Basic Principle of Physics II Heat and thermodynamics

Basic Principle of Physics II – Heat and thermodynamics Lecturers: Dr. F. O. Ogundare Dr. T. A. Otunla

Reversible Processes • The state of a fixed mass of an ideal gas is defined by three variables: pressure p, volume V and temperature T. • The variables are related by the equation of state: or • The gas is in equilibrium when the variables are related as given in the equation above. • A REVERSIBLE PROCESS OR CHANGE in the state of a gas is a process where the gas is taken from an equilibrium state to another through a number of small stages such that the gas is always in equilibrium at every intermediate state. The process can be reversed so as to return to its initial state via the same small stages but in the opposite direction.

Reversible Isothermal process • Reversible isothermal compression Reversible isothermal expansion p p A 0 V 1 A V 2 V 0 V 1 V 2 V

Reversible Isothermal process • The process is carried out as follows: 1. The gas is contained in a thin-walled container made of a good conducting material so that heat can easily flow out or into the gas. 2. The container is fitted with a light frictionless piston 3. The container is surrounded with a bath maintained at constant temperature 4. The compression or expansion is done by moving the piston through small steps, each step being done very slowly

Reversible Adiabatic Process • Adiabatic compression or expansion is carried out as follows: 1. The container must be thick-walled and made of insulating materials so that heat does not enter or escape from the gas 2. The light frictionless piston must be moved through small steps, each step being done very quickly. Perfect reversible change are not possible because there is no perfect conductor or perfect insulator

Heat engine This is a device that takes in energy by heat and, operating in cyclic process, expels a fraction of that energy by means of work

Heat Engine • In other words, the principle of a heat engine is to extract useful mechanical work from a machine and to replace the energy lost by heating the machine. By so doing, a certain amount of heat can be converted into work 1 st law of thermodynamics

Heat engine contd Heat engine carries some working fluid through a cyclic process during which 1. The working fluid absorbs energy by heat from a high temperature energy reservoir 2. Work is done by the engine, and 3. Energy is expelled by heat to a lower-temperature reservoir. Examples of heat engines : 1. internal combustion engine, 2. the turbines that generate electricity in power stations 3. simple steam engines
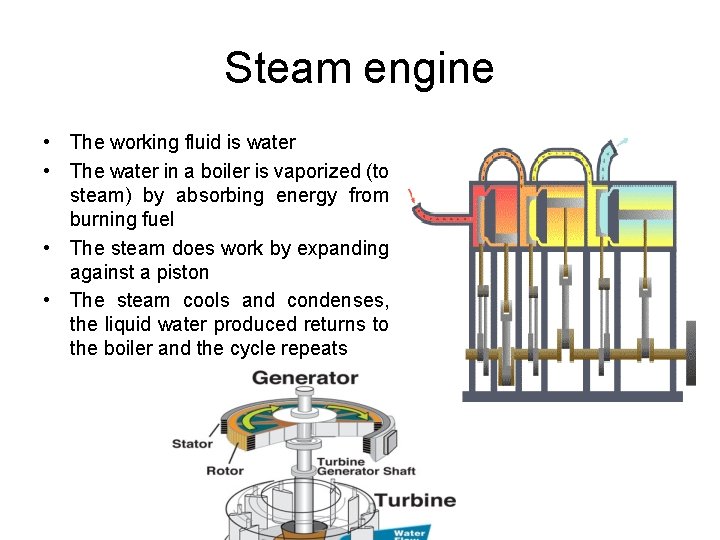
Steam engine • The working fluid is water • The water in a boiler is vaporized (to steam) by absorbing energy from burning fuel • The steam does work by expanding against a piston • The steam cools and condenses, the liquid water produced returns to the boiler and the cycle repeats

2 nd law of thermodynamics • Kelvin-Planck form of the second law states: It is impossible to construct a heat engine that, operating in a cycle, produces no other effect other than the input energy by heat from a reservoir and the performance of an equal amount of work The implication of this law is that the efficiency of heat engine is less than 100% • Thermal efficiency, e From 1 st law of thermodynamics In terms of temperature:

Heat pumps and Refrigerators • • These devices transfer energy from the cold reservoir to the hot reservoir The direction of the energy transfer is opposite to the heat engine Heat engine does work on the surrounding while work is put into the heat pumps or refrigerators for heat to be transferred from cold to hot reservoir Example of heat pump is air conditioner which transfers energy from the cool room in the house to the warm air outside • Diagram TH QH Engine W QC TC

Clausius statement of the law nd 2 • In practice, it is desirable to carry out the process involved in heat pump with minimum or no work. But this will violate the second in the form of Clausius statement: • It is impossible to construct a cyclical machine whose sole effect is to transfer energy continuously by heat from one object to another at higher temperature without the input of energy by work •

Heat pumps or refrigerator • In practice, a heat pump includes a circulating fluid that passes through two sets of metal coils that can exchange energy with the surroundings. The fluid is cold and at low pressure when it is in the coils located in a cool environment, where it absorbed energy by heat. The resulting warm fluid is then compressed and enters the other coils as hot, high pressure fluid. There it releases its stored energy to the warm surrounding. • In air conditioners, energy is absorbed into the fluid in coils located in building’s interior, after the fluid is compressed, energy leave the fluid through coils located outdoors

Heat pumps * • 4. Compressor. • Red = Gas at high pressure and temperature. • Pink = Gas at high pressure and reduced temperature. • Blue = Liquid at low pressure and greatly reduced temperature. • Light Blue = Gas at low pressure and warmer temperature.

Heat pump effectiveness • It is described using coefficient of performance (cop) • cop (cooling mode): • cop(heating mode) •

Example 1 • An ideal (carnot ) engine operates between a hot reservoir at 360 K and a cold reservoir at 270 K. It absorbs 600 J of heat per cycle at the hot reservoir. • (a) How much work does it do at each cycle? • (b) If the same engine is operated in reverse as a refrigerator, how much work must be done each cycle in order to remove

• 1200 J of heat from the cold reservoir each cycle?

Exercise 1 • A certain refrigerator has a cop of 5. 00 when the refrigerator is running, its power input is 500 W. A sample of water of 5. 00 g and temperature of 25°C is placed in the freezer compartment. How long does it take to freeze the water to ice at 0°C? Assume all other parts of the refrigerator stay at the same temperature and there is no leakage of energy from the exterior, so the operation of the refrigerator results only in energy being extracted from the water.

Carnot Engine • This is an engine operating in an ideal, reversible cycle (also known as carnot cycle) between two energy reservoir. • This ideal engine establishes an upper limit on the efficiencies of all other engines • The best way to study carnot cycle is to represent the process on a p. V diagram. The cycle consists of several individual steps assumed to be operating on an ideal gas in a frictionless piston. • The cycle consists of two reversible adiabatic processes and two reversible isothermal processes

Carnot cycle A→B Isothermal Expansion QH is absorbed through thermal contact with hot (TH) energy reservoir D→ A Adiabatic B→ C Adiabatic Compression QH =0 Thermal contact ≠ 0 Expansion QH =0 Thermal contact ≠ 0 C→D Isothermal Compression Qc is dumped through to cold (TH) energy reservoir

Carnot cycle p A QH B D TH Qc C TC VD VB VC

Process A →B (Isothermal expansion) The system starts in a state indicated by point on the p. V diagram (i. e temperature TH, pressure p. A and volume VA ) The gas is then allowed to expand to a new volume VB while remaining in thermal contact with a hot reservoir of temperature TH. This means that the gas can draw in energy as heat QH and so the expansion to a new volume VB and pressure Pa is isothermal. The work done by the gas WAB=QH and ∆U=0 p. V diagram p A QH B D TH Qc C TC VD VB VC

Process B→C Adiabatic expansion The gas is now thermal isolated from the hot reservoir by replacing the cylinder with a thermally non-conducting thickwalled cylinder. The gas expands adiabatically since no energy enters or leaves the system by heat. ∆U<0 as the energy to do work on the surrounding has to come from the internal energy of the gas. Once the temperature has dropped to TC the expansion stopped at volume VC and pressure p. C p. V diagram p A QH B D TH Qc C TC V VD VD VB VC

Process C→D Isothermal compression The gas is placed in thermal contact with the cold reservoir of temperature TC and compressed (isothermal again) until the volume and pressure are now VD and p. D respectively. During this part of the cycle work is done on the gas, but the energy gained QC is dumped to the cold reservoir so ∆U=0 • p. V diagram p A QH B D TH Qc C TC V VD VD VB VC

Process D→A Adiabatic compression • The final stage is the adiabatic compression from D→A restoring the gas back to its initial state so that it can be taken round the cycle again. The work done on the gas during this compression is equal to the work it did in moving from B →C p A QH B D TH Qc C TC V VD VD VB VC

Example 2 • The gas in the cylinder of a diesel engine can be considered to undergo a cycle of changes of pressure, volume and temperature. One such cycle, for an ideal gas, is shown on the graph P/Pa 16 x 106 B C 7. 9 x 106 D 1. 0 x 106 A 1. 4 x 10 -4 6. 0 x 10 -4 10. 0 x 10 -4 V/m 3

(i). The temperature of the gas at A and B are 300 K and 660 K respectively. Use the ideal gas equation and data from the graph to find the temperature at C and D. (ii). During each of the four sections of the cycle, changes are being made to the internal energy of the gas. Some of the factor affecting these changes are given in the table below Section of cycle Heat supplied to Work done on gas/J A to B 0 -300 B to C 2580 740 C to D 0 440 D to A -1700 Internal energy of gas/J Find the work done on the gas in section D to A Deduce the values of the ‘increase in internal energy of the gas’ for each section and list them (iii). What is the net work out put during a complete cycle? (iv). Calculate the efficiency of this machine

Example 3 (i). A quantity of 0. 220 mol of air enters a diesel engine at a pressure of 1. 04 x 105 Pa and temperature of 297 K. Assuming that air behaves as an ideal gas find the volume of this quantity of air (ii). The air is then compressed to one twentieth of this volume the pressure having risen to 6. 8 x 106 Pa. Find the new temperature. (iii). Heating of the air then takes place by burning a small quantity of fuel in it to supply 6150 J. This is done at a constant pressure of 6. 09 x 106 Pa as the volume of air increases and the temperature rises to 2040 K Find 1. The molar heat capacity of air at constant pressure 2. The volume of air after burning the fuel 3. The work done by the air during this expansion 4. The change in internal energy of air during this expansion

Third law of thermodynamics • This law does not really fit into the pattern establishes by the others • Zeroth law-establishes the existence of thermal equilibrium and allows temperature to be defined • 1 st law establishes that work and heat help us to increase the internal energy of an object • 2 nd law establishes that the efficiency of heat engines cannot be 100% • 3 rd law states that objects can never be cooled down to absolute zero (i. e -273 K).

3 rd law • In order to cool something down energy must be drained from it into a colder object. • The task of reducing the temperature become increasingly difficult as the temperature approaches absolute zero. • The task reducing temperature to absolute temperature has led to many wonderful discoveries • Superconductivity: resistivity of some materials drops to zero at temperatures of a few Kelvin • Superfluidity: Helium-3 cools to a liquid that flows with no viscosity. •

Thermal Conductivity • Thermal conduction in crystalline solid is due to lattice vibration. • In metals- in addition to lattice vibration there is conduction through diffusion of free electrons. • Silver is the best conductor of heat, follow by copper, aluminum and iron among common metals • Wood, paper, cork and polystyrene are poor conductor or good insulator • Air is a good insulator. • A metal knob of a door feels colder to the touch than the wooden door. Why? • Since the two objects are in thermal equilibrium, there temperature must be the same. • However, metal knob feels colder to touch because of it conducts heat away from the hand quicker than wood, an insulator.

Thermal conductivity • If a metal rod is placed with one end kept at a high constant temperature (Thot) and the other end kept in contact with another object at a lower temperature (Tcold) Thot Tcold • Under steady state condition, the rate of flow of heat through each section of the rod depends on (1). The temperature gradient, which is the change in temperature per unit distance along the bar (2). The cross-sectional area A- A larger A will allow greater heat flow (3). The material of the rod Temperature gradient Rate of flow -ve sign denotes that the temperature is decreasing along the direction of heat flow is constant of proportionality known as thermal conductivity

Thermal conductivity • • • Thermal conductivity of a material is the rate of flow of heat through a unit cross-sectional area per unit temperature gradient OR It can also be defined as the rate of flow of heat through two opposite faces of a cube of the material of length 1 m when the temperature difference between the two faces is 1 K Examples: (1) A lagged copper has a uniform cross-sectional area of 1. 0 cm 2 and length 20. 0 cm. When steady state is attained, the temperature of one end of the rod is 120°C and other is 0°C. Find (a). The temperature gradient, (b). Rate of heat flow and (c). Temperature at a point 8 cm from the hot end ( λ=300 W/m/K). (2) Window glass has a thermal conductivity of 0. 8 W/m/K. Calculate the rate at which heat is conducted through a window of area 2. 0 m 2 and thickness 4. 0 mm if the temperature in an air-conditioned room is 20°C and the outdoors temperature is 35°C.

Thermal resistance • Both thermal and electrical conductions in metals are due to motion of free electrons. • On the macroscopic scale there is a lot of similarity between the two. Thermal conduction Electrical conduction Heat charge Temperature Electric potential Temperature difference Potential difference Thermal resistance Electrical resistance Rate of flow= temp. diff/ thermal resistance Rate of flow= Potential diff. / electrical resistance • Rate of flow

Thermal resistance • Thermal conductors in series: when two or more conductors are joined in series so that heat flows from one to the next, the rate of heat flow through all the conductors are the same • If the temperature between the ends are Thot and Tcold and there cross-sectional areas are the same • Rate of heat flow=

Examples 1. The external wall of a brick house has an area of 16 m 2 and thickness 0. 3 m. The temperature inside and outside the house are 20°C and 0° C respectively. (a). Calculate the rate of heat loss through the wall (b). What is the rate of heat loss when the inside surface of the wall is covered with a layer of expanded polystyrene of thickness 20 mm? (c). What is the temperature at the interface between the brick wall and the expanded polystyrene. (thermal conductivity of brick=0. 5 W/m/K; expanded polystyrene=0. 03 W/m/K). 2. Three metal rods, one copper, another aluminum and third brass are joined in series. The length of each rod is 60 cm and diameter is 1. 0 cm. The free end of the copper rod is maintained at 100°C and the brass rod at 0°C. If the rods are lagged and thermal conductivity of copper is twice that of aluminum and four times that of brass, find the equilibrium temperature at the copper-aluminium and aluminum-brass interface.

Radiation • • • Nuclear radiation: α, β and γ emissions from unstable nuclei Visible radiation: emission of visible wavelength of EM waves Thermal radiation: emissions of infrared wavelength of EM waves Thermal radiation is a method of transfer of heat as conduction and convection. Conduction and convection require material medium Radiation is a method of energy transfer that does not require material medium. Thermal radiation is emitted by all objects whose temperature is above 0 K. A wide spectrum of wavelengths are included The higher the temperature the shorter the wavelength

Thermal radiation detectors • Human skin • A thermometer with the bulb blackened with lamp soot • A bolometer: consists of narrow strip of platinum with the surface blackened. Thermal radiation makes the temperature of the platinum to rise. This is accompany with the corresponding rise in the resistance. It is measured by an electrical circuit. • A thermopile: consists of a number of thermocouples joined in series. The hot junction is blackened and exposed to thermal radiation. The cold junction is shielded from radiation. The thermo-emf produced is measured using a millvoltmeter. •

Absorption and Emission of Radiation • Thermal radiation are partly absorbed or reflected by the surface of any object. This depends on the nature of the surface. • Rule of thumb: • An object will radiate(absorb) more energy than it absorbs(emits) if the temperature is greater than (less than) the temperature of its surrounding. • Good radiators and good absorbers tend to be black with rough surface • Bad radiators and bad absorbers tend to be light colour (white), shiny, and with smooth surfaces.

Perfect thermal sources(Black bodies) • An object that is capable of emitting or absorbing radiation of every wavelength is a perfect thermal source. • The spectrum of radiation produced by black bodies depend on temperature. E X 10 -9 Wm-3 T = 1 500 K T = 1200 K T = 1000 K 0 Λ x 106 m

Wien’s law • The wavelength (λmax. ) at which the intensity is at a peak decreases as temperature decreases and it is related to the temperature T of black body • The constant k is known as wien’s constant = 2. 898 x 10 -3 m. K

Stefan’s law • The area under each of the curves above represents total radiant energy emitted per second per area of the black body and it is given as: Where

RADIANT ENERGY As black bodies radiate energy( ), they also absorbs EM radiation from the surrounding( )otherwise their temperatures will drop to absolute zero. Suppose an object is at temperature T and Its surrounding is at temperature To The net power radiated:

Example • A human being has a surface area of 1. 9 m 2 and a body temperature of 37°C. What is the total power radiated and the peak wavelength, assuming the human to be a perfect thermal source? (assume the surroundings to be at a temperature of 20°C)
- Slides: 44