BASIC PRINCIPLES OF ELECTRODE PROCESSES Heterogeneous kinetics II

BASIC PRINCIPLES OF ELECTRODE PROCESSES Heterogeneous kinetics II
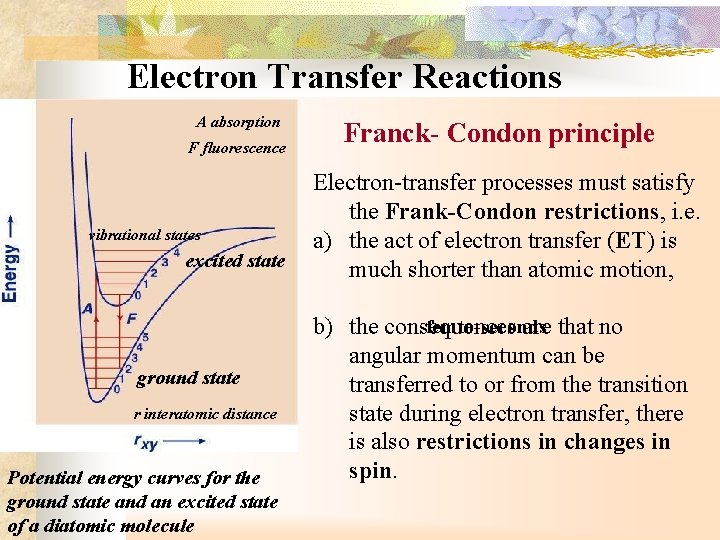
Electron Transfer Reactions A absorption F fluorescence vibrational states excited state ground state r interatomic distance Potential energy curves for the ground state and an excited state of a diatomic molecule Franck- Condon principle Electron-transfer processes must satisfy the Frank-Condon restrictions, i. e. a) the act of electron transfer (ET) is much shorter than atomic motion, femto-seconds b) the consequences are that no angular momentum can be transferred to or from the transition state during electron transfer, there is also restrictions in changes in spin.


Electron transfer in biochemistry Aox + Bred « Ared + Box Aox is the oxidized form of A (the oxidant in the reaction shown) Bred is the reduced form of B (the reductant). For such an electron transfer, one may consider two half-cell reactions: 1. A + z e- « A . . . e. g. , Fe+++ + e- « Fe++ 2. B + z e- « B ox red For each half reaction: E = E°' - RT/z. F (ln [red]/[ox]) e. g. , for the first half reaction: E = E°' - RT/z. F (ln [ared]/[aox]) [ared] = [aox], . . E = E°' is the mid-point potential, or standard redox potential. It is the potential at which [oxidant] = [reductant] for the half reaction. For an electron transfer: E°' = E°'(oxidant) - E°'(reductant) = E°'(e- acceptor) - E°'(e- donor) Go' = - n. F Eo'

½O 2 + NADH + H+ H 2 O + NAD+ Exercise: Consider ET of 2 electrons from NADH to oxygen: a. ½O 2 + 2 H+ + 2 e- H 2 O E°' = + 0. 815 V b. NAD+ + 2 H+ + 2 e- NADH + H+ E°' = - 0. 315 V Subtracting reaction b from reaction a: ½O 2 + NADH + H+ H 2 O + NAD+ E°' = + 1. 13 V Go' = - n. F Eo' = - 2(96485 Joules/Volt · mol)(1. 13 V) = - 218 k. J/mol
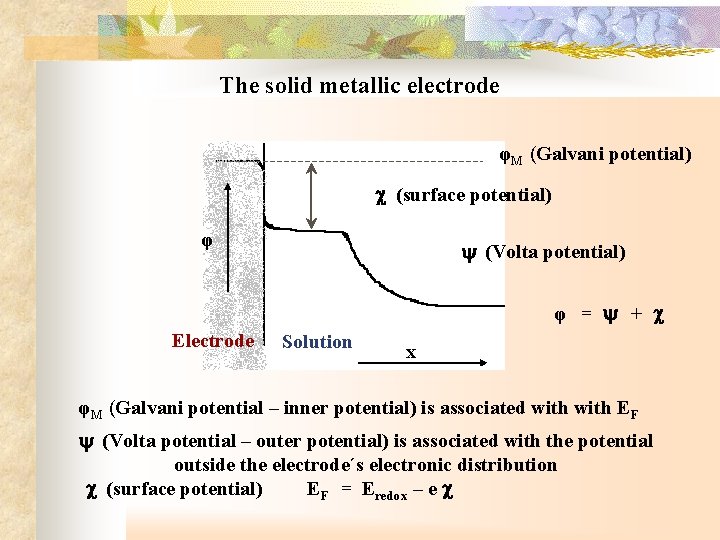
The solid metallic electrode φM (Galvani potential) (surface potential) φ (Volta potential) φ = + Electrode Solution x φM (Galvani potential – inner potential) is associated with EF (Volta potential – outer potential) is associated with the potential outside the electrode´s electronic distribution (surface potential) EF = Eredox – e

From without potential insertion on solid phase over / below Er more positive / more negative electrode polarization, overpotential = Ep - Er Ep = E polarization = Epolarizační Er = E equilibrium = Erovnovážný begins lead an electrode process As well as each electrode process, it consists of more follow steps – levels rds - rate determining step = overpotential = přepětí a most slow step

Electrode reactions steps Substance crossing from within of electrolyte to a level of maximal approximation transport (diffusion) overpotential three transport mechanisms migration – movement of ions through solution by electrostatic attraction to charged electrode diffusion – motion of a species caused by a concentration gradient convection – mechanical motion of the solution as a result of stirring or flow 2. Adsorption (localization) of ions or molecules in space of electric double layer 3. Dehydration (desolvation) absolute partial none = overpotential = přepětí

4. Chemical reactions on a metal surface, coupled with making of intermediates capable of obtaining or losing of electrons reaction overpotential 5. Electrode reaction - solitary electron crossing through interface activation overpotential 6. Adsorption of primary product of electrochemical process on a metal surface

7. Desorption of a primary product 8. Transport of product from a metal surface a) Soluble product – by diffusion (the most used style) b) Gas products – by bubbling c) Products can be integrated to an electrode crystal lattice crystalization ( nucleation) overpotential g) By diffusion to inside of electrode (for ex. amalgam)
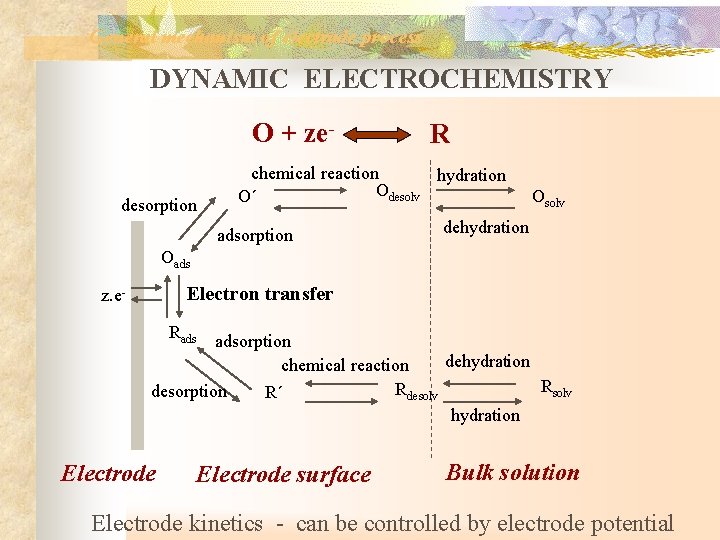
General mechanism of electrode process DYNAMIC ELECTROCHEMISTRY O + zedesorption R chemical reaction hydration Odesolv O´ adsorption Osolv dehydration Oads Electron transfer z. e- Rads adsorption dehydration chemical reaction Rsolv Rdesolv desorption R´ hydration Electrode surface Bulk solution Electrode kinetics - can be controlled by electrode potential

Velocity of electrode process Faraday : I t = N n. F = Q = for transformation of 1 mol of substance with a charge of z, charge of n. F coulomb is consumed ; F = 96484 coulomb/mol For transformation of dn mol of substance at time dt, a current I is consumed Heterogeneous reactions – velocity to surface unit: Current density Experimental dependences: = f ( j ) j = f ( ) polarization curves current-potential curves

Activation overpotential kred kox

An expression for the rate of electrode reaction Arrhenius Gibbs-Helmholtz
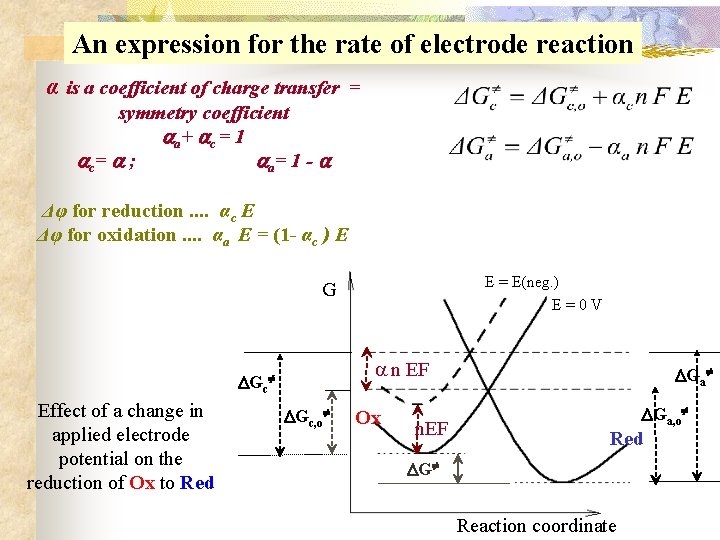
An expression for the rate of electrode reaction α is a coefficient of charge transfer = symmetry coefficient a + c = 1 c = ; a = 1 - Δφ for reduction. . αc E Δφ for oxidation. . αa E = (1 - αc ) E E = E(neg. ) E = 0 V G n EF Gc Effect of a change in applied electrode potential on the reduction of Ox to Red Gc, o Ox n. EF Ga, o Red G Reaction coordinate
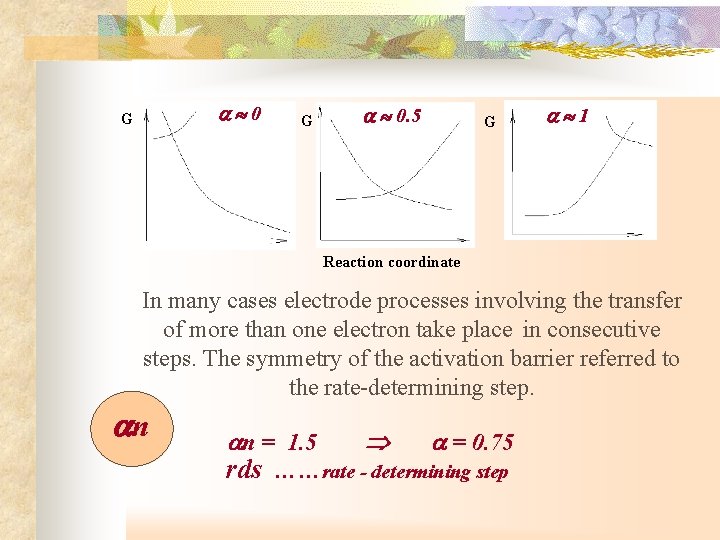
0 G G 0. 5 G 1 Reaction coordinate In many cases electrode processes involving the transfer of more than one electron take place in consecutive steps. The symmetry of the activation barrier referred to the rate-determining step. n n = 1. 5 = 0. 75 rds ……rate - determining step

Balance of electrode process vox = vred j 0 is charge current density

k 0 = standard velocity constant (members independence on E) jred = jc determination of Eeq jox = ja +

Current - overpotential crossing jox = ja jred = jc Butler- Volmer equation for electrode process, where rds is charge transfer

Butler-Volmer equation j, ka and kc depends exponentially on potential linear free energy relationship the parameters: I and E Eeq gives the exchange current Io= jo. A standard rate constant electrode as a powerful catalyst for transport the Tafel law must be corrected

the observed current is proportional to the difference between the rate of the oxidation and reduction reactions at the electrode surface concentrations of Red and Ox next to the electrode do not grow indefinitely – limited by the transport of species to electrode Id diffusion-limited current
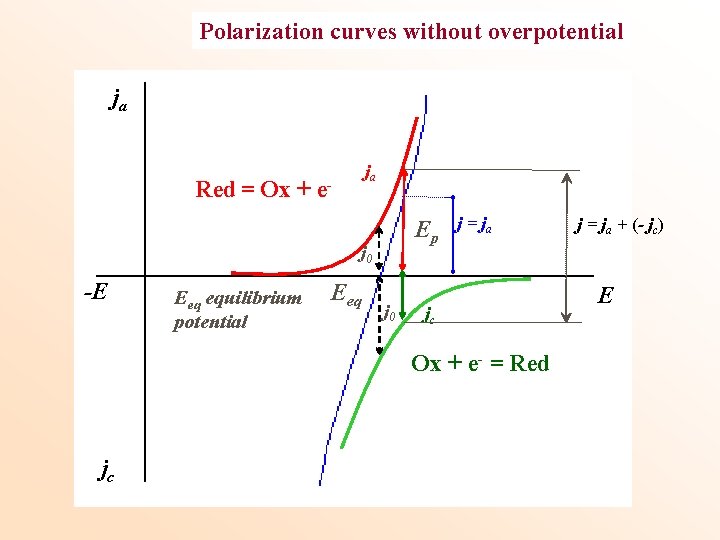
Polarization curves without overpotential ja ja Red = Ox + e- Ep j 0 -E Eeq equilibrium potential Eeq j 0 j = ja jc Ox + e- = Red jc j = ja + (- jc) E
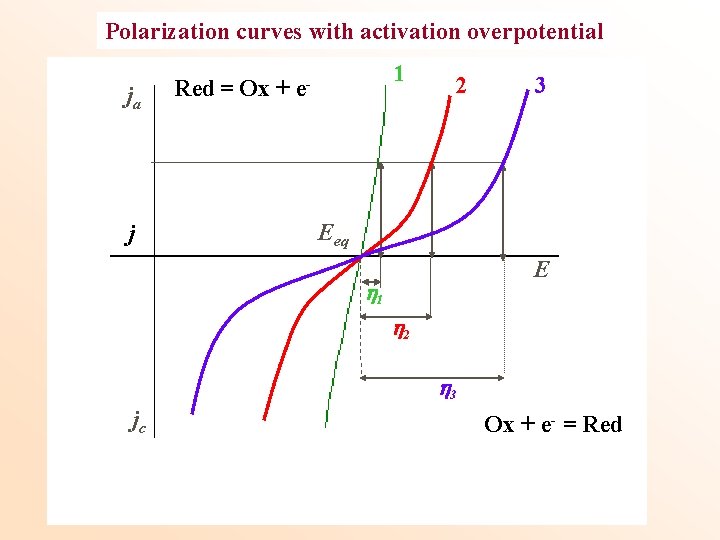
Polarization curves with activation overpotential ja j 1 Red = Ox + e- 2 3 Eeq E 1 2 3 jc Ox + e- = Red
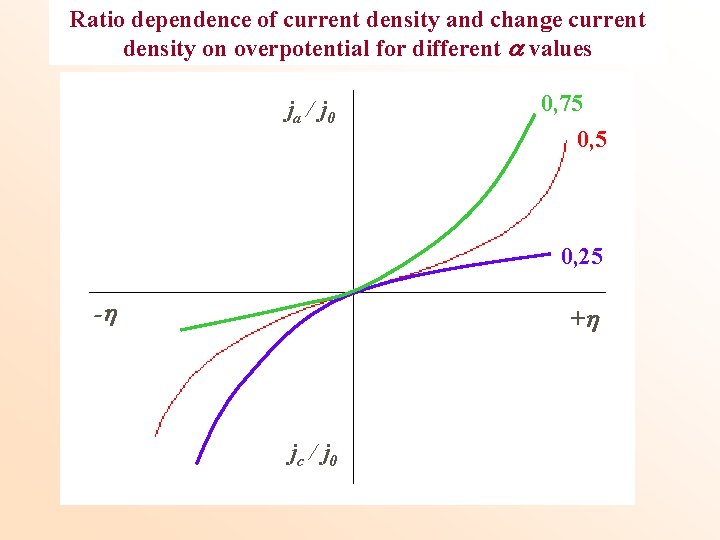
Ratio dependence of current density and change current density on overpotential for different values ja / j 0 0, 75 0, 25 - + jc / j 0

Polarization curve: = f ( j ) or I-E curves: j = f ( ) a) Small values of overpotential Development of e-x function Rp = polarization resistance b) Large values of overpotential negative process of reduction positive process of oxidation Tafel relations
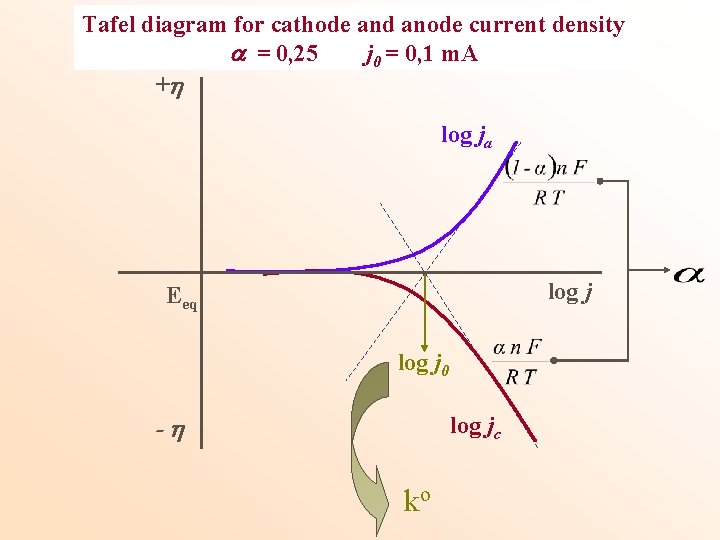
Tafel diagram for cathode and anode current density = 0, 25 j 0 = 0, 1 m. A + log ja log j Eeq log j 0 - log jc ko
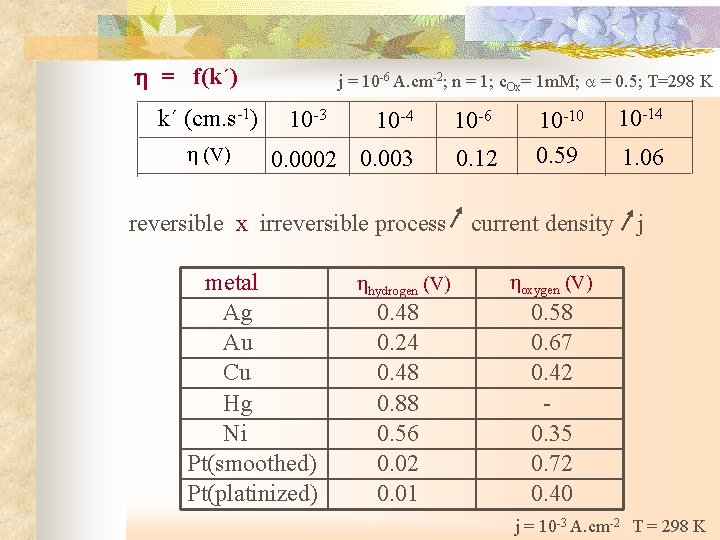
= f(k´) k´ (cm. s-1) (V) j = 10 -6 A. cm-2; n = 1; c. Ox= 1 m. M; = 0. 5; T=298 K 10 -3 10 -4 10 -6 0. 0002 0. 003 0. 12 10 -10 0. 59 10 -14 1. 06 reversible x irreversible process current density j metal Ag Au Cu Hg Ni Pt(smoothed) Pt(platinized) hydrogen (V) 0. 48 0. 24 0. 48 0. 88 0. 56 0. 02 0. 01 oxygen (V) 0. 58 0. 67 0. 42 - 0. 35 0. 72 0. 40 j = 10 -3 A. cm-2 T = 298 K

DIFFUSION: the natural movement of species in solution without the effects of the electrical field I. Fick law: the natural movement of species isolution without the effects of the electrical field …. . concentration gradient diffusion coefficient [cm 2 s-1] ; 10 -5 - 10 -6 in aqeous solutions II. Fick law: What is the variation of concentration with time ? ? ?
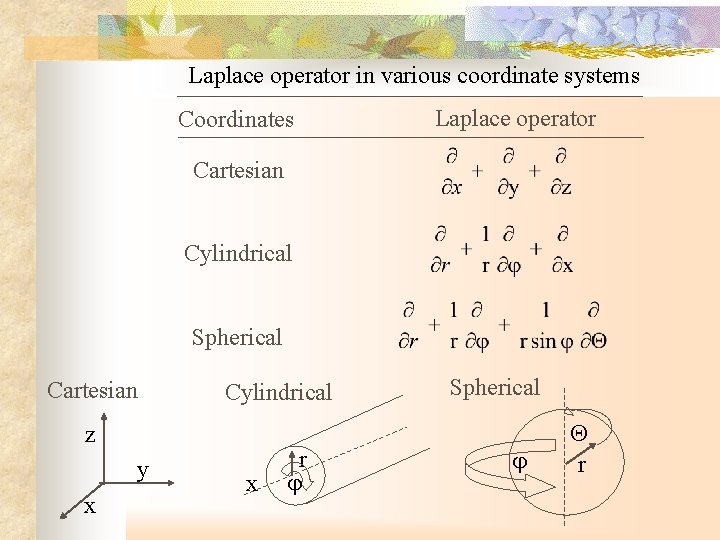
Laplace operator in various coordinate systems Coordinates Laplace operator Cartesian Cylindrical Spherical Cartesian Cylindrical z y x x r Spherical r

For any coordinate system LT for Fick´s second law under conditions of pure diffusion control Id § the potential is controlled, the current response and its variation in time is registrated chronoamperometry § the current is controlled and the variation of potential with time is registrated chronopotentiometry

Diffusion-limited current: planar and spherical electrodes Mass transport Reaction of all species reaching the electrode E t No reaction Id Potential step to obtain a diffusion-limited current of the electroactive species t = 0 planar electrode …. . semi-infinite linear diffusion Boundary conditions t = 0 c 0 = c no electrode reaction t 0 lim c = c bulk solution t 0 and c 0 = 0 diffusion –limited current Id x = 0

Cottrell equation to planar electrode linear diffusion to spherical electrode spherical diffusion

§ small t (spherical diffusion linear diffusion) § large t (the spherical diffusion dominates, which represents a steady state current) Microelectrodes -electrodes and ultra- -electrodes § small size at least one dimension 0. 1 -0. 5 m § steady state § high current density § low total current (% electrolysis is small) § interference from natural convection is negligible (supporting electrolyte)
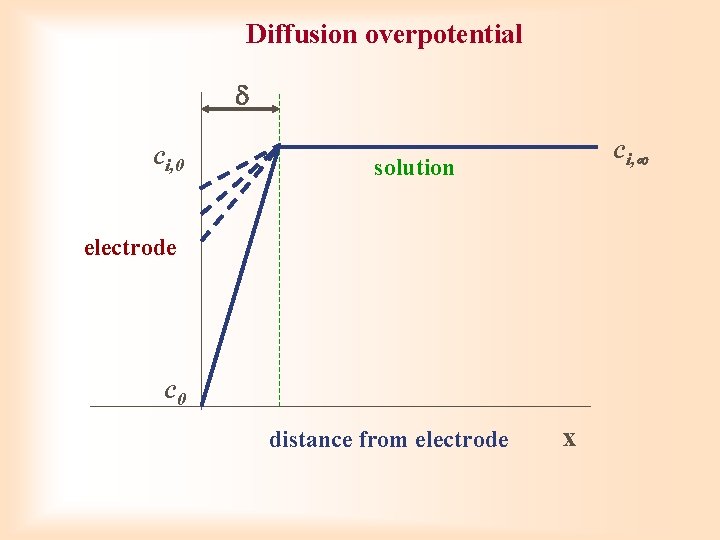
Diffusion overpotential ci, 0 ci, solution electrode c 0 distance from electrode x

Diffusion overpotential Solitary electrode process is in balance ( = 1 ai = ci) (E 0 i = standard potential) overpotential required for getting over of concentration difference

I. Fick law: Nernst diffusion layer Faraday: in stationary state

in limit: ci, 0 = 0 jk, lim= ni F D (ci/ ) Diffusion overpotential
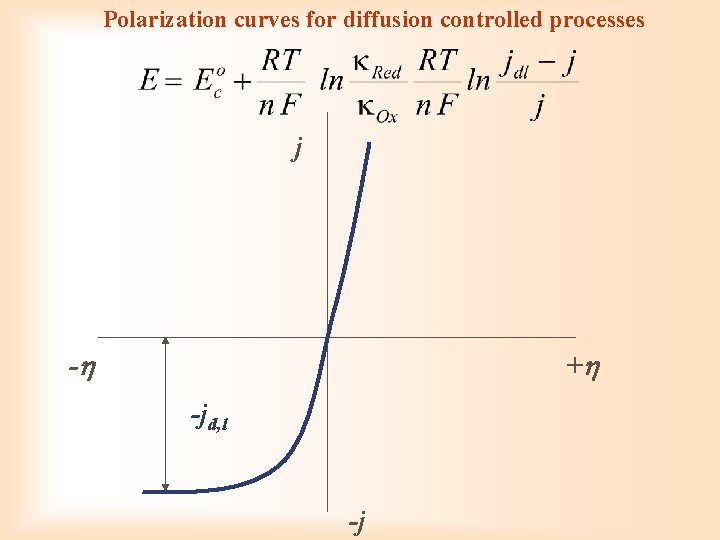
Polarization curves for diffusion controlled processes j - + -jd, l -j
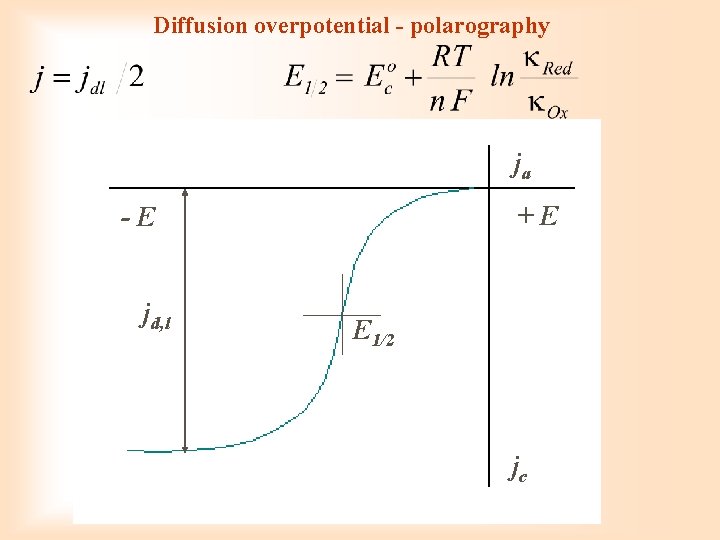
Diffusion overpotential - polarography ja +E -E jd, l E 1/2 jc
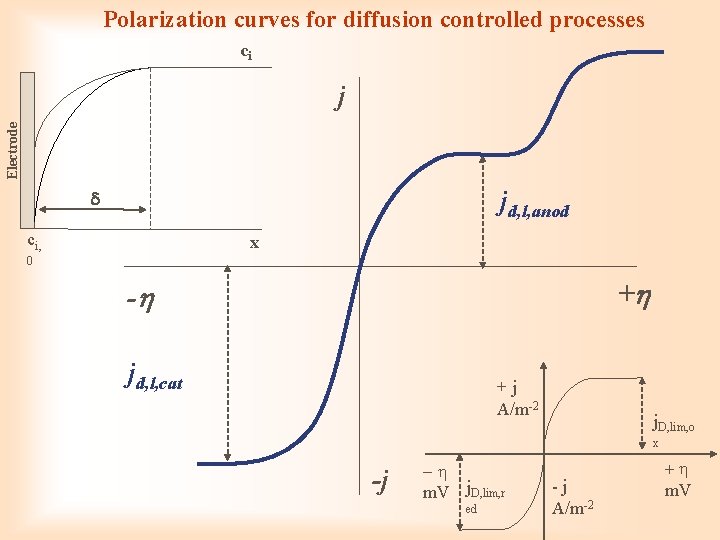
Polarization curves for diffusion controlled processes ci Electrode j jd, l, anod ci, x 0 + - jd, l, cat + j A/m-2 j. D, lim, o x -j - m. V j. D, lim, r ed - j A/m-2 + m. V
![Jaroslav Heyrovský * Dec. 20, 1890, Prague, Bohemia, Austro-Hungarian Empire [now Czech Rep. ] Jaroslav Heyrovský * Dec. 20, 1890, Prague, Bohemia, Austro-Hungarian Empire [now Czech Rep. ]](http://slidetodoc.com/presentation_image/1c144af47e41faeb5a63a1493e28222f/image-41.jpg)
Jaroslav Heyrovský * Dec. 20, 1890, Prague, Bohemia, Austro-Hungarian Empire [now Czech Rep. ] † March 27, 1967, Prague, Czechoslovakia Jaroslav Heyrovský was an inventor of the polarographic method, father of electroanalytical chemistry, recipient of the Nobel Prize (1959). His contribution to electroanalytical chemistry can not be overestimated. All voltammetry methods used now in electroanalytical chemistry originate from polarography developed by him.
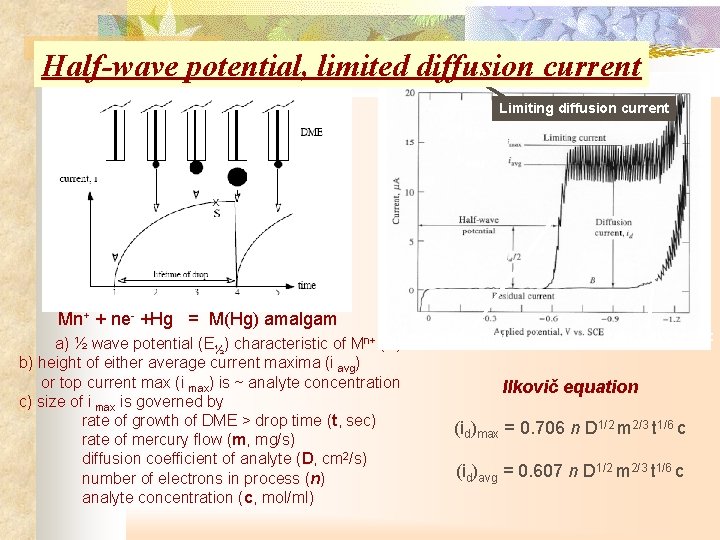
Half-wave potential, limited diffusion current Limiting diffusion current i max i avg Mn+ + ne- +Hg = M(Hg) amalgam potential Residual current a) ½ wave potential (E½) characteristic of Mn+ Half-wave (E) b) height of either average current maxima (i avg) or top current max (i max) is ~ analyte concentration Ilkovič equation c) size of i max is governed by rate of growth of DME > drop time (t, sec) (id)max = 0. 706 n D 1/2 m 2/3 t 1/6 c rate of mercury flow (m, mg/s) diffusion coefficient of analyte (D, cm 2/s) (id)avg = 0. 607 n D 1/2 m 2/3 t 1/6 c number of electrons in process (n) analyte concentration (c, mol/ml)

Instrumentation, common techniques Electrochemical analyzer AUTOLAB Autolab 20 Autolab Ecochemie Utrecht The Netherlands VA-Stand 663 Metrohm Zurich Switzerland Autolab 30

Thank you for your attention
- Slides: 44