Basic Principles of GMP Sanitation and Hygiene Section

Basic Principles of GMP Sanitation and Hygiene Section 3 | Module 3 January 2006 Slide 1 of 30 STOP

Sanitation and Hygiene Objectives l Review measures to ensure good sanitation in: ä Premises and personnel ä Equipment and apparatus ä Processes, materials and containers l To review measures to ensure good personal hygiene l Group session - to discuss the situation in your country and to look at some bad sanitation and hygiene practices | Module 3 January 2006 Slide 2 of 30 STOP

Sanitation and Hygiene Scope High level of sanitation and hygiene practised – in every aspect of manufacturing. It covers: l. Personnel l. Premises l. Equipment and apparatus l. Production materials and containers l. Products for cleaning and disinfection l. All potential sources of cross-contamination 3. 1 | Module 3 January 2006 Slide 3 of 30 STOP

Sanitation and Hygiene Personal Hygiene (1) l Health examinations: ä Before and during employment ä Periodic eye examinations for those who do visual inspections l Training: ä Practices in personal hygiene ä Written procedures and instructions ä Signs in areas 11. 1 - 2 | Module 3 January 2006 Slide 4 of 30 STOP

Basic Principles of GMP ä Written procedures and instructions - to wash hands before entering production areas ä Some also use disinfectants 11. 1 - 2 | Module 3 January 2006 Slide 5 of 30 STOP

Basic Principles of GMP ä Wash hands before entering production areas ä Signs in areas (e. g. changing rooms) 11. 1 - 2 | Module 3 January 2006 Slide 6 of 30 STOP

Sanitation and Hygiene Personal Hygiene (2) l Illness or open lesions: ä May affect the quality of products ä Should not handle starting materials, intermediates or finished products, etc. ä Instruction and encouragement to report to supervisors l Direct contact between product and operator: ä Should be avoided ä Starting materials, primary packaging materials, intermediate and bulk product 11. 2 - 5 | Module 3 January 2006 Slide 7 of 30 STOP

Sanitation and Hygiene Personnel Hygiene (3) l Protection of product from contamination: ä Clean clothes appropriate to personnel activities ä Including hair covering (e. g. caps) l Check change rooms/changing facilities ä Hand washing, signs, drying of hands ä Used clothing stored in separate closed containers while awaiting cleaning ä Laundering of clean area clothing according to an SOP and in an appropriate facility ä Procedure for disinfecting and sterilizing when required 11. 6 | Module 3 January 2006 Slide 8 of 30 STOP

Sanitation and Hygiene Personnel Hygiene (4) l Smoking, eating and drinking not allowed in production areas, laboratories and storage areas l No chewing (e. g. gum), or keeping food or drinks allowed l No plants kept inside these areas l Rest and refreshment areas should be separate from manufacturing and control areas 11. 7, 12. 11 | Module 3 January 2006 Slide 9 of 30 STOP
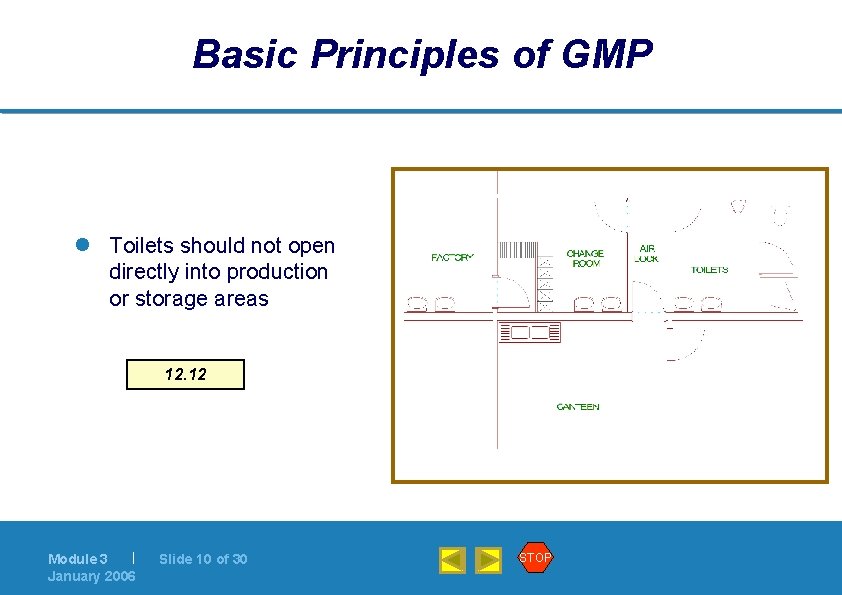
Basic Principles of GMP l Toilets should not open directly into production or storage areas 12. 12 | Module 3 January 2006 Slide 10 of 30 STOP

Sanitation and Hygiene Personnel Hygiene (5) l Personal hygiene procedures including wearing protective clothing apply to all persons entering into production areas: ä Full-time employees ä Temporary workers ä Contractor's employees ä Visitors ä Managers ä Inspectors 11. 8 | Module 3 January 2006 Slide 11 of 30 STOP

Sanitation and Hygiene Design of Premises l Design ä Walls, floors, ceilings, ledges, drains, air supply, dust extraction l Prevention of build-up of dirt and dust to avoid unnecessary risks of contamination ä Cleaning programme, appropriate cleaning, cleaning records l Effective cleaning and disinfection ä choice of materials and chemicals, validation l Drains – prevent backflow 12. 2, 12. 3, 12. 7, 12. 9, 12. 29 | Module 3 January 2006 Slide 12 of 30 STOP

Basic Principles of GMP l Protection from insects, birds, vermin and weather – from receipt of raw materials to dispatch of released product 12. 9 | Module 3 January 2006 Slide 13 of 30 STOP

Sanitation and Hygiene Avoidance of Cross-Contamination (1) l Special precautions should be taken to prevent generation and dissemination of dust l Proper air control – supply and extraction, suitable quality l Due to uncontrolled release of: ä dust, gas, particles, vapours, sprays, organisms, residue, insects 16. 10 - 11 | Module 3 January 2006 Slide 14 of 30 STOP

Basic Principles of GMP Measures that can be taken to prevent cross-contamination also include: l Segregated areas l Ventilation systems l Airlocks l Clothing l Closed processing systems l Cleaning and decontamination | Module 3 January 2006 Slide 15 of 30 STOP

Sanitation and Hygiene Avoidance of Cross-Contamination (2) l Dedicated and self-contained areas for: ä Live vaccines ä Live bacterial preparations ä Certain other biological materials ä Penicillin products 16. 12(a) | Module 3 January 2006 Slide 16 of 30 STOP

Sanitation and Hygiene Avoidance of Cross-Contamination (3) l Campaign production: ä Separation in time ä Followed by appropriate cleaning ä Validated cleaning procedure 16. 12(b) | Module 3 January 2006 Slide 17 of 30 STOP

Sanitation and Hygiene Avoidance of Cross-Contamination (4) l Ventilation systems and airlocks ä Appropriately designed ventilation system with air supply and extraction systems ä Supply or incoming air should be filtered ä Recirculation of air versus 100% fresh air supply ä Proper airflow patterns ä Pressure differentials ä Appropriately designed airlocks 16. 12 (c and d) | Module 3 January 2006 Slide 18 of 30 STOP

Basic Principles of GMP ä Appropriately designed ventilation system with air supply and extraction systems ä Supply or incoming air should be filtered l Detailed modules in the supplementary training deal with recommendations for HVAC systems | Module 3 January 2006 Slide 19 of 30 STOP

Sanitation and Hygiene Avoidance of Cross-Contamination (5) l Clothing ä Protection of operator and product ä Highly potent products or those of particular risk - need for special protective clothing ä Personnel should not move between areas producing different products ä Garments need to be cleaned 16. 12(e) | Module 3 January 2006 Slide 20 of 30 STOP

Sanitation and Hygiene Avoidance of Cross-Contamination (6) l Cleaning and decontamination ä Procedure for removing soil and dirt ä Remove all cleaning chemical residues or disinfectant residues ä Remove and/or reduce micro-organisms ä Validated (known effectiveness of the procedure) ä Use cleanliness status labels ä Test for residues 16. 12(f, h and i) | Module 3 January 2006 Slide 21 of 30 STOP

Sanitation and Hygiene Avoidance of Cross-Contamination (7) l Closed processing systems ä For example: totally enclosed water purification systems ä Tanks fitted with appropriate filtration - without removable lids ä Present special cleaning difficulties, sometimes use clean-in-place (CIP) 16. 12(g) | Module 3 January 2006 Slide 22 of 30 STOP

Sanitation and Hygiene Production Operations – Sanitation (1) l Cleaning and cleaning validation ä Degree of cleaning depends on whether consecutive batches are of same or different product l Check cleaning agent is fully removed l If possible hot water alone used for cleaning ä all cleaning and disinfecting solutions carefully prepared and expiry dated l Final rinse with purified water, or water for injection (for sterile products) l Full records kept | Module 3 January 2006 Slide 23 of 30 STOP

Sanitation and Hygiene Production Operations – Sanitation (2) l Full records kept l Water systems l Water - major constituent of most products l SOP for cleaning and sanitization of the water purification system should include distribution line l Validation and removal of disinfectant before reuse | Module 3 January 2006 Slide 24 of 30 STOP

Sanitation and Hygiene Production Operations – Sanitation (3) l Maintenance and repair ä activities inevitable in manufacturing area ä Should present no risk to product l Whenever possible, all planned maintenance outside normal operating hours l Emergency work in working area followed by thorough clean down and disinfection before manufacturing recommences l Area clearance by QC | Module 3 January 2006 Slide 25 of 30 STOP

Basic Principles of GMP | Module 3 January 2006 Slide 26 of 30 STOP

Sanitation and Hygiene Group Session - Option 1 Look at the photographs in the handout and record as many sanitation and hygiene issues as you can | Module 3 January 2006 Slide 27 of 30 STOP

Sanitation and Hygiene Group Session - Option 2 You are inspecting a new factory. What are the key issues for sanitation and the key issues for personnel hygiene that the company should have in place? | Module 3 January 2006 Slide 28 of 30 STOP

Sanitation and Hygiene Possible Issues – I Sanitation l Mixed production l Penicillins l Product versus batch changeovers l Water systems l How long should a “cleaned” status last for? l What should happen if a clearance check is required when no QC personnel are on duty? l Procedures and records | Module 3 January 2006 Slide 29 of 30 STOP

Sanitation and Hygiene Possible Issues – II Hygiene l Personal hygiene l Health checks l Dealing with health problems l Personal responsibility l Training records l Frequency of handwashing | Module 3 January 2006 Slide 30 of 30 STOP
- Slides: 30