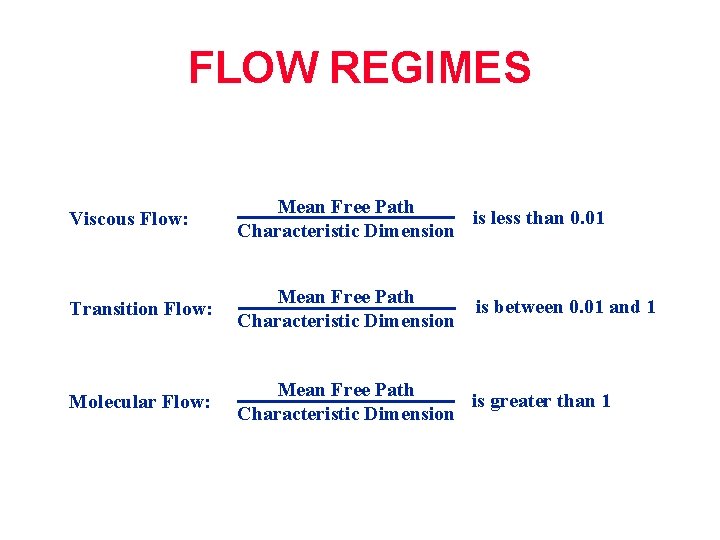
FLOW REGIMES Viscous Flow: Mean Free Path is less than 0. 01 Characteristic Dimension Transition Flow: Mean Free Path Characteristic Dimension Molecular Flow: Mean Free Path is greater than 1 Characteristic Dimension is between 0. 01 and 1

Conductance in Viscous Flow Under viscous flow conditions doubling the pipe diameter increases the conductance sixteen times. The conductance is INVERSELY related to the pipe length (Page 28 manual)

Conductance in Molecular Flow Under molecular flow conditions doubling the pipe diameter increases the conductance eight times. The conductance is INVERSELY related to the pipe length.
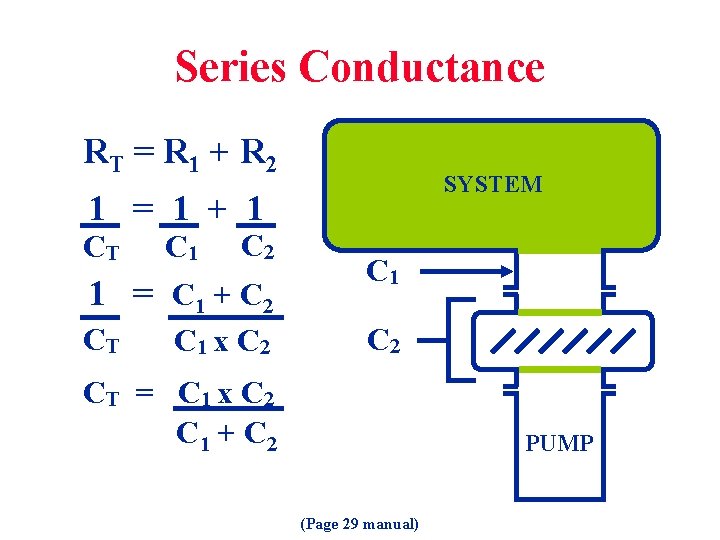
Series Conductance RT = R 1 + R 2 SYSTEM 1 = 1 + 1 CT C 1 C 2 1 = C 1 + C 2 CT C 1 x C 2 C 1 C 2 CT = C 1 x C 2 C 1 + C 2 PUMP (Page 29 manual)
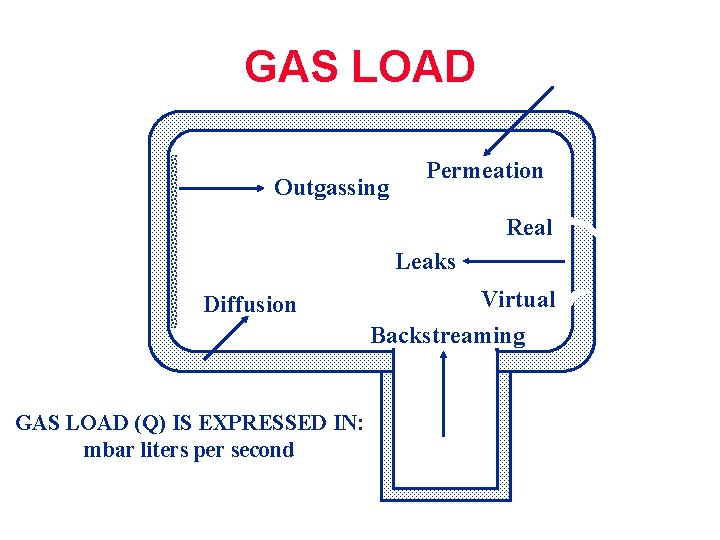
GAS LOAD Outgassing Permeation Real Leaks Diffusion GAS LOAD (Q) IS EXPRESSED IN: mbar liters per second Virtual Backstreaming
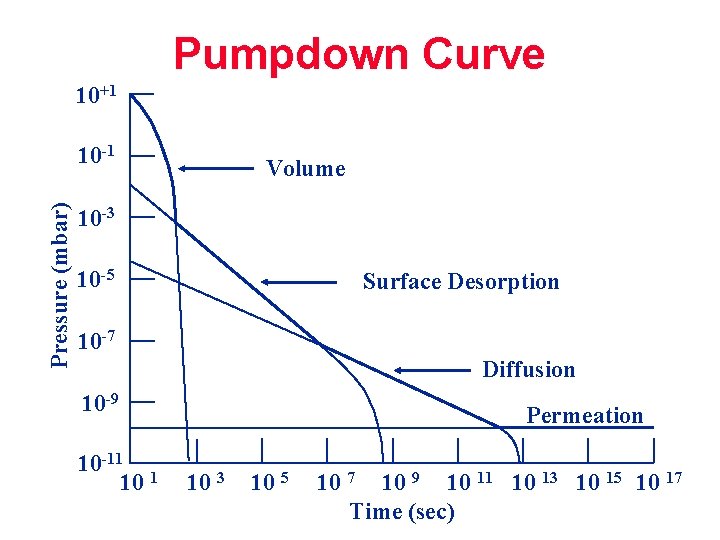
Pumpdown Curve 10+1 Pressure (mbar) 10 -1 Volume 10 -3 10 -5 Surface Desorption 10 -7 Diffusion 10 -9 10 -11 1 10 Permeation 10 3 10 5 10 7 10 9 10 11 10 13 10 15 10 17 Time (sec)

Roughing Pumps 2 (Page 39 manual)
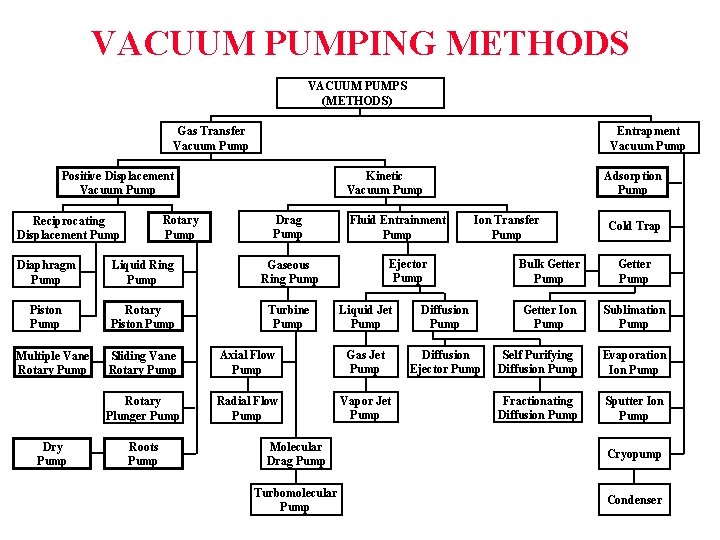
VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Gas Transfer Vacuum Pump Entrapment Vacuum Pump Kinetic Vacuum Pump Positive Displacement Vacuum Pump Rotary Pump Reciprocating Displacement Pump Drag Pump Diaphragm Pump Liquid Ring Pump Gaseous Ring Pump Piston Pump Rotary Piston Pump Turbine Pump Multiple Vane Rotary Pump Dry Pump Fluid Entrainment Pump Ion Transfer Pump Ejector Pump Liquid Jet Pump Diffusion Ejector Pump Sliding Vane Rotary Pump Axial Flow Pump Gas Jet Pump Rotary Plunger Pump Radial Flow Pump Vapor Jet Pump Roots Pump Adsorption Pump Cold Trap Bulk Getter Pump Getter Ion Pump Sublimation Pump Self Purifying Diffusion Pump Evaporation Ion Pump Fractionating Diffusion Pump Sputter Ion Pump Molecular Drag Pump Cryopump Turbomolecular Pump Condenser
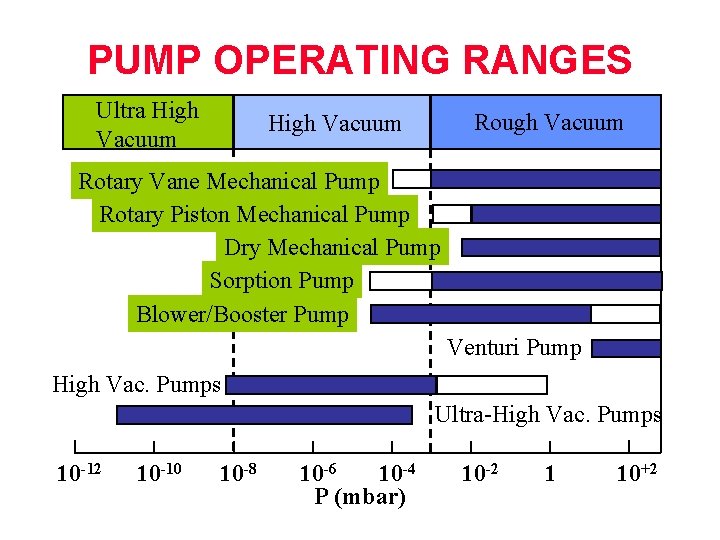
PUMP OPERATING RANGES Ultra High Vacuum Rough Vacuum High Vacuum Rotary Vane Mechanical Pump Rotary Piston Mechanical Pump Dry Mechanical Pump Sorption Pump Blower/Booster Pump Venturi Pump High Vac. Pumps Ultra-High Vac. Pumps 10 -12 10 -10 10 -8 10 -6 10 -4 P (mbar) 10 -2 1 10+2
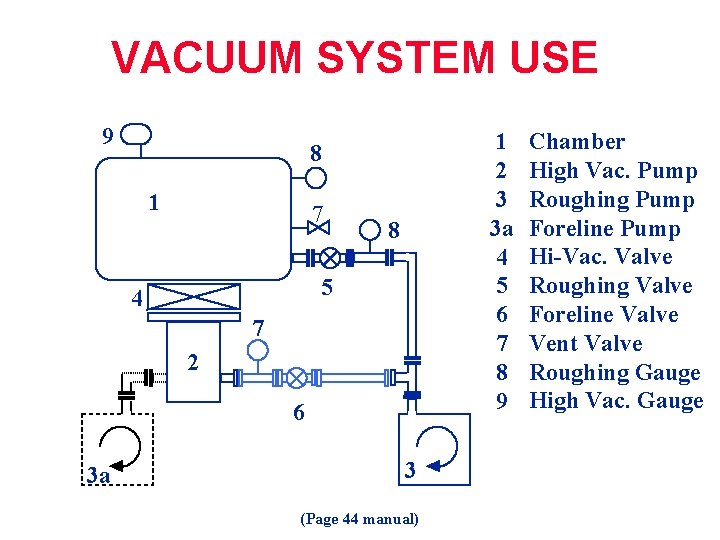
VACUUM SYSTEM USE 9 1 2 3 3 a 4 5 6 7 8 9 8 1 7 8 5 4 7 2 6 3 a 3 (Page 44 manual) Chamber High Vac. Pump Roughing Pump Foreline Pump Hi-Vac. Valve Roughing Valve Foreline Valve Vent Valve Roughing Gauge High Vac. Gauge

Rotary Vane, Oil-Sealed Mechanical Pump (Page 45 manual)

Pump Mechanism
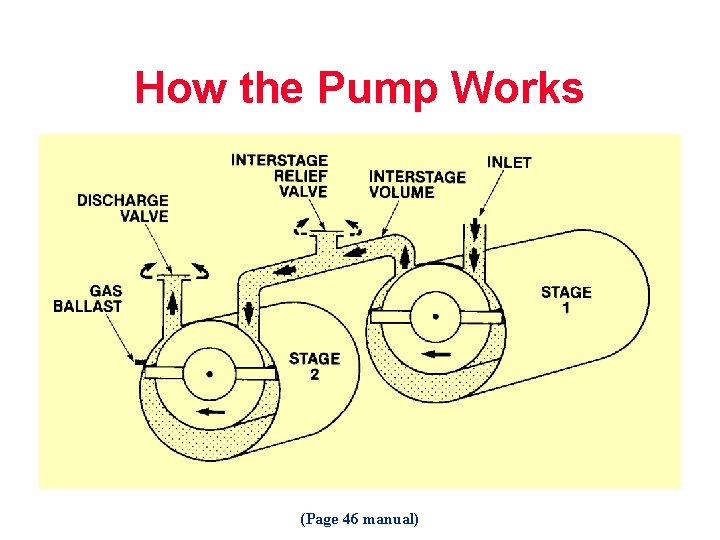
How the Pump Works (Page 46 manual)
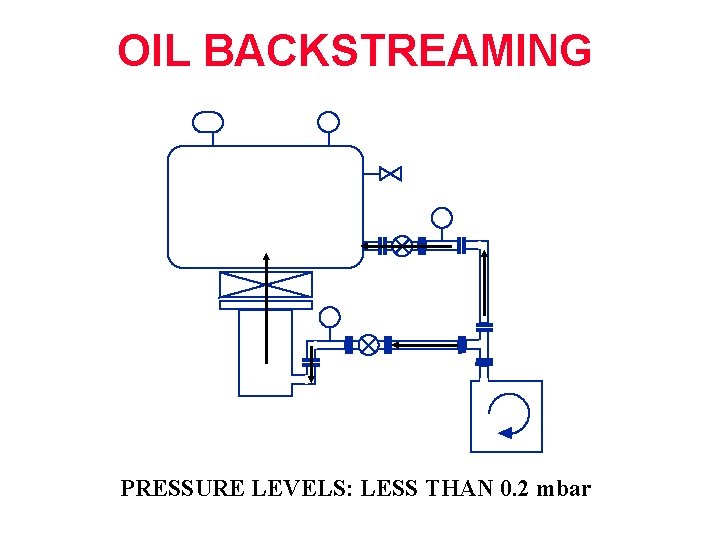
OIL BACKSTREAMING 2 PRESSURE LEVELS: LESS THAN 0. 2 mbar
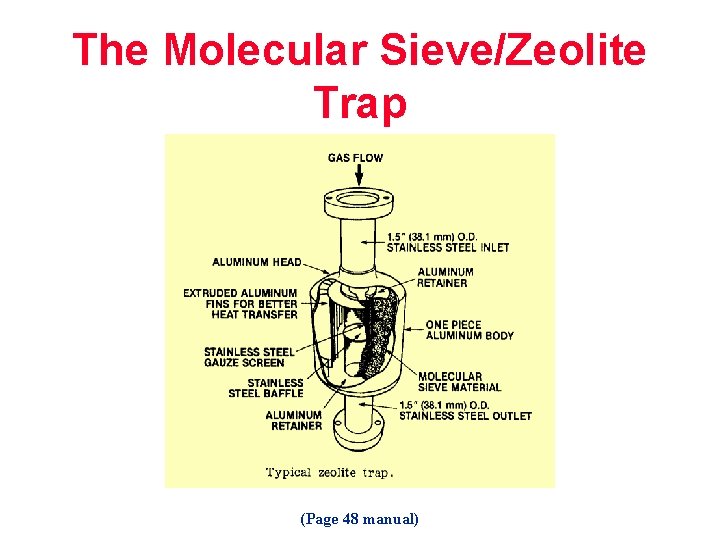
The Molecular Sieve/Zeolite Trap (Page 48 manual)

Dry Vacuum Pumps
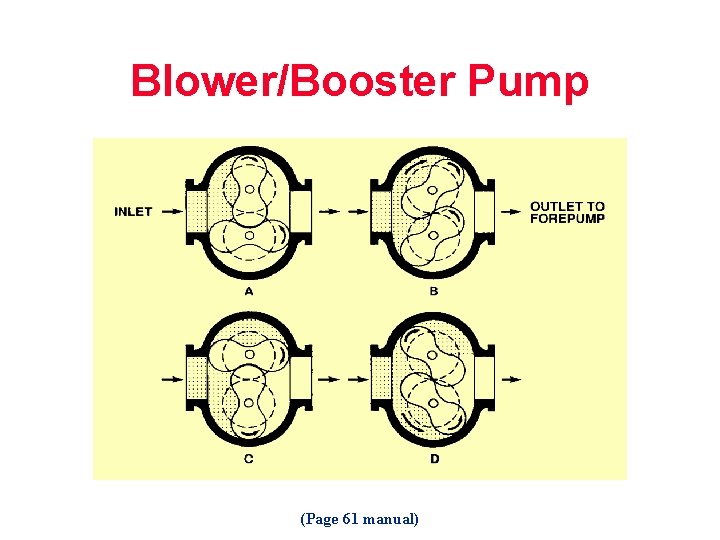
Blower/Booster Pump (Page 61 manual)

One Stage Roots Blower Pump Assembly
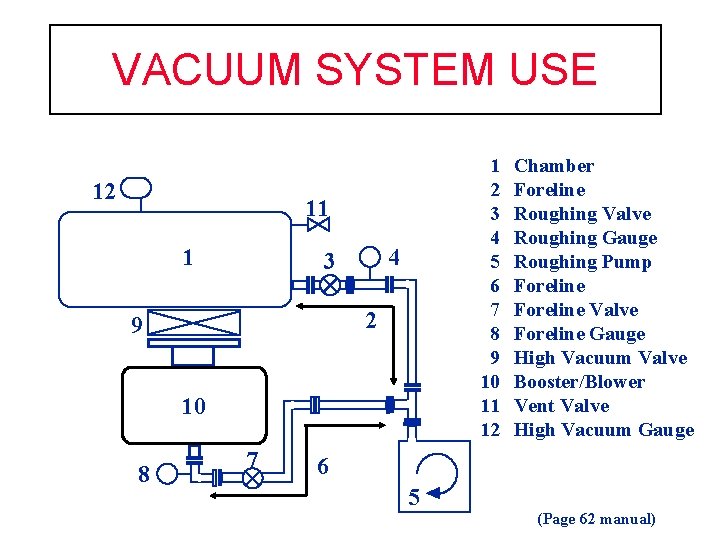
VACUUM SYSTEM USE 12 1 2 3 4 5 6 7 8 9 10 11 12 11 1 4 3 2 9 10 8 7 Chamber Foreline Roughing Valve Roughing Gauge Roughing Pump Foreline Valve Foreline Gauge High Vacuum Valve Booster/Blower Vent Valve High Vacuum Gauge 6 5 (Page 62 manual)

Sorption Pump Components (Page 54 manual)
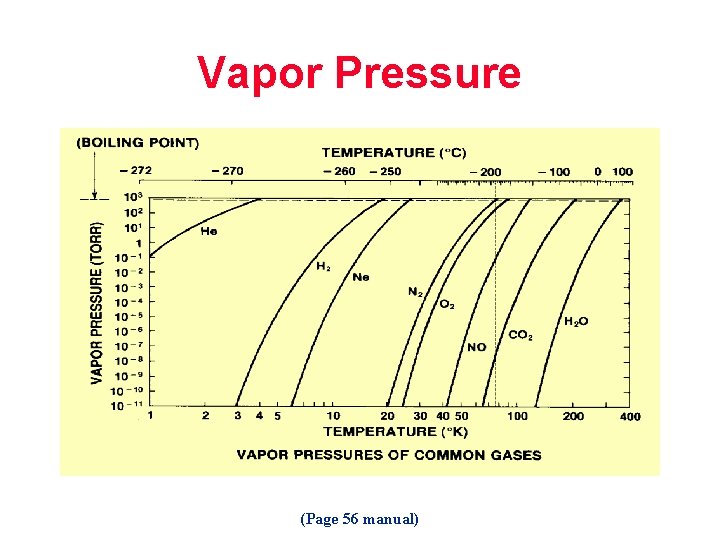
Vapor Pressure (Page 56 manual)

Cryo-condensation
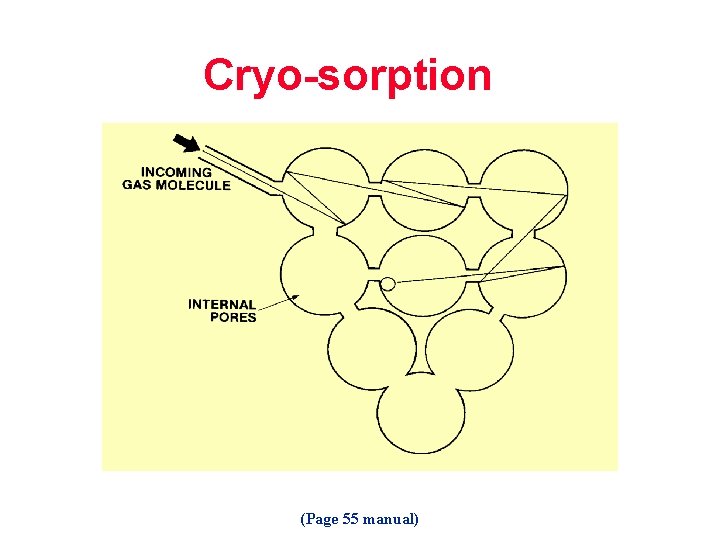
Cryo-sorption (Page 55 manual)

HIGH VACUUM PUMPS 3 (Page 63 manual)
VACUUM PUMPING METHODS VACUUM PUMPS (METHODS) Gas Transfer Vacuum Pump Entrapment Vacuum Pump Kinetic Vacuum Pump Positive Displacement Vacuum Pump Rotary Pump Reciprocating Displacement Pump Diaphragm Pump Liquid Ring Pump Piston Pump Rotary Piston Pump Multiple Vane Rotary Pump Drag Pump Fluid Entrainment Pump Liquid Jet Pump Diffusion Ejector Pump Sliding Vane Rotary Pump Axial Flow Pump Gas Jet Pump Rotary Plunger Pump Radial Flow Pump Vapor Jet Pump Roots Pump Ion Transfer Pump Ejector Pump Gaseous Ring Pump Turbine Pump Adsorption Pump Cold Trap Bulk Getter Pump Getter Ion Pump Sublimation Pump Self Purifying Diffusion Pump Evaporation Ion Pump Fractionating Diffusion Pump Sputter Ion Pump Molecular Drag Pump Cryopump Turbomolecular Pump Condenser
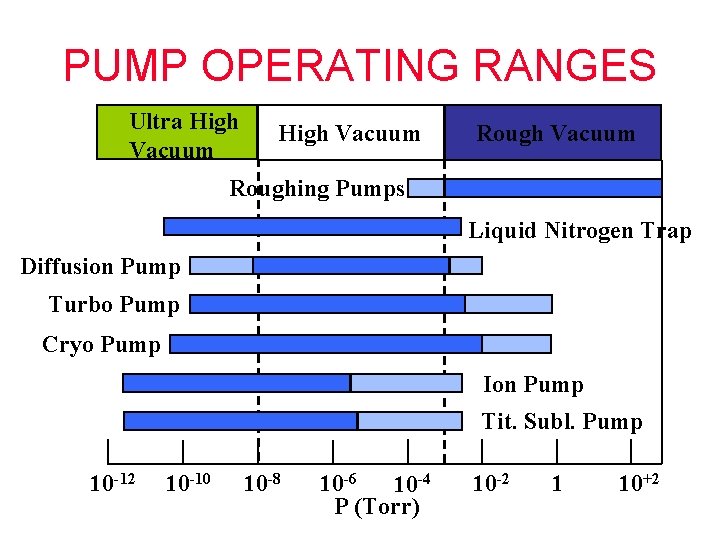
PUMP OPERATING RANGES Ultra High Vacuum Roughing Pumps Liquid Nitrogen Trap Diffusion Pump Turbo Pump Cryo Pump Ion Pump Tit. Subl. Pump 10 -12 10 -10 10 -8 10 -6 10 -4 P (Torr) 10 -2 1 10+2
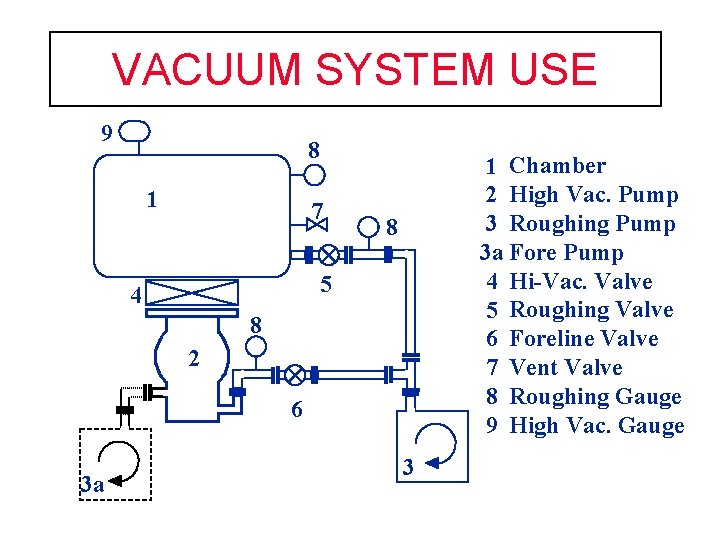
VACUUM SYSTEM USE 9 8 1 7 1 Chamber 2 High Vac. Pump 3 Roughing Pump 3 a Fore Pump 4 Hi-Vac. Valve 5 Roughing Valve 6 Foreline Valve 7 Vent Valve 8 Roughing Gauge 9 High Vac. Gauge 8 5 4 8 22 6 3 a 3

Oil Diffusion Pump

Pump Construction (Page 66 manual)
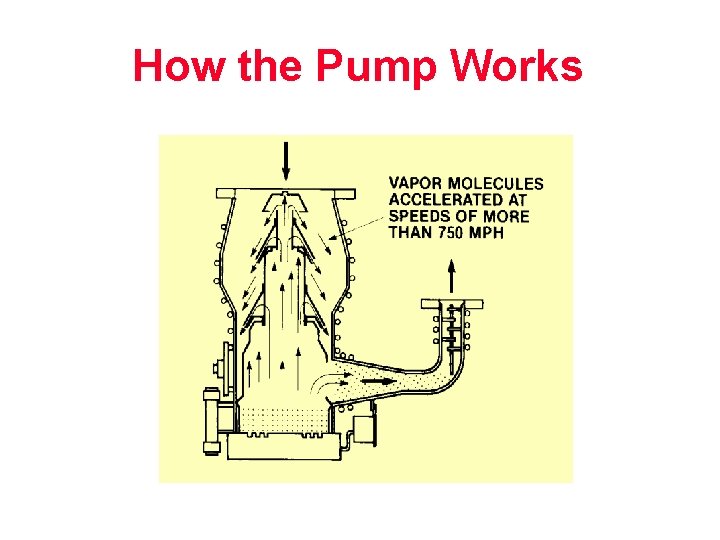
How the Pump Works
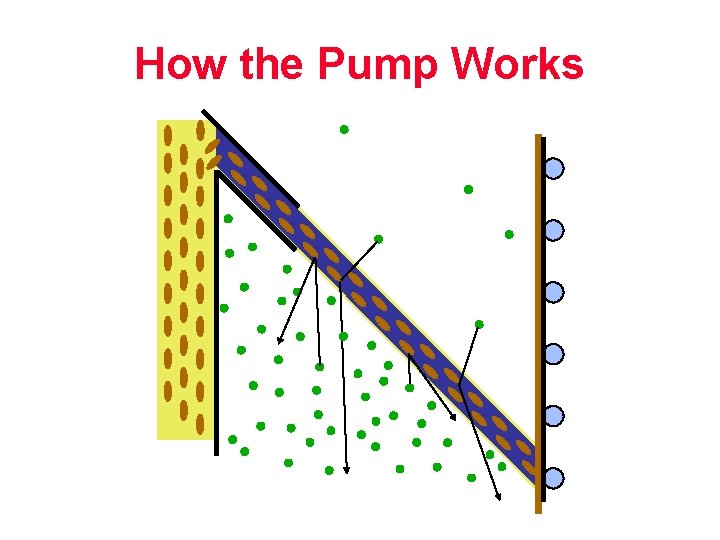
How the Pump Works
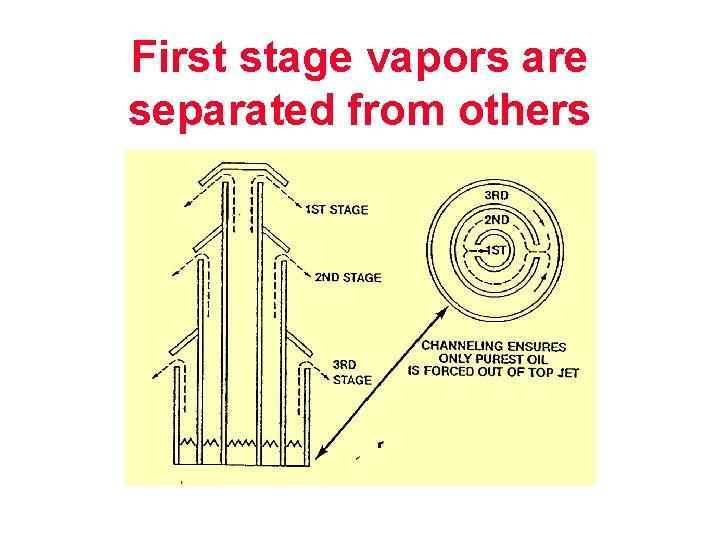
First stage vapors are separated from others

Pumping Speed (Air) 1 2 3 4 Critical Point 1. Compression Ratio Limit 2. Constant Speed 3. Constant Q (Overload) 4. Mechanical Pump Effect 10 -10 10 --3 Inlet Pressure (Torr) 10 --1
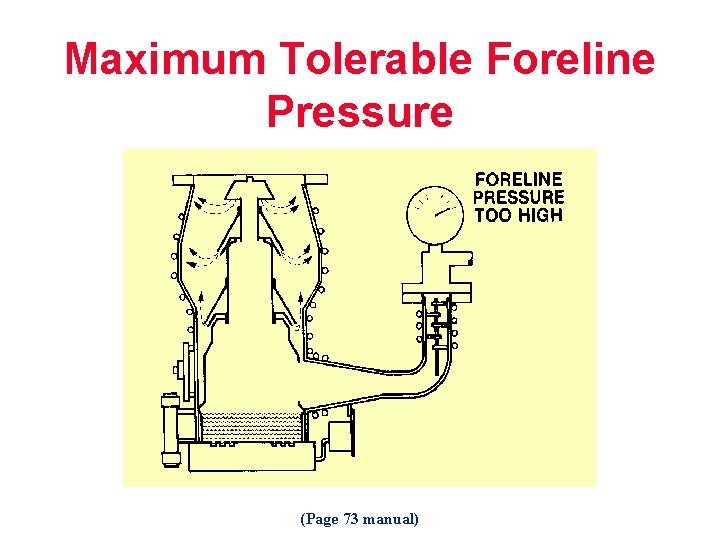
Maximum Tolerable Foreline Pressure (Page 73 manual)
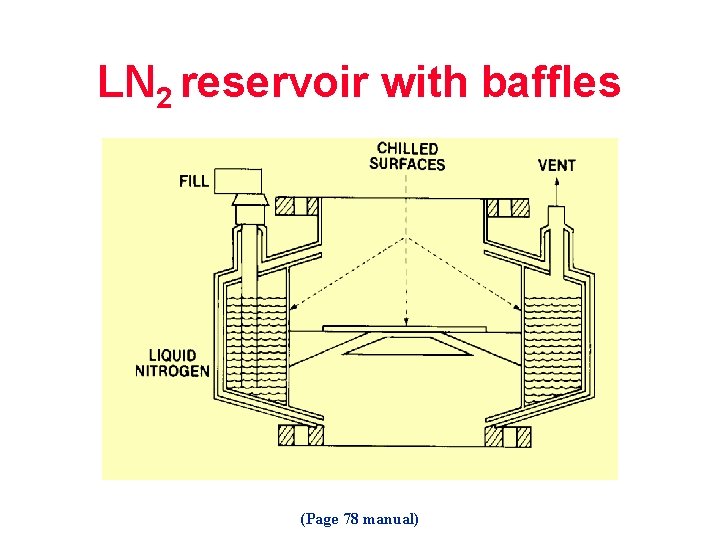
LN 2 reservoir with baffles (Page 78 manual)

How the LN 2 Trap Works Approximate Vapor Pressure (mbar) Gas Water (H 2 O) Argon (A) Carbon Dioxide (CO 2) Carbon Monoxide (CO) Helium (He) Hydrogen (H 2) Oxygen (O 2) Neon (Ne) Nitrogen (N 2) Solvents (Page 79 manual) 10 -22 500 10 -7 >760 350 >760 <10 -10
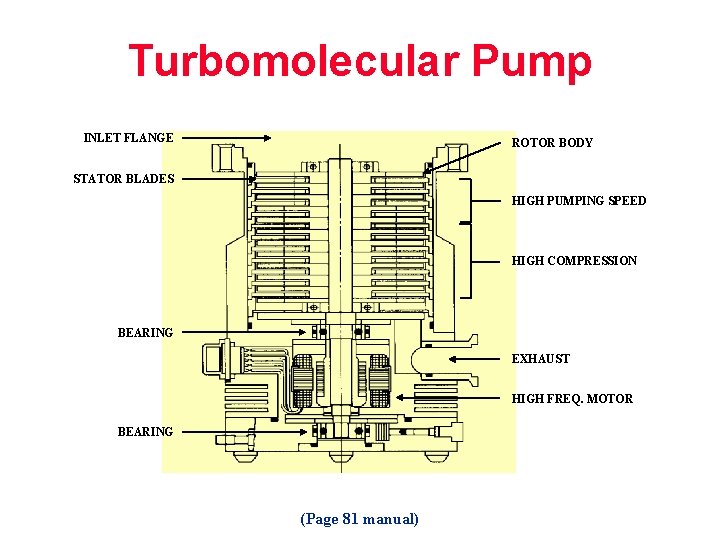
Turbomolecular Pump INLET FLANGE ROTOR BODY STATOR BLADES HIGH PUMPING SPEED HIGH COMPRESSION BEARING EXHAUST HIGH FREQ. MOTOR BEARING (Page 81 manual)
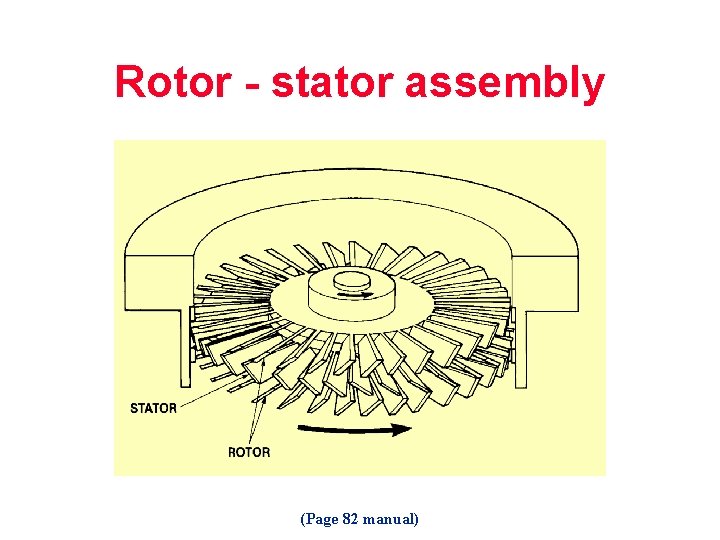
Rotor - stator assembly (Page 82 manual)
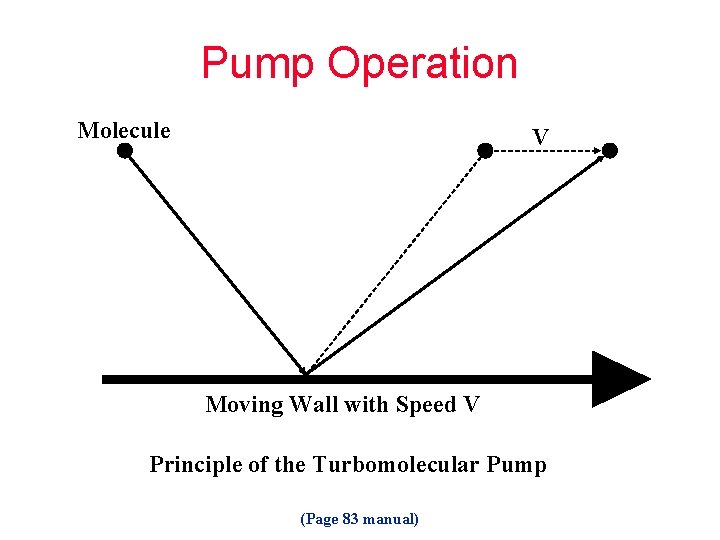
Pump Operation Molecule V Moving Wall with Speed V Principle of the Turbomolecular Pump (Page 83 manual)
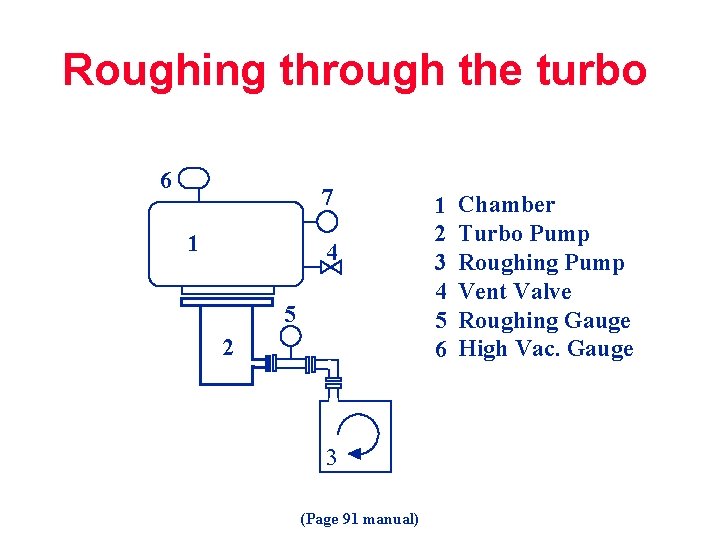
Roughing through the turbo 6 7 1 4 5 22 3 (Page 91 manual) 1 2 3 4 5 6 Chamber Turbo Pump Roughing Pump Vent Valve Roughing Gauge High Vac. Gauge
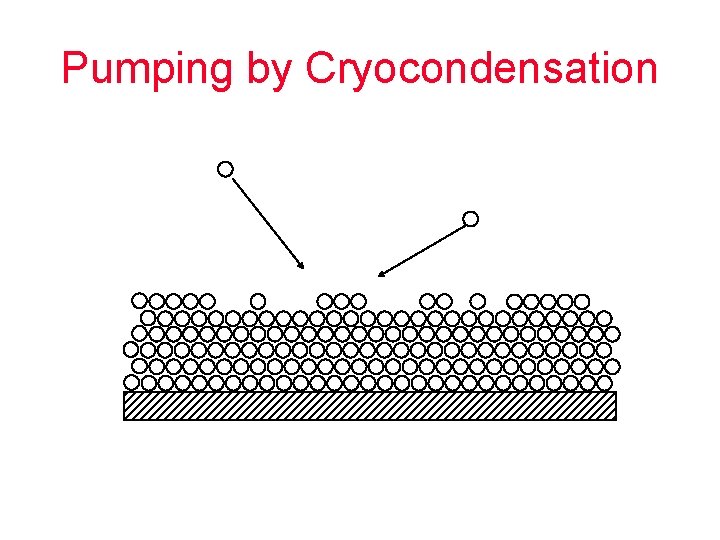
Pumping by Cryocondensation
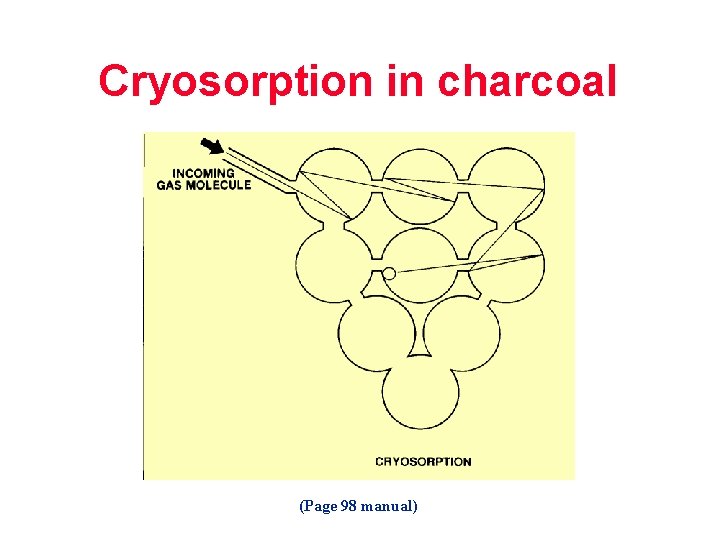
Cryosorption in charcoal (Page 98 manual)
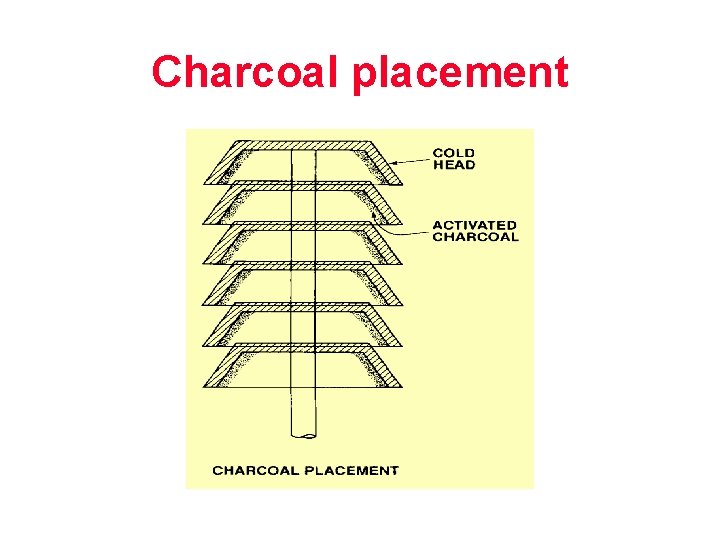
Charcoal placement

Gauges 5 (Page 123 manual)
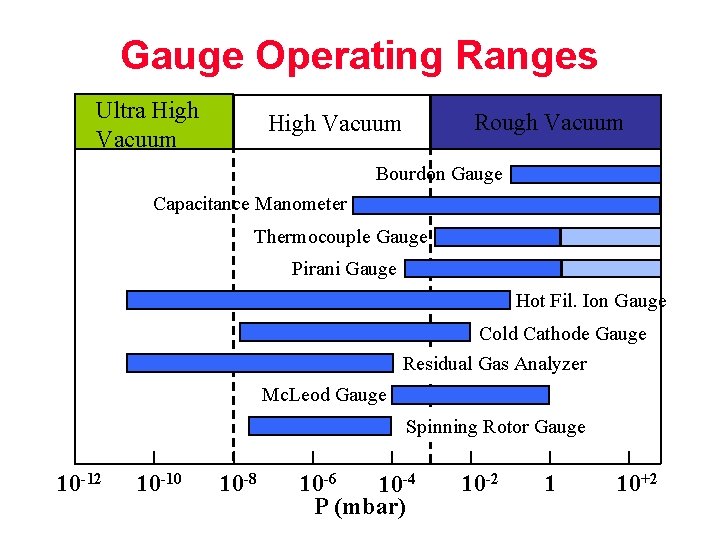
Gauge Operating Ranges Ultra High Vacuum Rough Vacuum High Vacuum Bourdon Gauge Capacitance Manometer Thermocouple Gauge Pirani Gauge Hot Fil. Ion Gauge Cold Cathode Gauge Residual Gas Analyzer Mc. Leod Gauge Spinning Rotor Gauge 10 -12 10 -10 10 -8 10 -6 10 -4 P (mbar) 10 -2 1 10+2

Bourdon Gauge
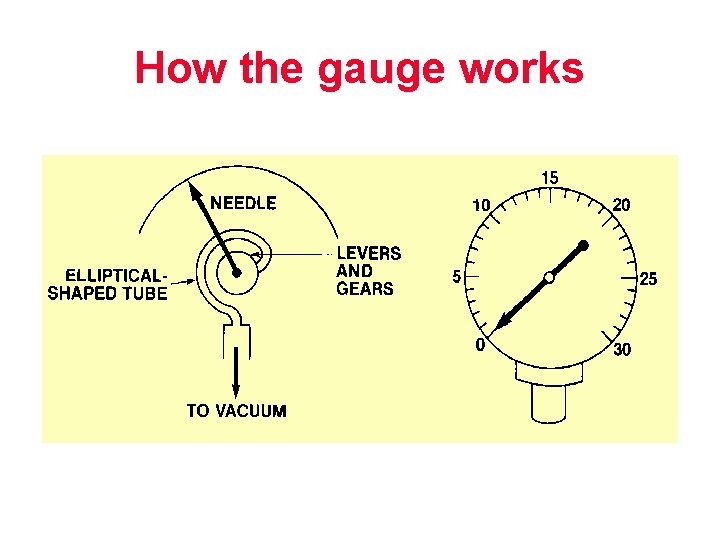
How the gauge works

Heat Transfer Gauges Thermocouple gauge and Pirani Gauge

Thermocouple Gauge
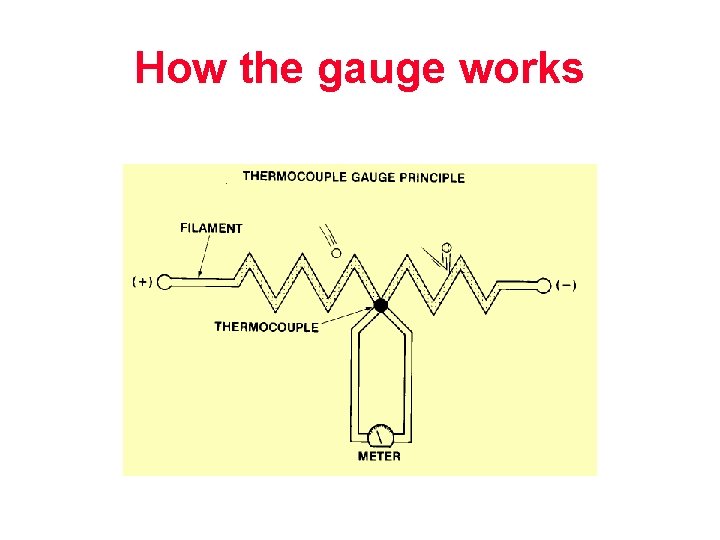
How the gauge works
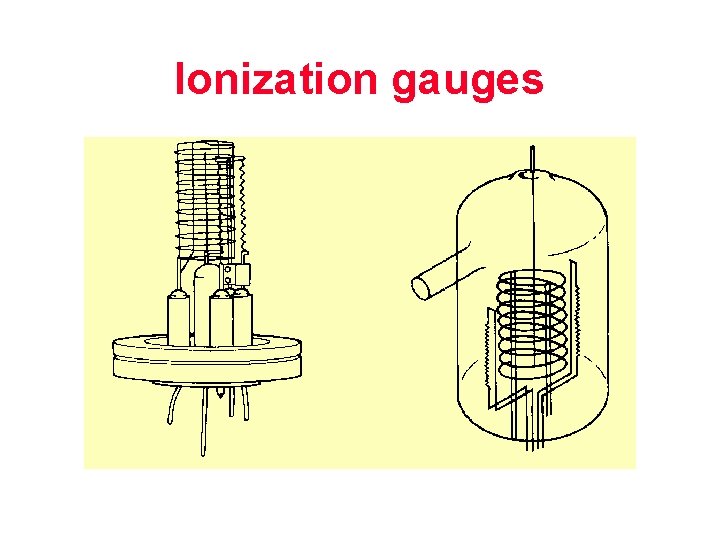
Ionization gauges
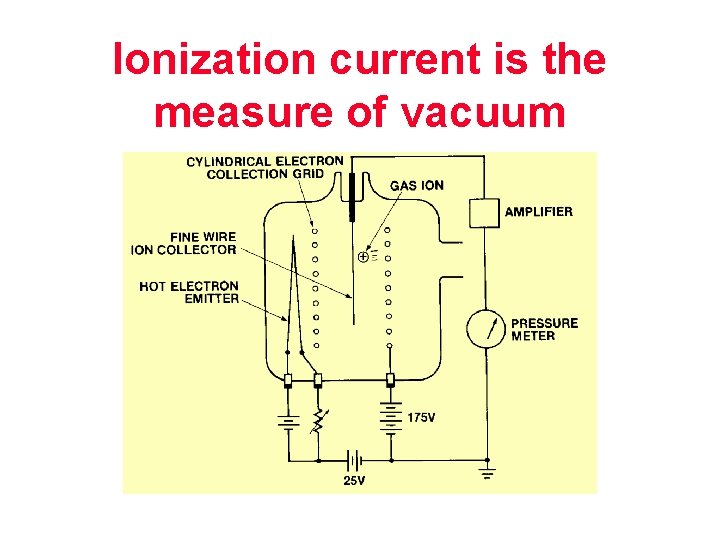
Ionization current is the measure of vacuum
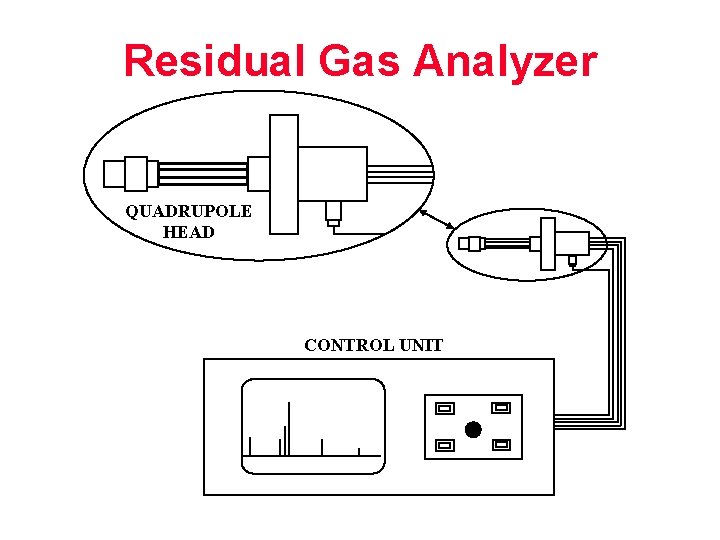
Residual Gas Analyzer QUADRUPOLE HEAD CONTROL UNIT
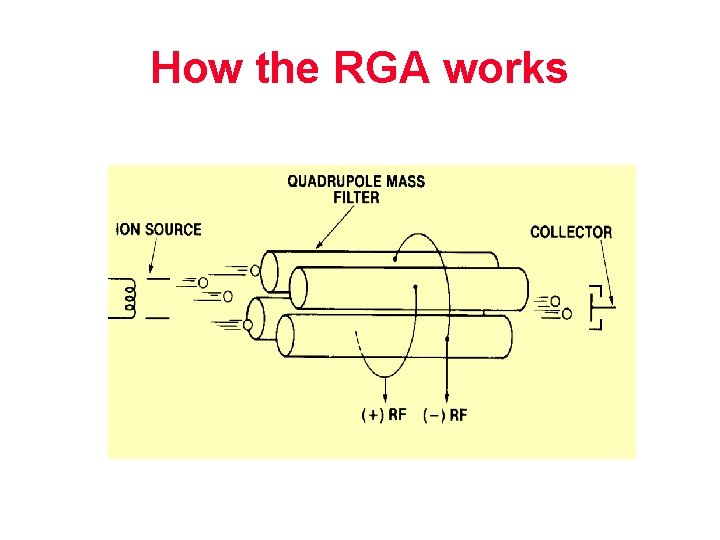
How the RGA works
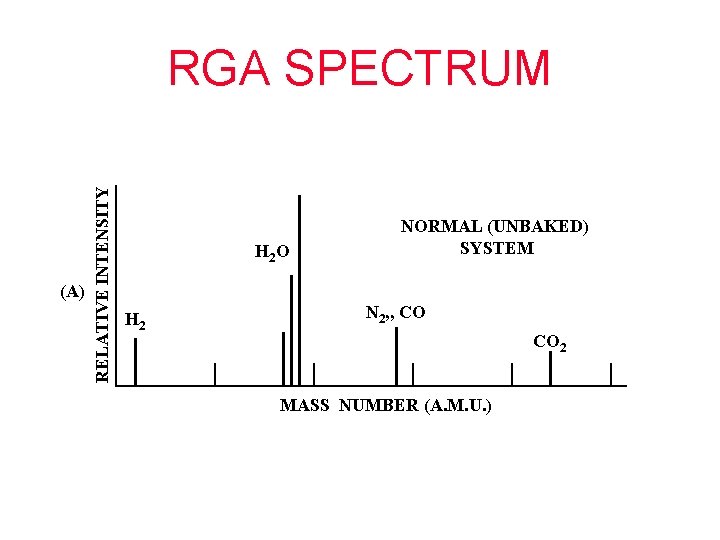
(A) RELATIVE INTENSITY RGA SPECTRUM H 2 O H 2 NORMAL (UNBAKED) SYSTEM N 2, , CO CO 2 MASS NUMBER (A. M. U. )

(B) RELATIVE INTENSITY RGA SPECTRUM N 2 SYSTEM WITH AIR LEAK H 2 O O 2 H 2 CO 2 MASS NUMBER (A. M. U. )

LEAK DETECTION 9 (Page 249 manual)
Introduction
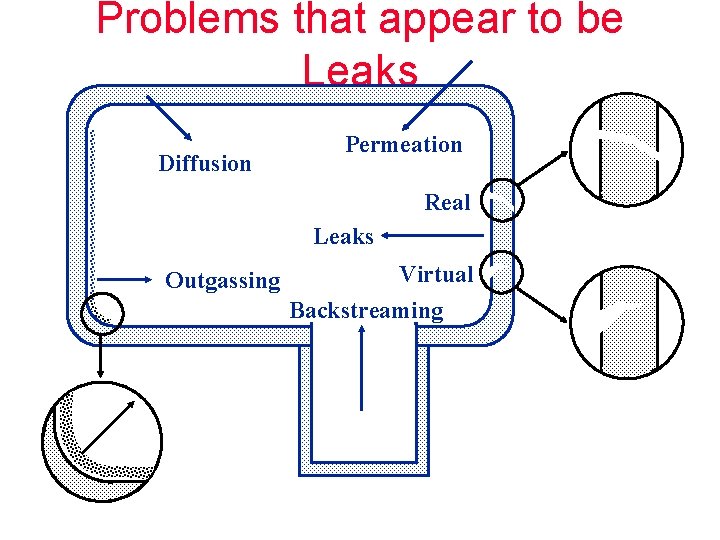
Problems that appear to be Leaks Diffusion Permeation Real Leaks Outgassing Virtual Backstreaming
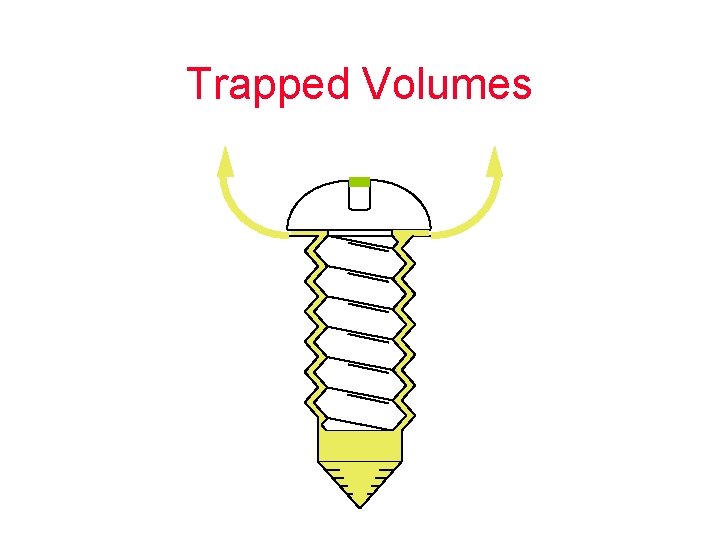
Trapped Volumes
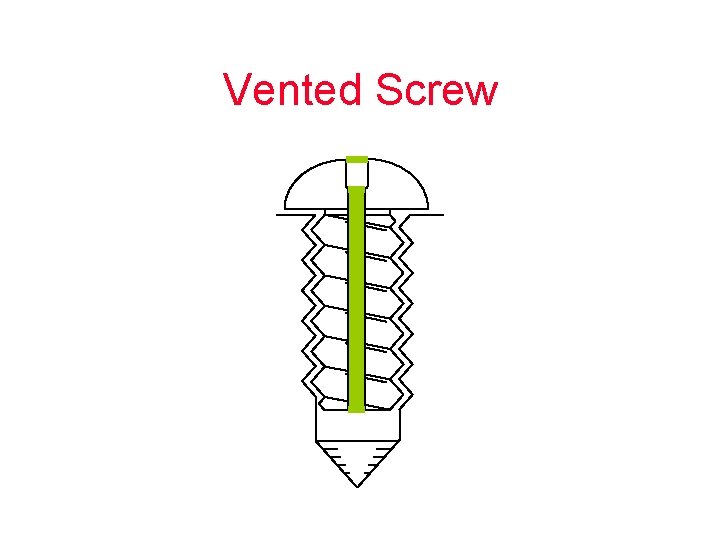
Vented Screw
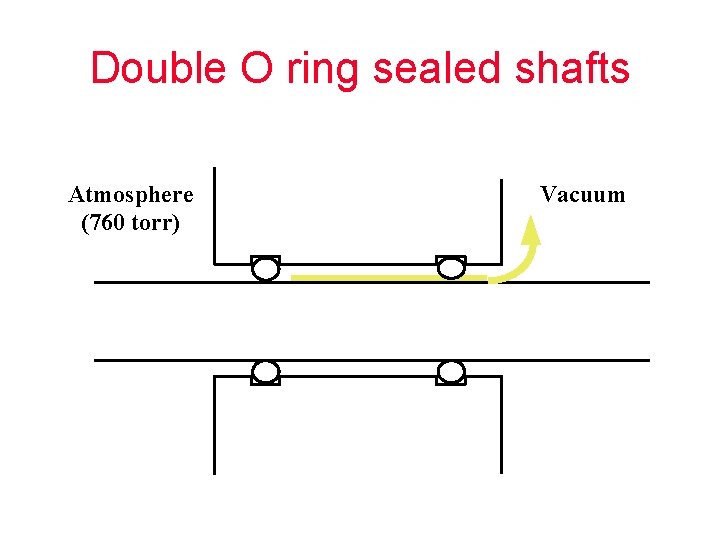
Double O ring sealed shafts Atmosphere (760 torr) Vacuum
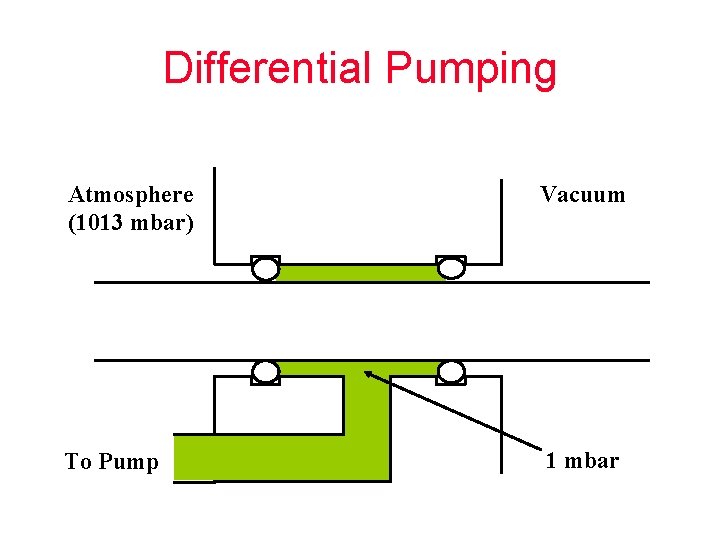
Differential Pumping Atmosphere (1013 mbar) Vacuum To Pump 1 mbar

PERMEATION LEAKS Permeation “leaks” are different than real leaks because the only way to stop them is to change to a less permeable material
One standard cubic centimeter/sec (std. cc/sec)

Leak rate of 1 x 10 -1 std cc/sec
Leak rate of 1 x 10 -3 std cc/sec

Leak Rates over Time LEAK RATES 10 -1 STD CC/SEC --- 1 CC/10 SEC 10 -3 STD CC/SEC --- 3 CC/HOUR 10 -5 STD CC/SEC --- 1 CC/DAY 10 -6 STD CC/SEC --- 1 CC/2 WEEKS 10 -7 STD CC/SEC --- 3 CC/YEAR 10 -9 STD CC/SEC --- 1 CC/30 YEARS
Why Helium is used

HELIUM • • • Helium is very light and small Low concentration in air (0. 0005%) Permits dynamic testing Permits non-destructive testing Helium is safe

CONVENTIONAL LEAK DETECTOR 1 10 9 2 11 12 7 8 3 5 6 4 1 2 3 4 5 6 7 8 9 10 11 12 Test Piece Test Port High Vac. Pump Roughing Pump Fore Pump Roughing. Valve Test Valve Pump Valve Spectrometer Tube Cold Trap Roughing Gauge Vent Valve
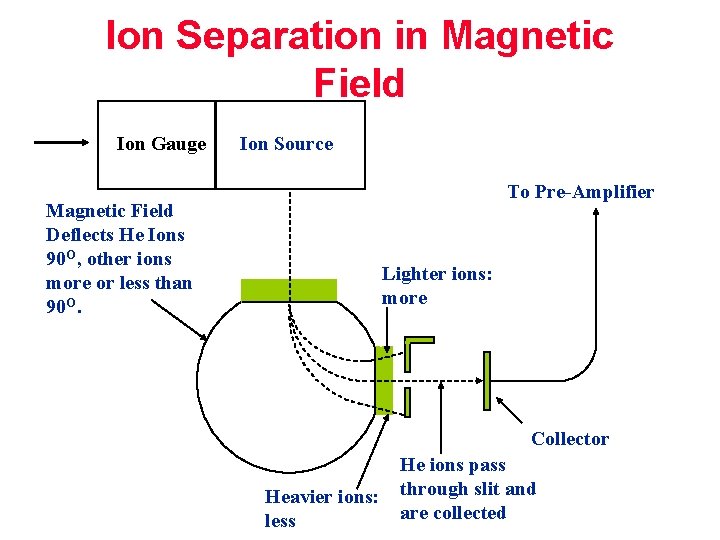
Ion Separation in Magnetic Field Ion Gauge Ion Source To Pre-Amplifier Magnetic Field Deflects He Ions 90 O, other ions more or less than 90 O. Lighter ions: more Collector Heavier ions: less He ions pass through slit and are collected
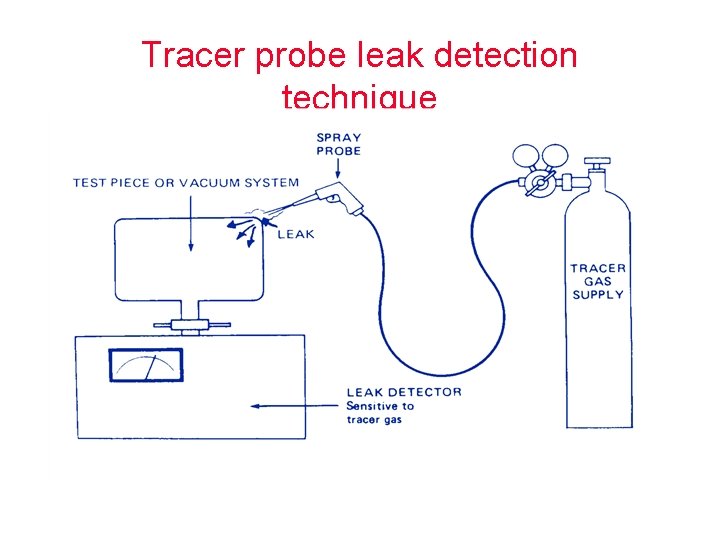
Tracer probe leak detection technique
- Slides: 90