Basic HIV Course for Health Professionals Session 5

Basic HIV Course for Health Professionals Session 5: Antiretroviral Therapy

Learning Objectives By the end of this session participants should be able to: • Describe the process of starting antiretroviral (ARV) medication in adults • Describe ARV mechanism of action • Explain ARV side effects • Describe ARV drug-drug interactions

Introduction: Overview of ART • Antiretroviral therapy = ART = drugs to treat HIV • Goals of ART include: • • Maximum and long-lasting suppression of viral load Restoration and/or preservation of immune function Improvement of quality of life Reduction of HIV-related morbidity (OIs) and mortality

Preparation for ART Initiation (1) • All HIV positive patients are eligible for same day ART, regardless of CD 4 count and clinical staging • Preparation for initiation should include: • • TB screening Clinical staging and CD 4 count Screening and management of STIs Counselling This Photo by Unknown Author is licensed under CC BY-NC-SA

Preparation for ART Initiation (2) • Support for disclosure and partner notification • Screening and management of comorbidities • Repeat CD 4 testing and WHO clinical staging 6 -monthly in those not ready to begin ART • Annual pap smear for all HIVpositive women

Preparation for ART Initiation (3) • Educate him/her on benefits of treatment and possible side-effects • Consider nutritional status of patient, co-morbidities and possible drug-interactions, and address any mental health and substance abuse issues
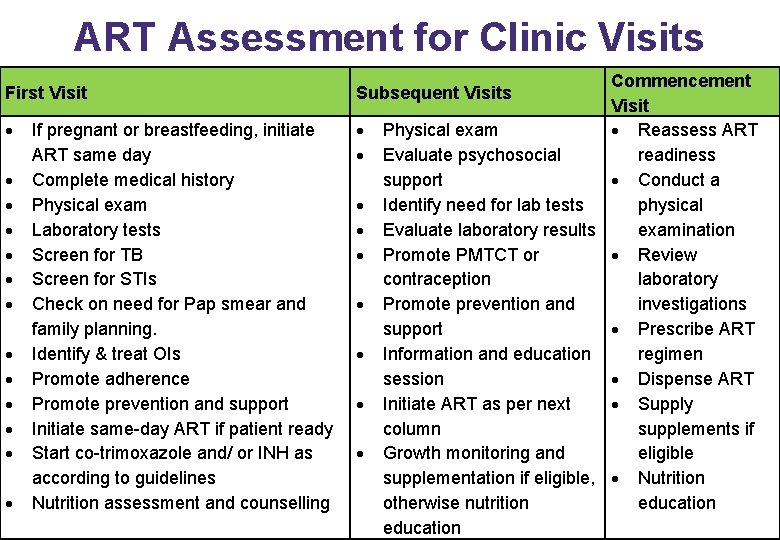
ART Assessment for Clinic Visits Commencement Visit Physical exam Reassess ART Evaluate psychosocial readiness support Conduct a Identify need for lab tests physical Evaluate laboratory results examination Promote PMTCT or Review contraception laboratory Promote prevention and investigations support Prescribe ART Information and education regimen session Dispense ART Initiate ART as per next Supply column supplements if Growth monitoring and eligible supplementation if eligible, Nutrition otherwise nutrition education First Visit Subsequent Visits If pregnant or breastfeeding, initiate ART same day Complete medical history Physical exam Laboratory tests Screen for TB Screen for STIs Check on need for Pap smear and family planning. Identify & treat OIs Promote adherence Promote prevention and support Initiate same-day ART if patient ready Start co-trimoxazole and/ or INH as according to guidelines Nutrition assessment and counselling

Timing of ART Initiation ART should be started as soon as the patient is ready and within 2 weeks of CD 4 count being done. Immediate • All HIV-positive pregnant or breastfeeding women, with no Priority active TB or contradiction provide DTG and EFV based regimen and their implication for this and subsequent pregnancies Fast Track • Should be done with those patients who are: Initiation • HIV stage 4 • Patients with CD 4 ≤ 200 cells Co. Infection with TB • Start TB treatment first, followed by ART as soon as possible and within 8 weeks • If CD 4 <50 cells, then initiate ART within 2 weeks after starting TB treatment, when patient’s symptoms are improving and TB treatment is tolerated. • If CD 4 >50 cells, then initiate ART within 2 -8 weeks after
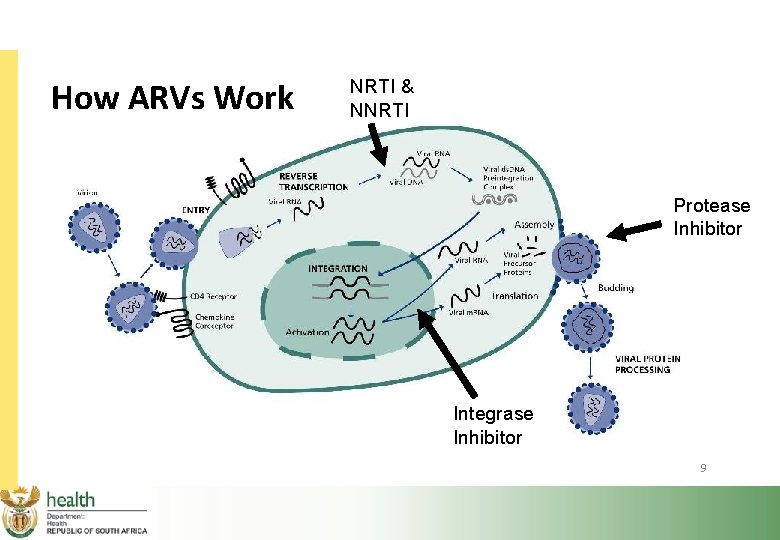
How ARVs Work NRTI & NNRTI Protease Inhibitor Integrase Inhibitor 9
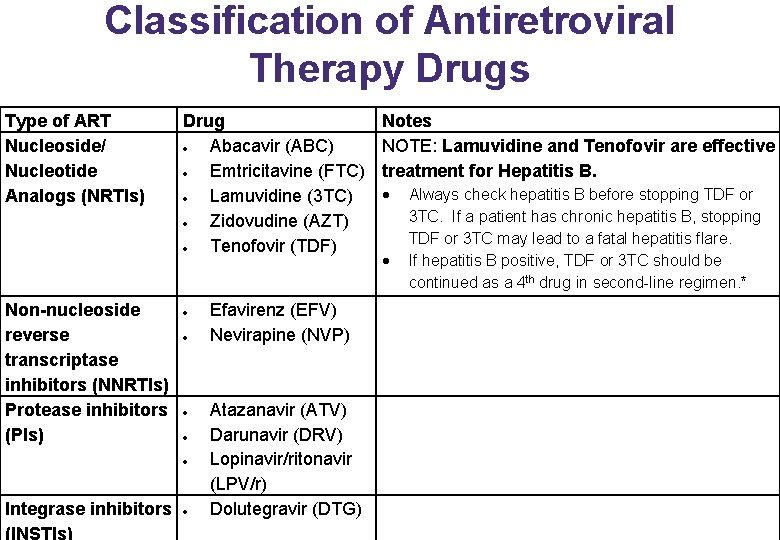
Classification of Antiretroviral Therapy Drugs Type of ART Nucleoside/ Nucleotide Analogs (NRTIs) Non-nucleoside reverse transcriptase inhibitors (NNRTIs) Protease inhibitors (PIs) Drug Abacavir (ABC) Emtricitavine (FTC) Lamuvidine (3 TC) Zidovudine (AZT) Tenofovir (TDF) Integrase inhibitors (INSTIs) Efavirenz (EFV) Nevirapine (NVP) Atazanavir (ATV) Darunavir (DRV) Lopinavir/ritonavir (LPV/r) Dolutegravir (DTG) Notes NOTE: Lamuvidine and Tenofovir are effective treatment for Hepatitis B. Always check hepatitis B before stopping TDF or 3 TC. If a patient has chronic hepatitis B, stopping TDF or 3 TC may lead to a fatal hepatitis flare. If hepatitis B positive, TDF or 3 TC should be continued as a 4 th drug in second-line regimen. *

Dolutegravir Why are all the excitement about DTG? • TDF, 3 TC, and DTG (TLD) is a potent ART regimen • Superior efficacy and faster rate of viral suppression • Well tolerated • High genetic barrier to resistance • No drug interactions with contraception

DTG: Dosages and Formulations • Class of ARV: Integrase Inhibitor (In. STI) • Standard Dose: DTG 50 mg daily • Who may get DTG? • Children, adolescents and adults > 20 kg: • Formulations: • DTG 50 mg tablet > 20 kg • children and adolescents 20 - 35 kg • Fixed-dose combination: TLD • Tenofovir (TDF) 300 mg + lamivudine (3 TC) 300 mg + DTG 50 mg (TLD). • TLD can be prescribed for patients > 35 kg and > 10 years of age (restricted by TDF). • DTG dose with concomitant TB Treatment: • Double DTG dose to 50 mg 12 -hourly. • If on TLD FDC, add DTG 50 mg 12 hours after TLD dose
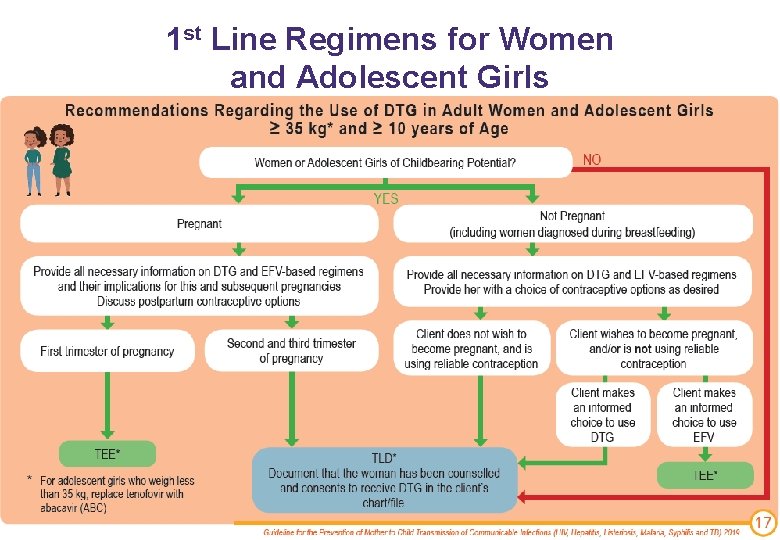
1 st Line Regimens for Women and Adolescent Girls

Switching between EFV and DTG in stable patients Overarching principle: Never change only one drug in a failing regimen! A single drug switch to DTG requires a VL of < 50 c/m. L in last 6 months Also remember to: o Counsel patients on risks and benefits of DTG vs EFV, and the risk for NTDs in subsequent pregnancies in WOCP o Provide counselling on contraception post-partum to WOCP o Check for potential drug interactions o Warn the client about new side effects that may be experienced when switching to a new drug o Offer patient a choice of remaining on EFV or switching to DTG

Second Line Regimen for Late Adolescents and Adults Second line regimen: adolescents ≥ 15 years and adults First line virological failure Drugs Comments Failing on TDF/ 3 TC or FTC/NVP or EFV Switch to AZT/3 TC* + DTG Failing on AZT/3 TC/NVP or EFV Switch to TLD Dyslipidaemia (total cholesterol >6 mmol/L) or diarrhoea associated with LPV/r If non-adherent, address causes of non-adherence If the VL >1000 copies/m. L at any point, intensify adherence and repeat VL in 2 months If VL remains at >1000 copies/m. L after 2 months, then switch to second line regimen * All patients switching off Switch LPV/r to ATV/r TDF should have their Hepatitis B status checked

Case Studies: Determining When and What to Start Cases in Adults

ART Jeopardy Game!

Immune Reconstitution Inflammatory Syndrome (IRIS) IRIS is: “A spectrum of clinical signs and symptoms resulting from [body’s] restored ability to mount an inflammatory response associated with immune recovery” WHO, 2006

Risk Factors for IRIS • Initiating ART within close proximity of diagnosis of an OI • Decrease in plasma HIV RNA (viral load) level • Low baseline CD 4 (<50) prior to starting ART • Being ART naïve at time of diagnosis of OI

IRIS Presentation • Early IRIS (during first 3 months of ART) • Late IRIS (months to years after starting ART) – Results from an immune response against viable opportunistic pathogens, often present subclinically – Incidence of IRIS peaks in first several months of therapy – Results from an immune response against antigens of non-viable opportunistic pathogens – Cases of late IRIS have been detected even after 1 or 2 years of therapy

Features of IRIS • Usually occurs one week to several months after starting ART • Associated with: • low initial CD 4 counts (<100 cells/mm 3) • rapidly increased CD 4 count and reduced viral load once started on ART

Presentation of IRIS • • Fever Lymphadenopathy/lymphadenitis Abscesses Pneumonia CNS disease Hepatitis Dermatological Manifestations

Treatment of IRIS • Treat underlying active infection • If already on ART, continue ART, unless inflammatory response is life-threatening • Most studies support early initiation of ART during treatment of an active OI • Prolonged delays in ART initiation should be avoided to prevent morbidity and mortality from untreated HIV

Timing of ART • To prevent IRIS, start treatment for active infections prior to starting ART, including: • Tuberculosis • Cryptococcal Meningitis • CMV Retinitis

Drug Interactions • Can result in life-threatening emergencies, or can actually have a benefit • Occur when one drug affects how another drug is used in the body (metabolized, absorbed, etc. ) • Herbs and traditional remedies can also cause interactions

How Drug Interactions May Occur • Absorption inhibition • Enzyme inhibition or induction • Additive or antagonistic side effects (pharmacodynamics effects)
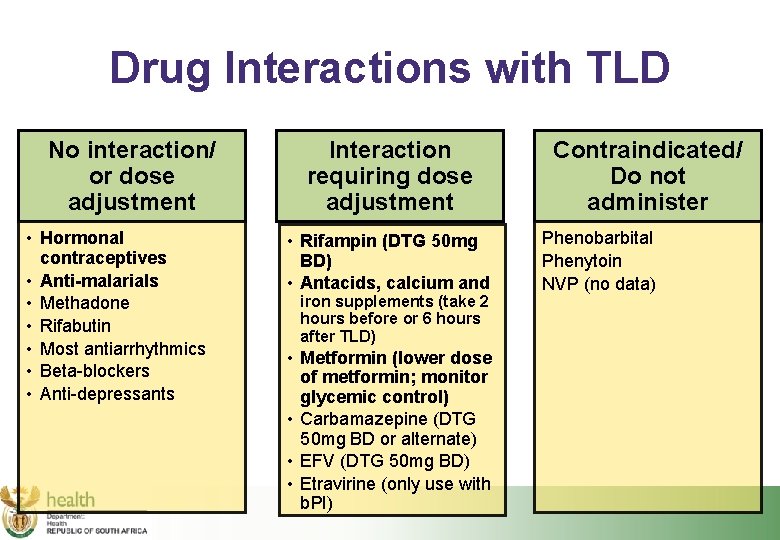
Drug Interactions with TLD No interaction/ or dose adjustment • Hormonal contraceptives • Anti-malarials • Methadone • Rifabutin • Most antiarrhythmics • Beta-blockers • Anti-depressants Interaction requiring dose adjustment • Rifampin (DTG 50 mg BD) • Antacids, calcium and iron supplements (take 2 hours before or 6 hours after TLD) • Metformin (lower dose of metformin; monitor glycemic control) • Carbamazepine (DTG 50 mg BD or alternate) • EFV (DTG 50 mg BD) • Etravirine (only use with b. PI) Contraindicated/ Do not administer Phenobarbital Phenytoin NVP (no data)

Worksheet: Recognising Common Drug Interactions and Drugs to Avoid

Treatment Goals ARV goal: My first Viral Load will be suppressed! And thereafter remain below 400 copies/m. L TB goal: I have completed 6 months TB treatment and I am cured of TB Hypertension My Blood Pressure is less than 140/90 mm. Hg goal: Diabetes goal: I monitor and keep my blood glucose within (FPG) 4 -7 mmol/L
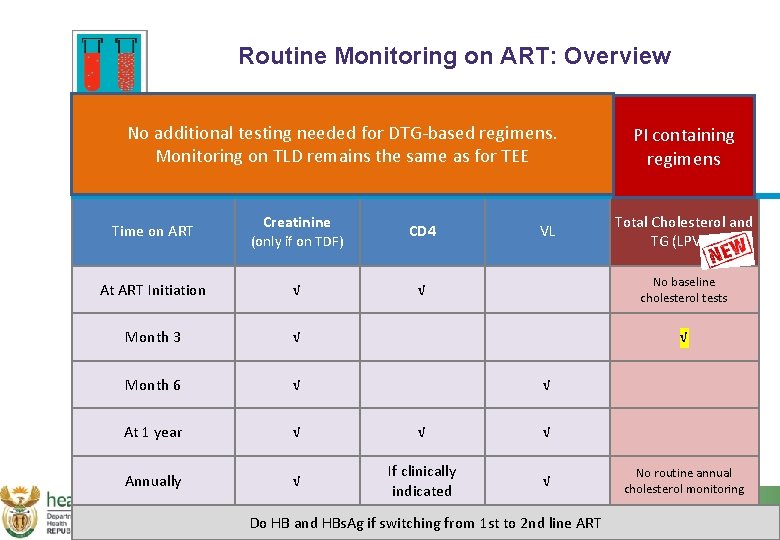
Routine Monitoring on ART: Overview No additional testing needed for DTG-based regimens. Monitoring on TLD remains the same as for TEE Time on ART Creatinine (only if on TDF) CD 4 VL PI containing regimens Total Cholesterol and TG (LPV/r) No baseline cholesterol tests At ART Initiation √ √ Month 3 √ Month 6 √ At 1 year √ √ √ Annually √ If clinically indicated √ √ √ Do HB and HBs. Ag if switching from 1 st to 2 nd line ART No routine annual cholesterol monitoring
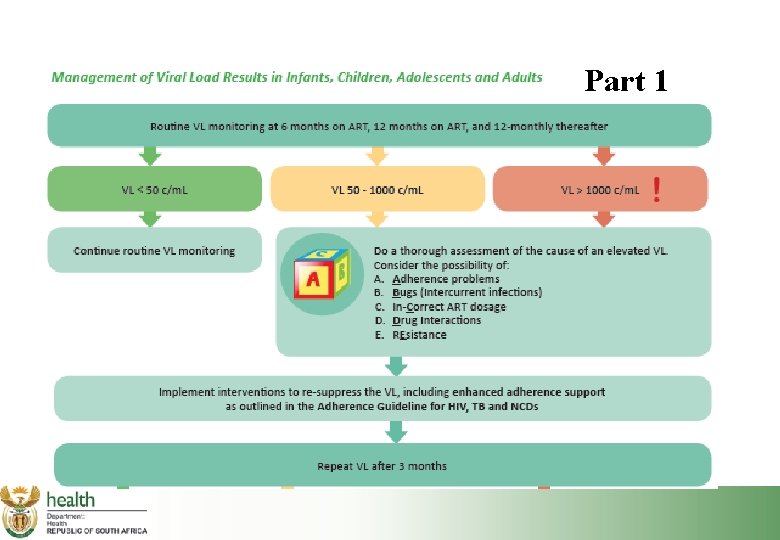
Part 1


Clinical and Laboratory Monitoring of Patient on First Line HIV Treatment Refer to page 65 of your participant manual to see the chart with this information

Any Questions? Thank you!
- Slides: 34