Basic Organic Chemistry CHM 207 Objective In this

Basic Organic Chemistry CHM 207

Objective • In this chapter, we have to understand the structure and bonding in organic compounds. • Able to give electrons configurations of different atoms. • Able to identify types of hybridization of different compounds.

Today’s Topic I. Introduction to Organic Chemistry II. Structures and bondings in organic compounds i. Electron configurations ii. Covalent bond formation and hybridization iii. Electronegativities of elements

I. Introduction to Organic Chemistry • Organic chemistry is the study of carbon compounds from living or non-living things. • Organic molecules are found in all aspects of life such as food, cosmetic, pharmaceuticals, environment, household products, textiles and others. • Organic chemistry refers to a branch of chemistry concerned with study of the compounds of carbon, excluding carbonate (CO 32 -); cyanide (CN-) bicarbonate (HCO 3 -), carbon dioxide and carbon monoxide.
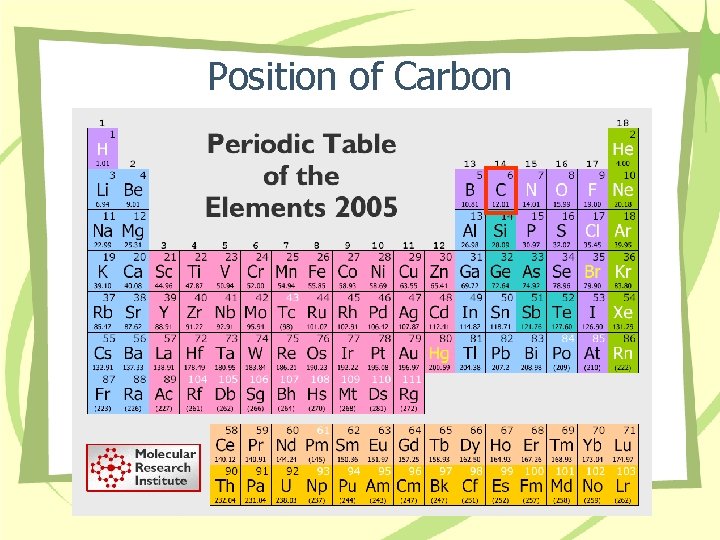
Position of Carbon

II. Structures and bondings in organic compounds
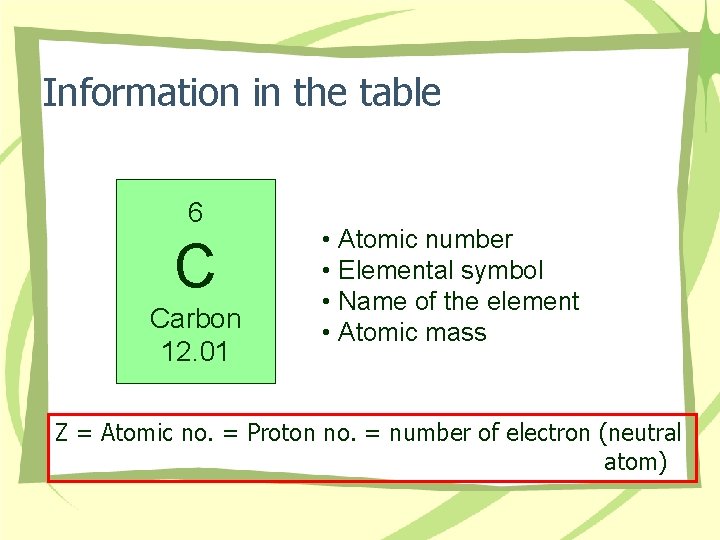
Information in the table 6 C Carbon 12. 01 • Atomic number • Elemental symbol • Name of the element • Atomic mass Z = Atomic no. = Proton no. = number of electron (neutral atom)

i. Electron configurations • Definition : description of the orbitals its electrons occupy. Element C Z 6 Electron configuration 1 s 22 p 2

Electron configurations-cont • Ground-state electron configuration – the electron configuration of lowest energy for an atom, molecule or ion. • The ground-state electron configuration is determine by using THREE rules.

Rule 1 Aufbau principle Orbitals fill in order of increasing energy, from lowest to highest. Rule 2 Pauli exclusion principle No more than two electrons may be present in an orbital. If two electrons are present, their spins will be paired. Rule 3 Hund’s rule When orbitals of equivalent energy are available but there are not enough electrons to fill all of them completely, then one electron is added to each equivalent orbital before a second electron is added to any one of them.
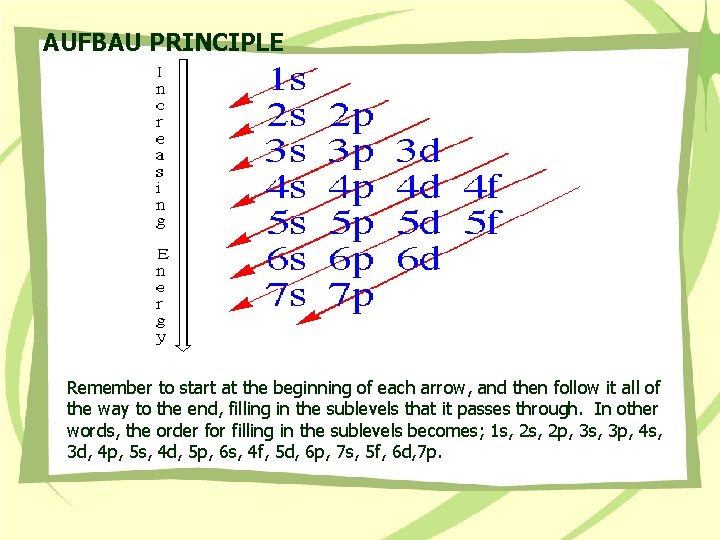
AUFBAU PRINCIPLE Remember to start at the beginning of each arrow, and then follow it all of the way to the end, filling in the sublevels that it passes through. In other words, the order for filling in the sublevels becomes; 1 s, 2 p, 3 s, 3 p, 4 s, 3 d, 4 p, 5 s, 4 d, 5 p, 6 s, 4 f, 5 d, 6 p, 7 s, 5 f, 6 d, 7 p.

Remember !!! Orbital s can be occupy with 2 electrons Orbital p can be occupy with 6 electrons





Activity • Gives the electron configurations and draw the orbital diagram of: a) O b) Cl c) C d) F

ii. Covalent bond formation and hybridization • In the valence bond theory, a covalent bond is formed when two atoms approach each other until their atomic orbitals (each containing one unpaired electrons) overlap with one another. For example: -

Bonding of Carbon Atoms • The covalent bonds present in organic compounds involve a number of hybrid orbitals such as sp 3 , sp 2 and sp.
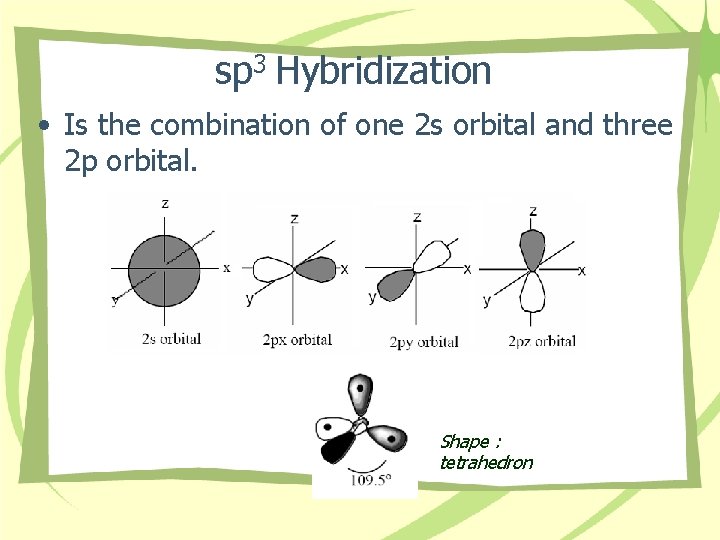
sp 3 Hybridization • Is the combination of one 2 s orbital and three 2 p orbital. Shape : tetrahedron
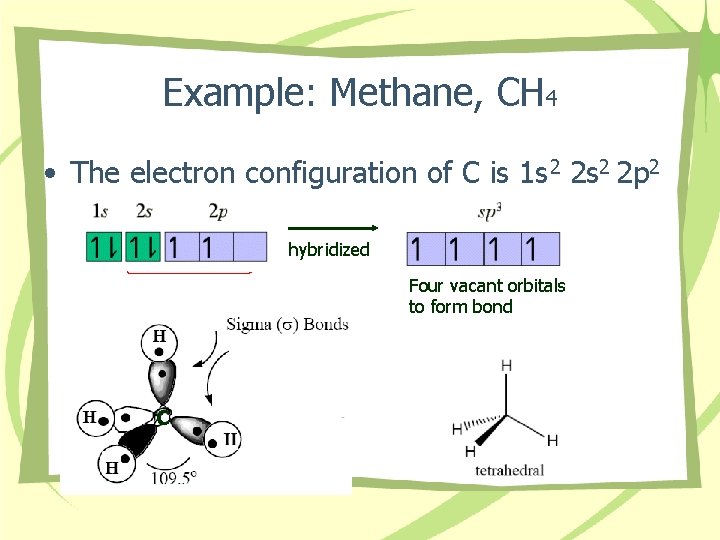
Example: Methane, CH 4 • The electron configuration of C is 1 s 2 2 p 2 hybridized Four vacant orbitals to form bond C
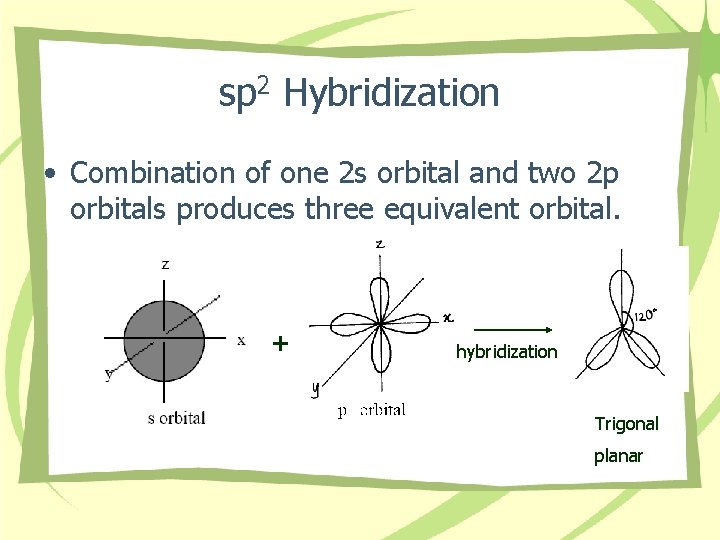
sp 2 Hybridization • Combination of one 2 s orbital and two 2 p orbitals produces three equivalent orbital. + hybridization Trigonal planar
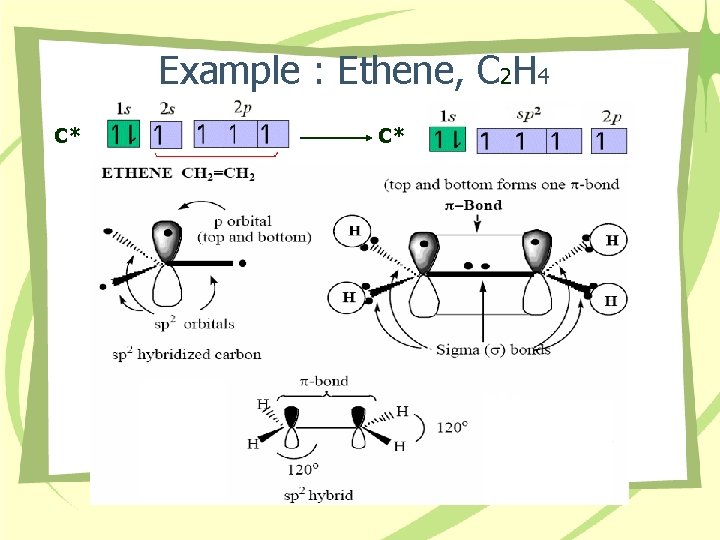
Example : Ethene, C 2 H 4 C* C*
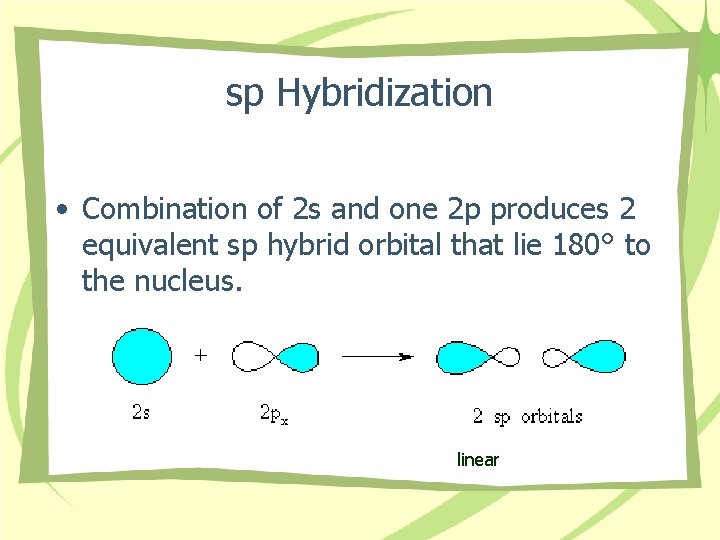
sp Hybridization • Combination of 2 s and one 2 p produces 2 equivalent sp hybrid orbital that lie 180° to the nucleus. linear
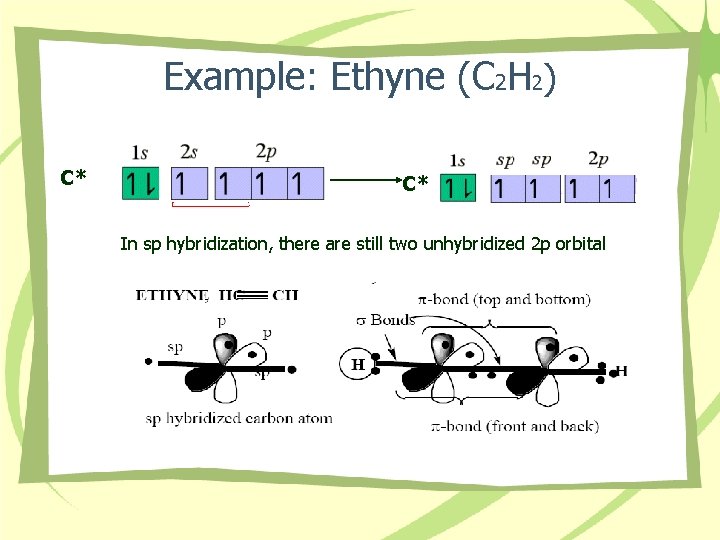
Example: Ethyne (C 2 H 2) C* C* In sp hybridization, there are still two unhybridized 2 p orbital

Questions • Determine types of hybridized orbital for each carbon in these compounds: – A) CH 3 COOH – B) CH 3 CH 2 OH – C) CH 3 C≡CCH 2 COCH 3 – D) CH 3 COOH – E) CH 3 CH=CH 2

iii. Electronegativities of elements • Electronegativity measure the ability of an atom to attract to itself the electron pair forming a covalent bond. • Among the main group elements, electronegativity increases moving from left to right in periodic table and decrease moving down a group. • Bond polarity is due to differences in electronegativities. H- H Non-polar C-H C-F polar covalent bond more polar The greater the differences in electronegativity the more polar the bond polarity.
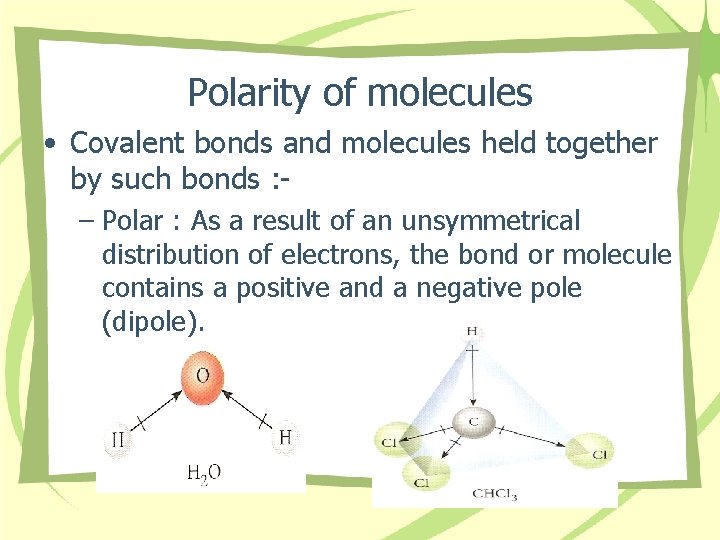
Polarity of molecules • Covalent bonds and molecules held together by such bonds : – Polar : As a result of an unsymmetrical distribution of electrons, the bond or molecule contains a positive and a negative pole (dipole).
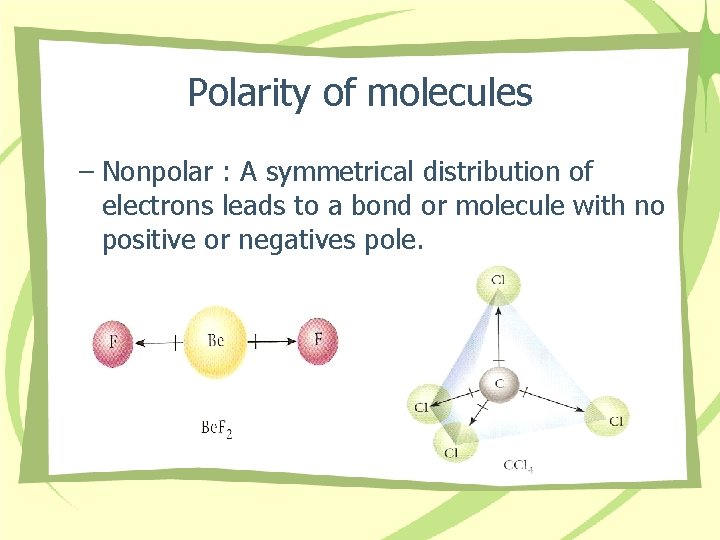
Polarity of molecules – Nonpolar : A symmetrical distribution of electrons leads to a bond or molecule with no positive or negatives pole.

References: • Ahmad Kamal, A. A. J. et. al. 2007. Chemistry for matriculation 2. 2 nd edition. 84 -106. Oriental academic publication. • Peter, Y. 2006. Organic and inorganic chemistry the complete text and guide. Arah pendidikan. • Masterton, W. and Hurley, C. N. 2001. Chemistry, Principle and reactions. 4 th edition. Harcourt College Publisher.

- Slides: 31