Basic Environmental Technology Water Supply Waste Management and

Basic Environmental Technology Water Supply, Waste Management, and Pollution Control SIXTH EDITION CHAPTER 4 Water Quality Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved

FIGURE 4 -1 a Portable water testing kits are used in the field to measure chemical and physical quality. (Courtesy of HACH Company. ) Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
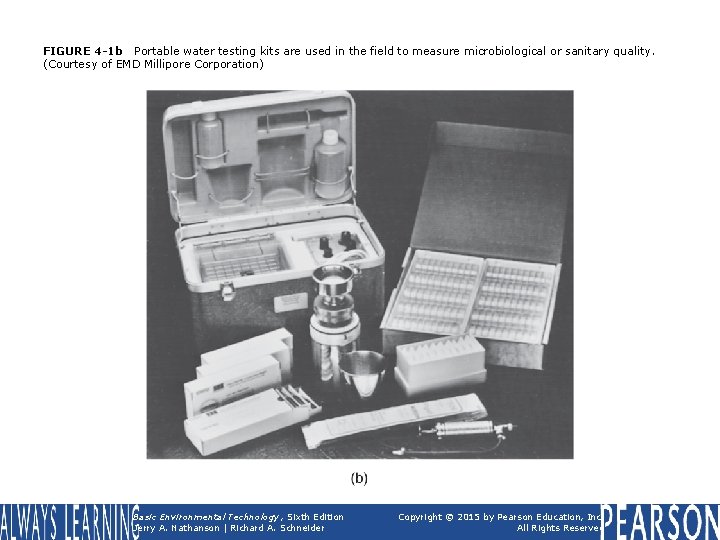
FIGURE 4 -1 b Portable water testing kits are used in the field to measure microbiological or sanitary quality. (Courtesy of EMD Millipore Corporation) Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
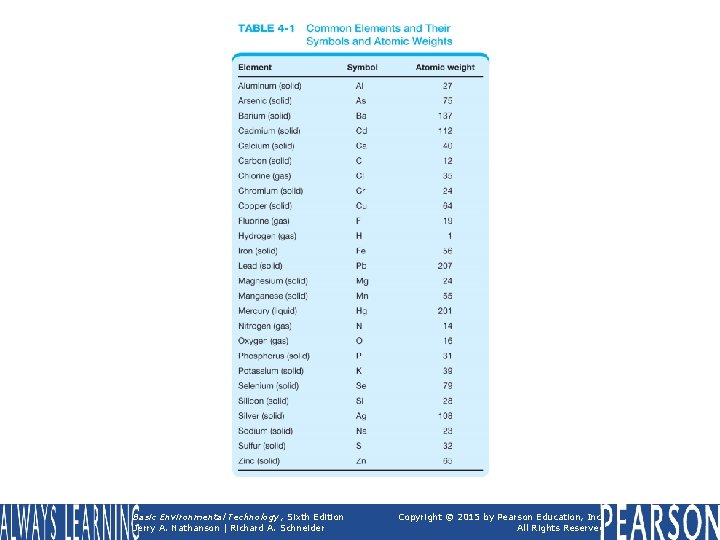
TABLE 4 -1 Common Elements and Their Symbols and Atomic Weights Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
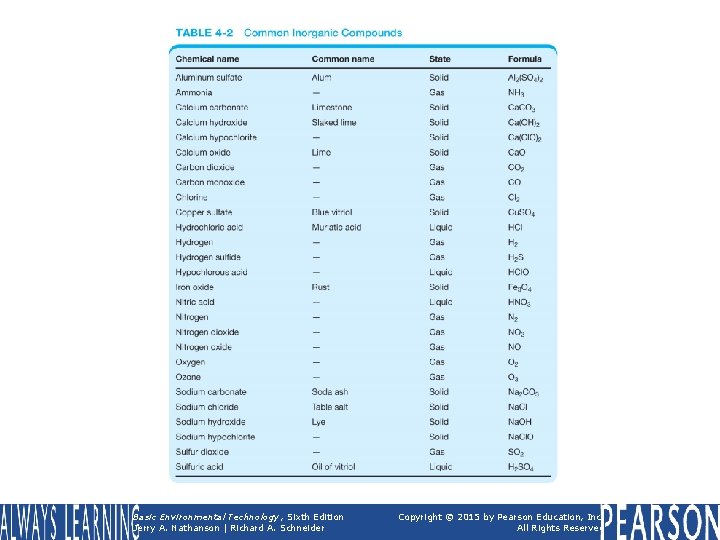
TABLE 4 -2 Common Inorganic Compounds Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
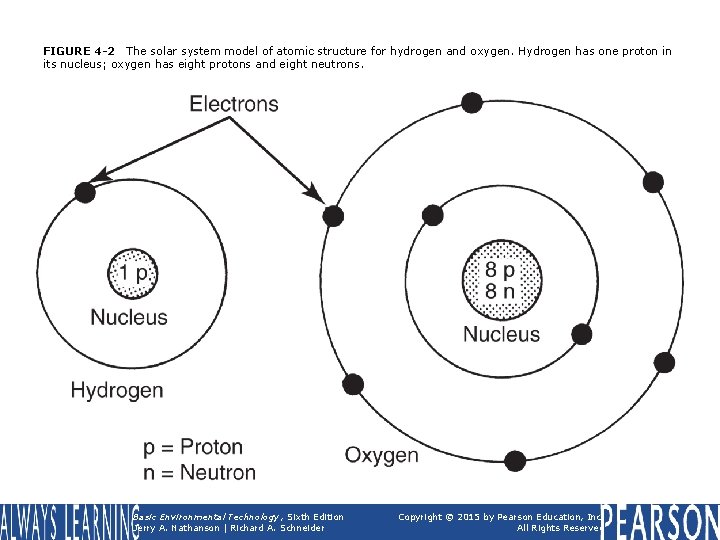
FIGURE 4 -2 The solar system model of atomic structure for hydrogen and oxygen. Hydrogen has one proton in its nucleus; oxygen has eight protons and eight neutrons. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
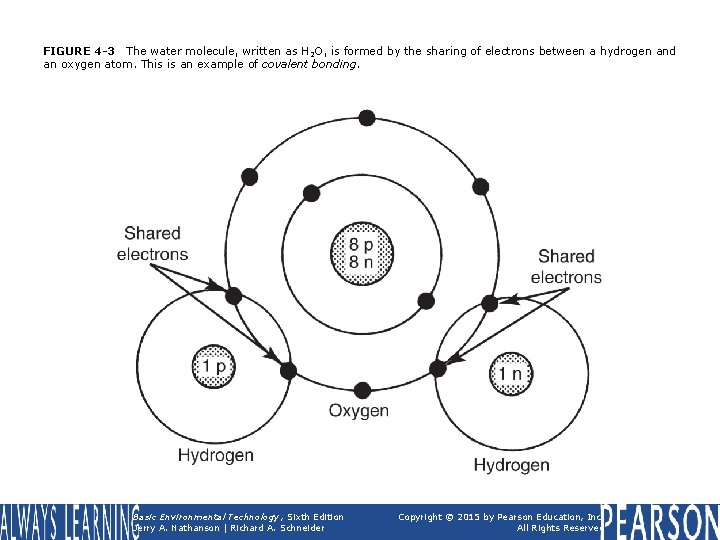
FIGURE 4 -3 The water molecule, written as H 2 O, is formed by the sharing of electrons between a hydrogen and an oxygen atom. This is an example of covalent bonding. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
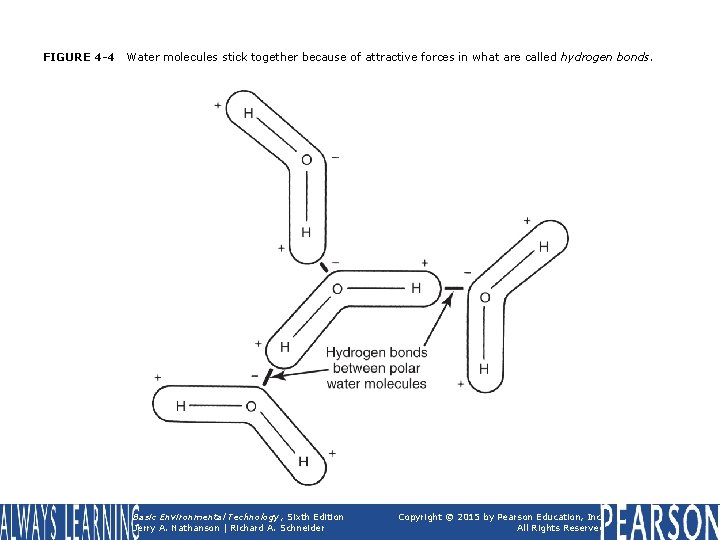
FIGURE 4 -4 Water molecules stick together because of attractive forces in what are called hydrogen bonds. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
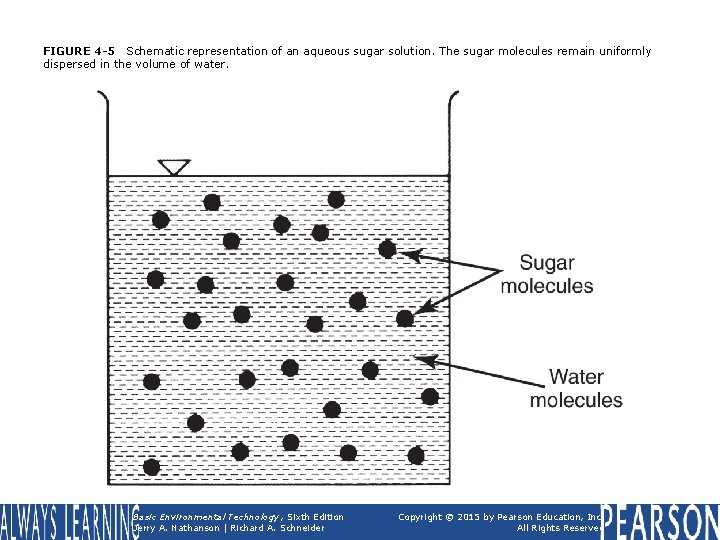
FIGURE 4 -5 Schematic representation of an aqueous sugar solution. The sugar molecules remain uniformly dispersed in the volume of water. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
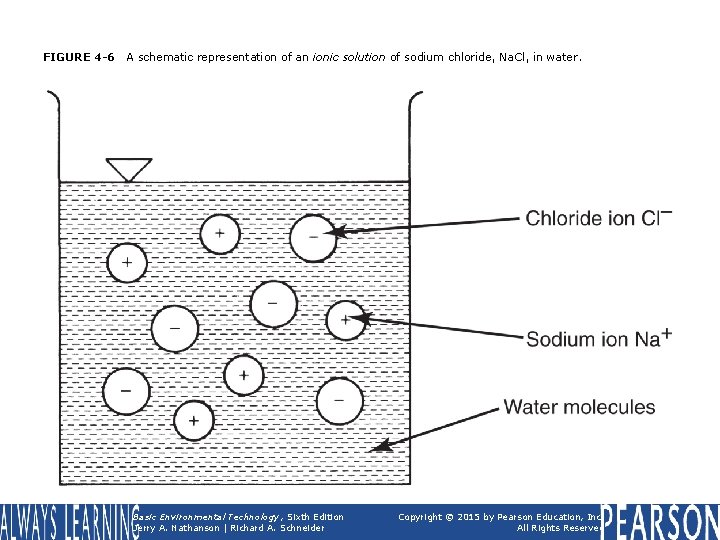
FIGURE 4 -6 A schematic representation of an ionic solution of sodium chloride, Na. Cl, in water. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
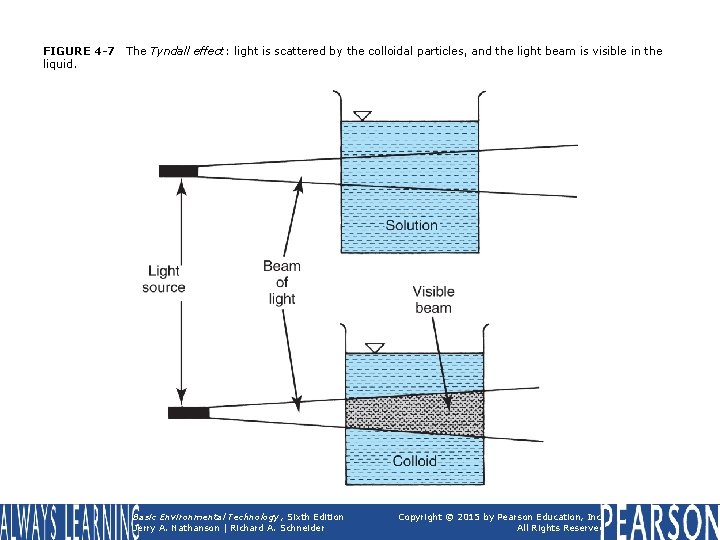
FIGURE 4 -7 liquid. The Tyndall effect: light is scattered by the colloidal particles, and the light beam is visible in the Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
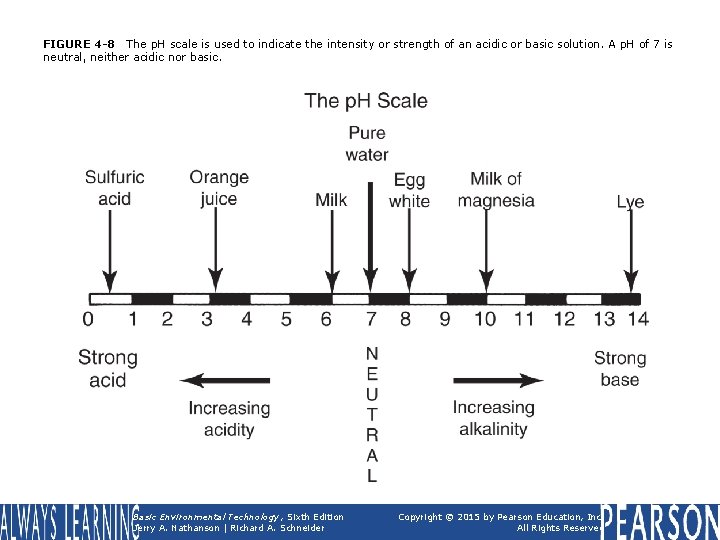
FIGURE 4 -8 The p. H scale is used to indicate the intensity or strength of an acidic or basic solution. A p. H of 7 is neutral, neither acidic nor basic. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
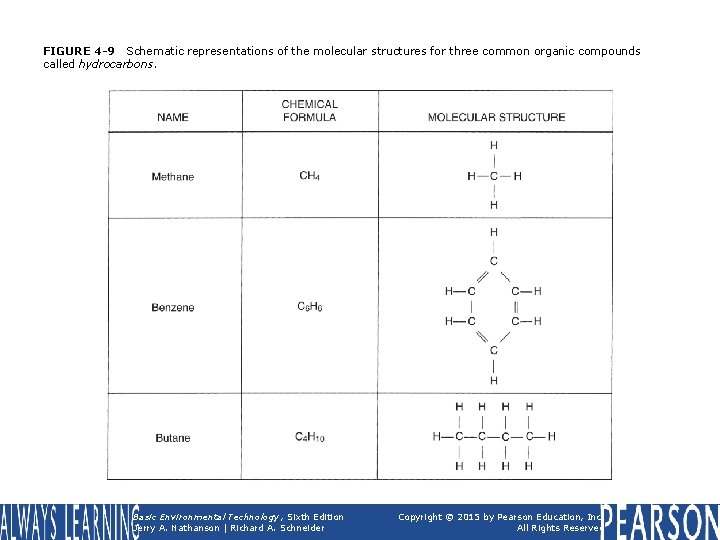
FIGURE 4 -9 Schematic representations of the molecular structures for three common organic compounds called hydrocarbons. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
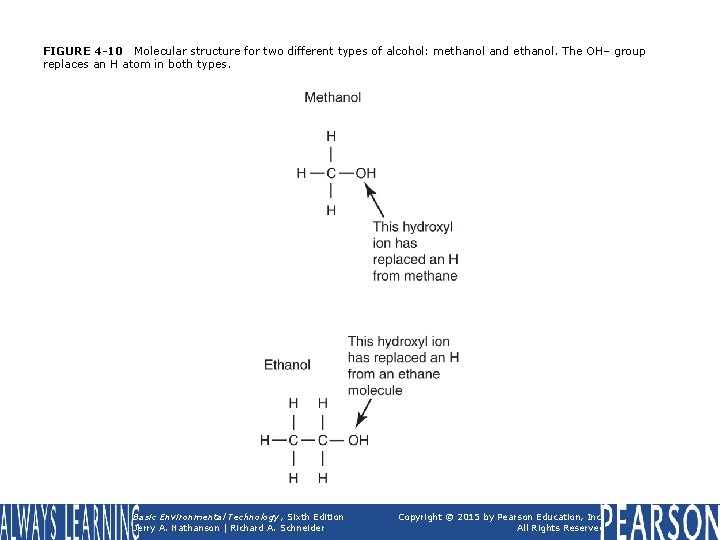
FIGURE 4 -10 Molecular structure for two different types of alcohol: methanol and ethanol. The OH– group replaces an H atom in both types. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
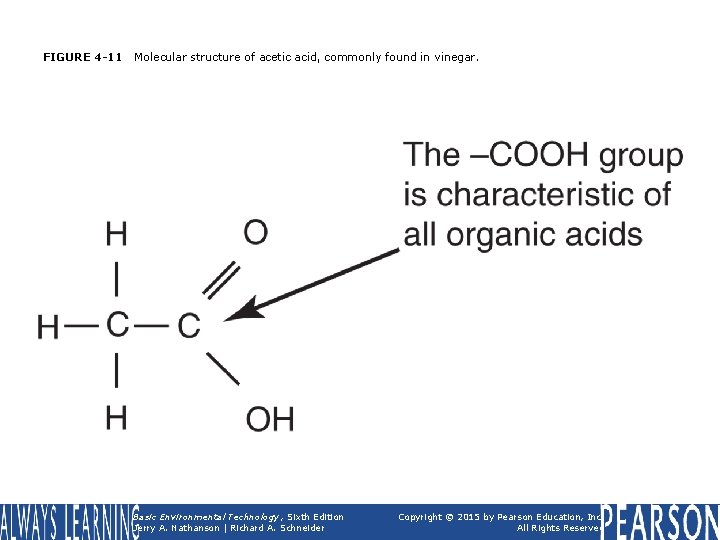
FIGURE 4 -11 Molecular structure of acetic acid, commonly found in vinegar. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
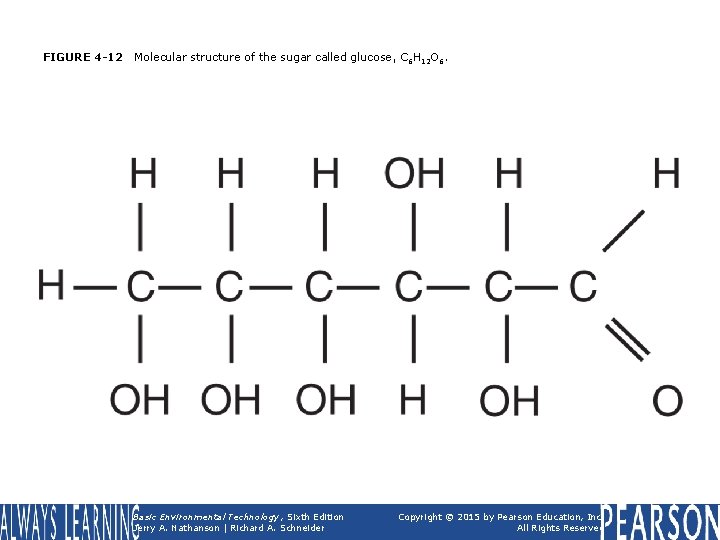
FIGURE 4 -12 Molecular structure of the sugar called glucose, C 6 H 12 O 6. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved

FIGURE 4 -13 Mass balance sketch. The total mass of salt entering the tank on the left must equal the total mass of salt leaving the tank on the right (1 input = output 2). If the salt concentrations and volume flow rates in pipes 1 and 2 are known, the concentration of salt in pipe 3 can be calculated using a mass balance equation. Note that Q 3 = Q 1 + Q 2. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
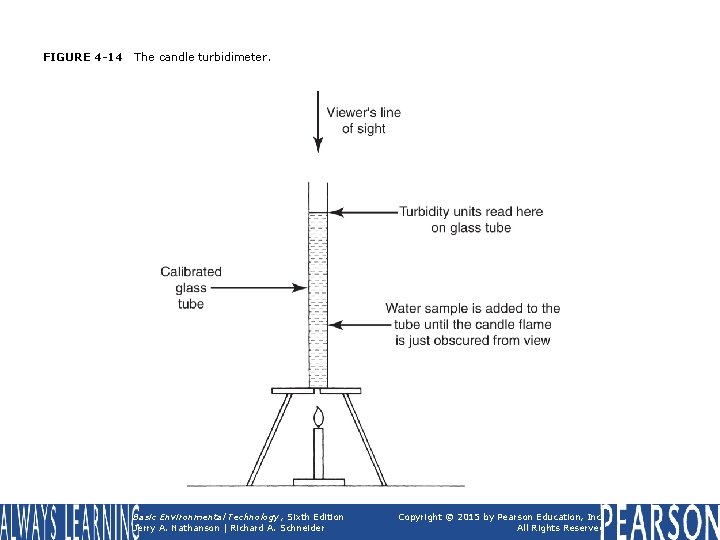
FIGURE 4 -14 The candle turbidimeter. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved

TABLE 4 -3 Solubility of Oxygen in Freshwater at Standard Atmospheric Pressure Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
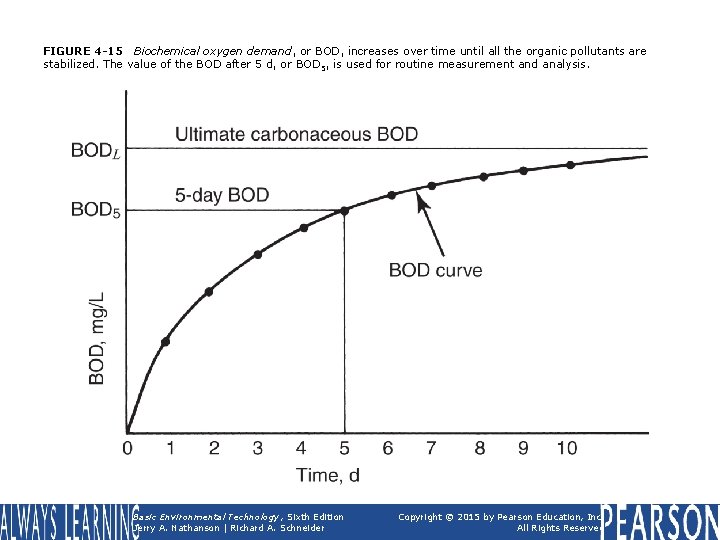
FIGURE 4 -15 Biochemical oxygen demand, or BOD, increases over time until all the organic pollutants are stabilized. The value of the BOD after 5 d, or BOD 5, is used for routine measurement and analysis. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
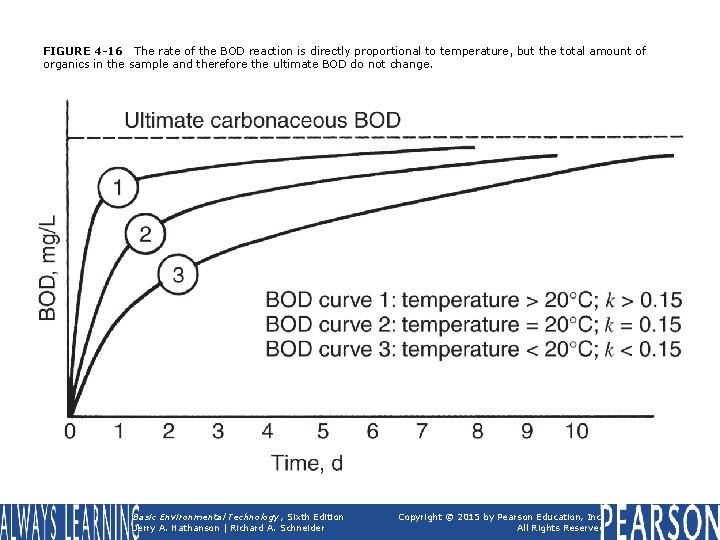
FIGURE 4 -16 The rate of the BOD reaction is directly proportional to temperature, but the total amount of organics in the sample and therefore the ultimate BOD do not change. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
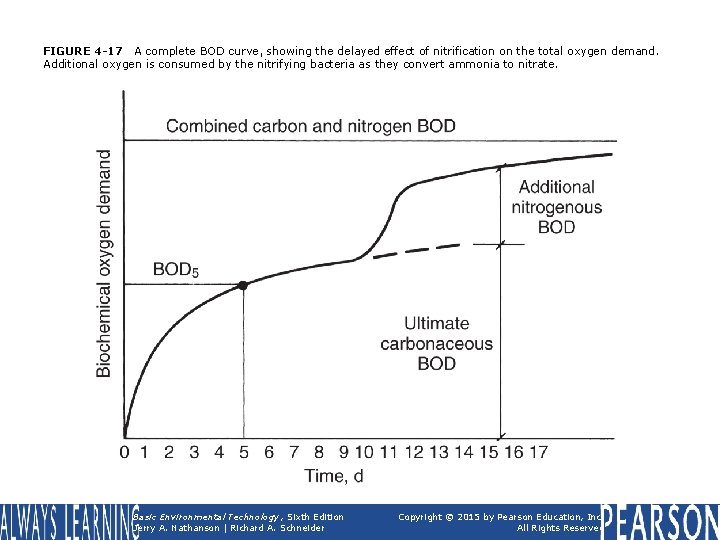
FIGURE 4 -17 A complete BOD curve, showing the delayed effect of nitrification on the total oxygen demand. Additional oxygen is consumed by the nitrifying bacteria as they convert ammonia to nitrate. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
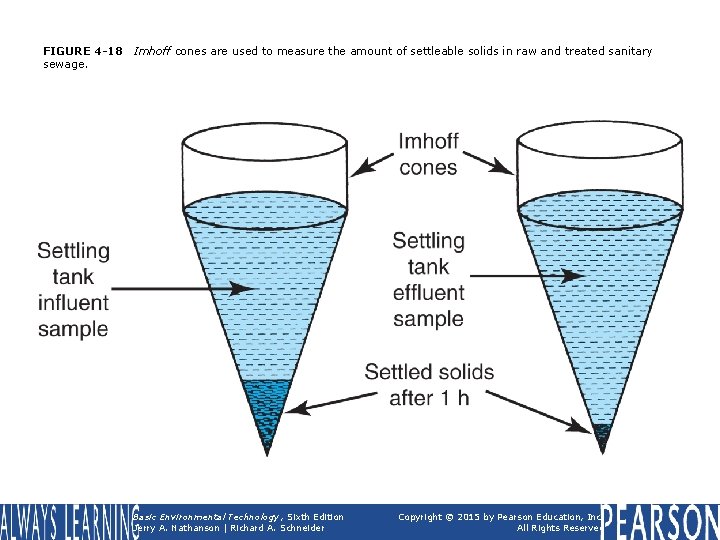
FIGURE 4 -18 sewage. Imhoff cones are used to measure the amount of settleable solids in raw and treated sanitary Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved

FIGURE 4 -19 A color comparator test kit for measuring chlorine residual. (Courtesy of HACH). Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
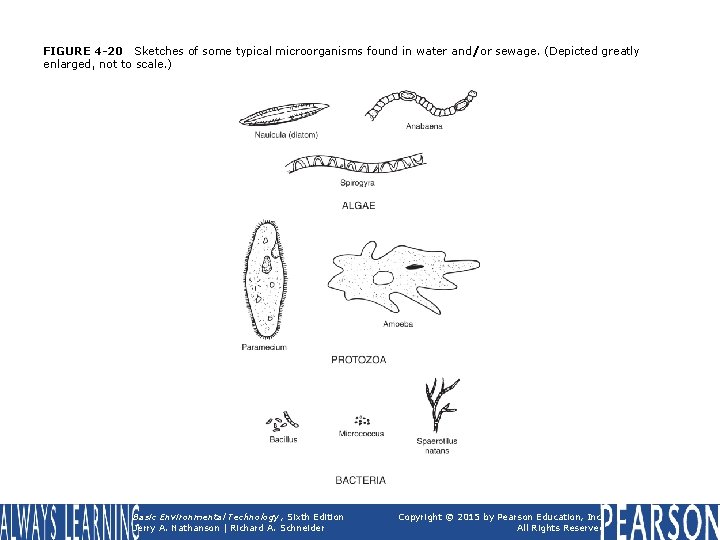
FIGURE 4 -20 Sketches of some typical microorganisms found in water and/or sewage. (Depicted greatly enlarged, not to scale. ) Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
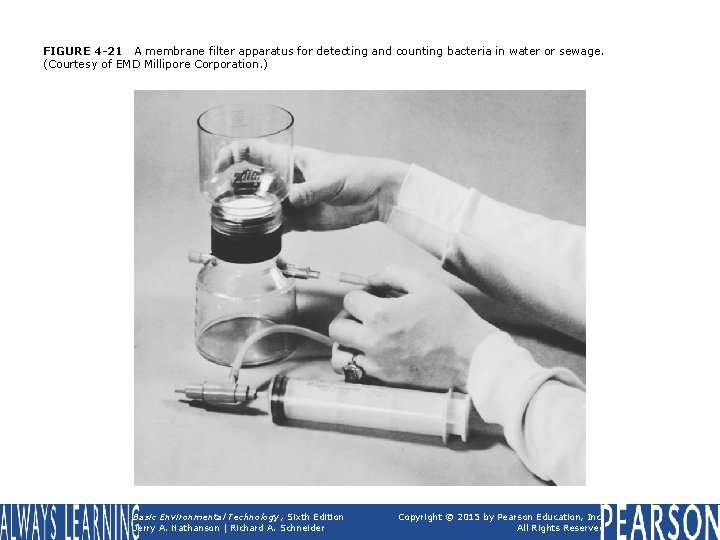
FIGURE 4 -21 A membrane filter apparatus for detecting and counting bacteria in water or sewage. (Courtesy of EMD Millipore Corporation. ) Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
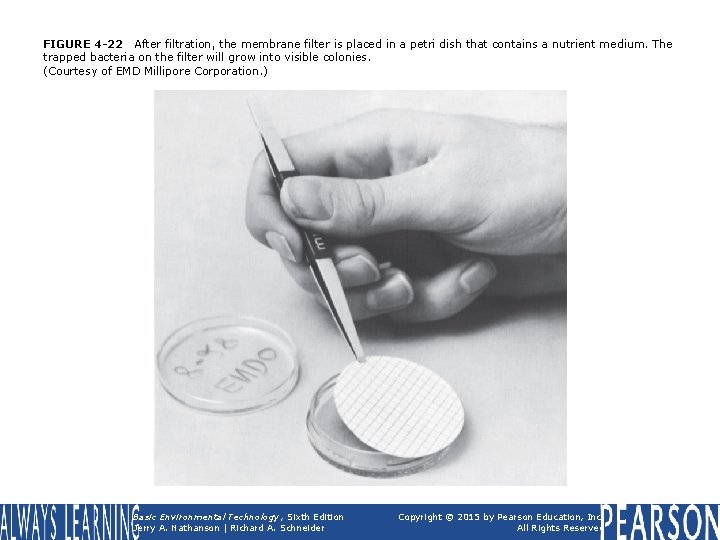
FIGURE 4 -22 After filtration, the membrane filter is placed in a petri dish that contains a nutrient medium. The trapped bacteria on the filter will grow into visible colonies. (Courtesy of EMD Millipore Corporation. ) Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
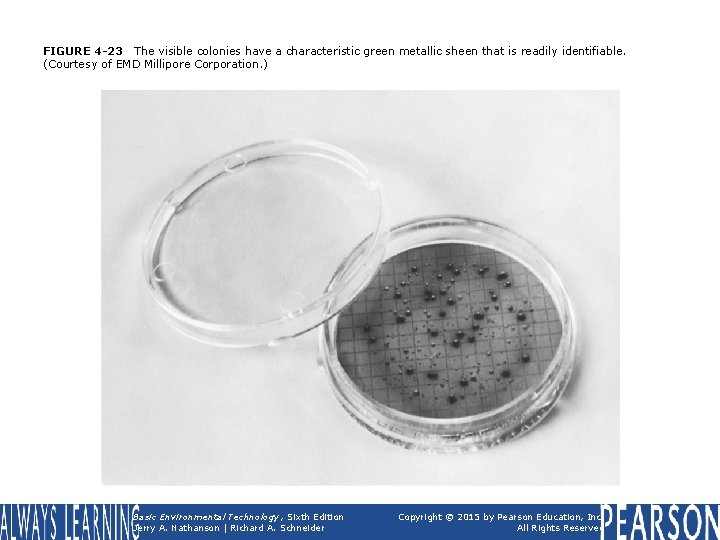
FIGURE 4 -23 The visible colonies have a characteristic green metallic sheen that is readily identifiable. (Courtesy of EMD Millipore Corporation. ) Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
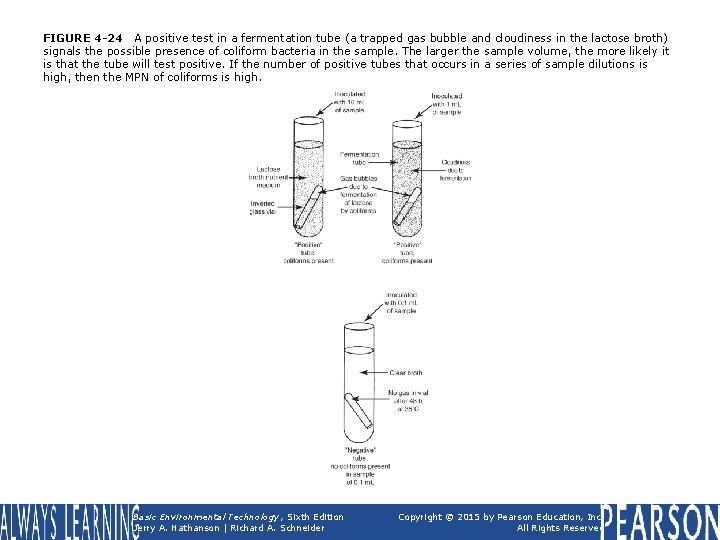
FIGURE 4 -24 A positive test in a fermentation tube (a trapped gas bubble and cloudiness in the lactose broth) signals the possible presence of coliform bacteria in the sample. The larger the sample volume, the more likely it is that the tube will test positive. If the number of positive tubes that occurs in a series of sample dilutions is high, then the MPN of coliforms is high. Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
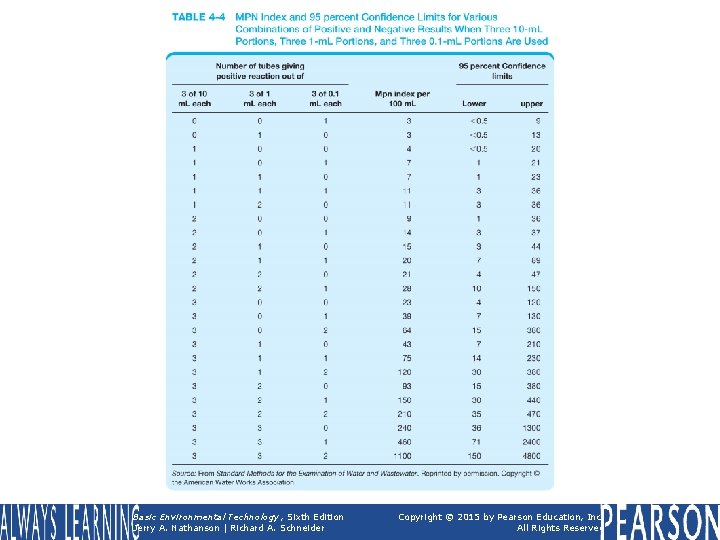
TABLE 4 -4 MPN Index and 95 percent Confidence Limits for Various Combinations of Positive and Negative Results When Three 10 -m. L Portions, Three 1 m. L Portions, and Three 0. 1 -m. L Portions Are Used Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
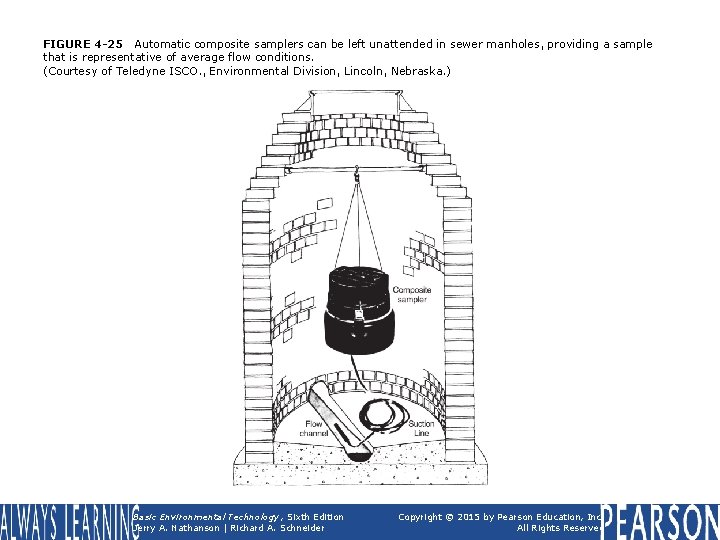
FIGURE 4 -25 Automatic composite samplers can be left unattended in sewer manholes, providing a sample that is representative of average flow conditions. (Courtesy of Teledyne ISCO. , Environmental Division, Lincoln, Nebraska. ) Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved

FIGURE 4 -26 Automatic composite samplers can be used to collect stream samples in order to determine the average water quality conditions over a period of time. (Isco. ) Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved

FIGURE 4 -27 All sample bottles should be properly identified, and records of the date, time, place, and type of sampling should be kept. (Reprinted by permission. Copyright © the American Water Works Association. ) Basic Environmental Technology, Sixth Edition Jerry A. Nathanson | Richard A. Schneider Copyright © 2015 by Pearson Education, Inc. All Rights Reserved
- Slides: 33