Basic Concepts to understand AEFI Data AEFI data

Basic Concepts to understand AEFI Data AEFI data management training course
Aspects discussed Understanding key AEFI data related terminology Understanding minor, severe and serious AEFI Applying terminology & concepts in the field | 2
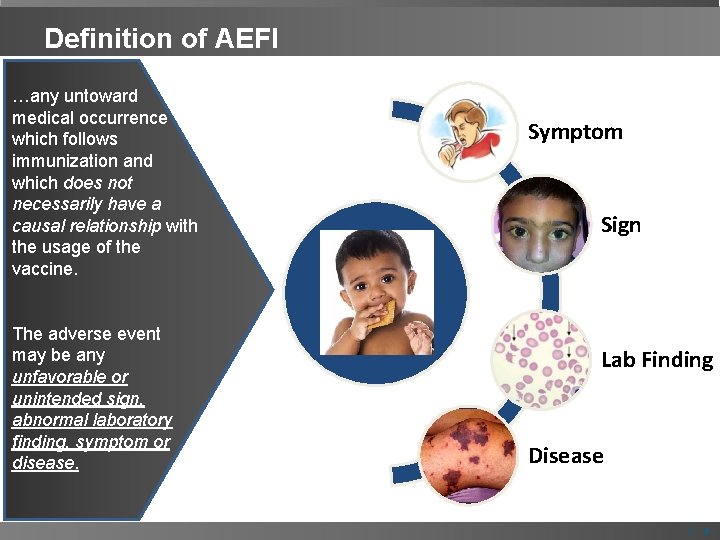
Definition of AEFI …any untoward medical occurrence which follows immunization and which does not necessarily have a causal relationship with the usage of the vaccine. The adverse event may be any unfavorable or unintended sign, abnormal laboratory finding, symptom or disease. Symptom Sign Lab Finding Disease | 3

Definition of AEFI (previous) An adverse event following immunization is a medical incident that takes place after an immunization, causes concern and is believed to be caused by the immunization. y l s u o i v e r (p ) d e us WHO Aide Memoire: AEFI Investigation, 2004 www. who. int/immunization_safety/en/ | 4
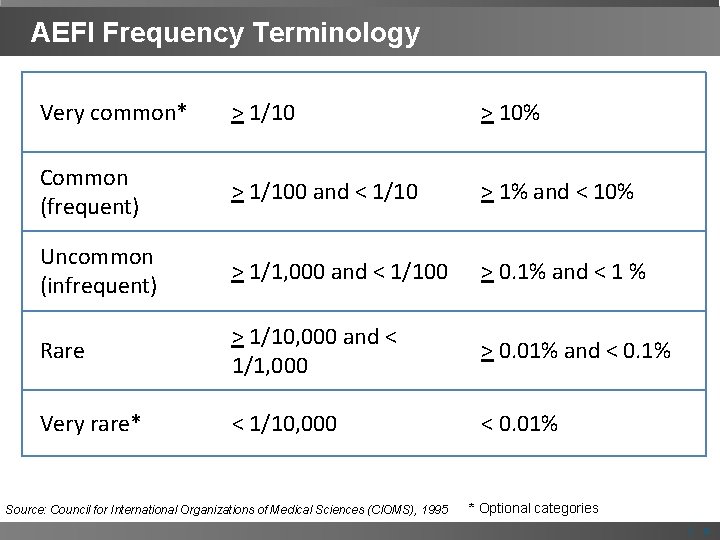
AEFI Frequency Terminology Very common* > 1/10 > 10% Common (frequent) > 1/100 and < 1/10 > 1% and < 10% Uncommon (infrequent) > 1/1, 000 and < 1/100 > 0. 1% and < 1 % Rare > 1/10, 000 and < 1/1, 000 > 0. 01% and < 0. 1% Very rare* < 1/10, 000 < 0. 01% Source: Council for International Organizations of Medical Sciences (CIOMS), 1995 * Optional categories | 5

CIOMS/ WHO cause specific definition of AEFIs 1 Vaccine productrelated reaction An AEFI that is caused or precipitated by a vaccine due to one or more of the inherent properties of the vaccine product. 2 Vaccine quality defect-related reaction An AEFI that is caused or precipitated by a vaccine that is due to one or more quality defects of the vaccine product including its administration device as provided by the manufacturer. 3 Immunization error-related reaction An AEFI that is caused by Inappropriate vaccine handling, prescribing or administration. 4 Immunization anxiety-related reaction An AEFI arising from anxiety about the immunization. 5 Coincidental event An AEFI that is caused by something other than the vaccine product, immunization error or immunization anxiety | 6
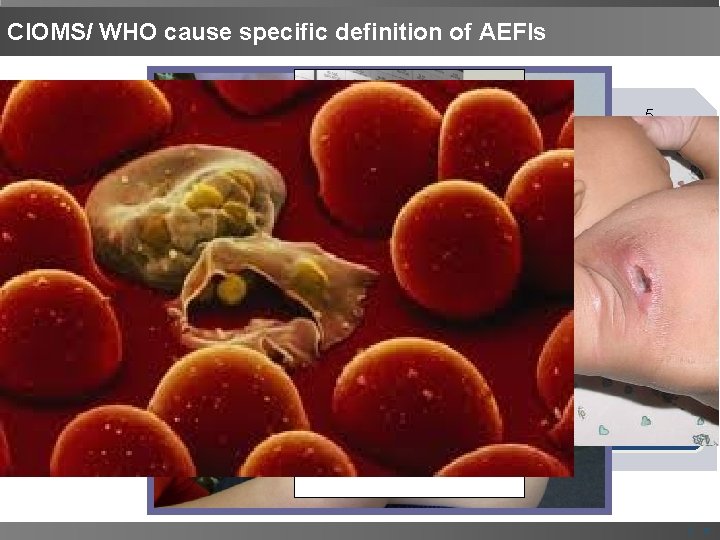
CIOMS/ WHO cause specific definition of AEFIs 1 Vaccine product -related reaction 2 Vaccine quality defect-related reaction 3 Immunization error-related reaction 4 Immunization anxiety-related reaction EXAMPLE Extensive limb swelling after DTP vaccine Failure by the manufacturer to completely inactivate a lot of inactivated polio vaccine leads to cases of paralytic polio. EXAMPLE Abscess formation due to improper technique of BCG 1955 - IPVvaccination. EXAMPLE Vasovagal syncope in an adolescent following vaccination. 120, 000 injected 40, 000 mild polio 200 paralysed 10 died 5 Coincidental event EXAMPLE A fever after vaccination (temporal association) and malarial parasite isolated from blood. | 7
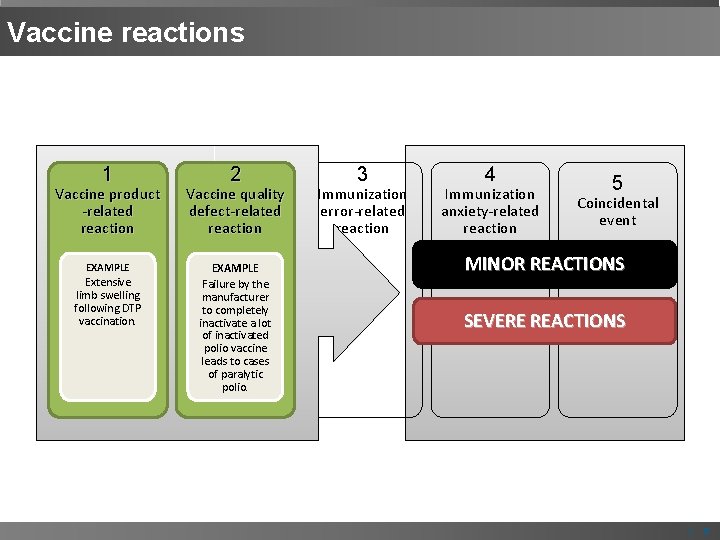
Vaccine reactions 1 2 Vaccine product -related reaction Vaccine quality defect-related reaction EXAMPLE Failure by the manufacturer to completely inactivate a lot of inactivated polio vaccine leads to cases of paralytic polio. Extensive limb swelling following DTP vaccination. 3 Immunization error-related reaction 4 Immunization anxiety-related reaction 5 Coincidental event MINOR REACTIONS SEVERE REACTIONS | 8

Vaccine reactions 1 Vaccine product -related reaction MINOR REACTIONS 2 Vaccine quality defect-related reaction SEVERE REACTIONS ÷ Usually occur within a few hours of injection. ÷ Resolve after short period of time and pose little danger. ÷ Local (includes pain, swelling or redness at the site of injection). ÷ Systemic (includes fever, malaise, muscle pain, headache or loss of appetite). | 9
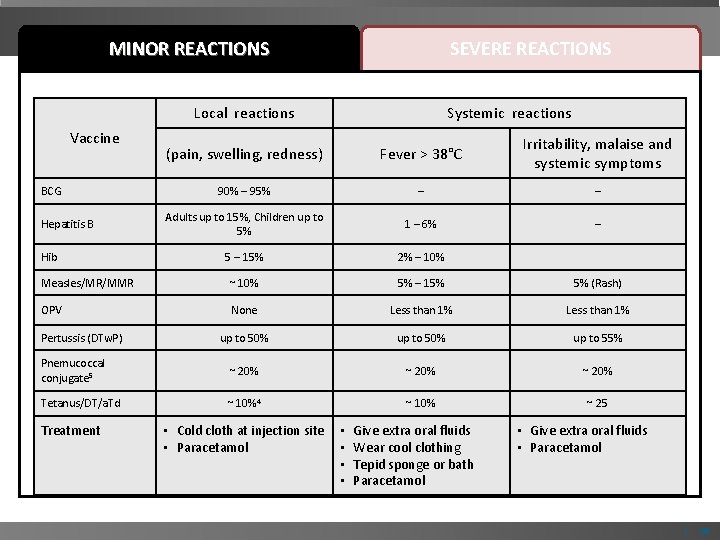
MINOR REACTIONS SEVERE REACTIONS Minor vaccine reactions, treatments and rates associated with childhood vaccines Local reactions Vaccine Systemic reactions (pain, swelling, redness) Fever > 38°C Irritability, malaise and systemic symptoms 90% – 95% – – Adults up to 15%, Children up to 5% 1 – 6% – 5 – 15% 2% – 10% Measles/MR/MMR ~ 10% 5% – 15% 5% (Rash) OPV None Less than 1% up to 50% up to 55% Pnemucoccal conjugate 5 ~ 20% Tetanus/DT/a. Td ~ 10%4 ~ 10% ~ 25 BCG Hepatitis B Hib Pertussis (DTw. P) Treatment • Cold cloth at injection site • Paracetamol • • Give extra oral fluids Wear cool clothing Tepid sponge or bath Paracetamol • Give extra oral fluids • Paracetamol | 10

Vaccine reactions 1 Vaccine product -related reaction MINOR REACTIONS 2 Vaccine quality defect-related reaction SEVERE REACTIONS ÷ Usually do not result in long-term problems. ÷ Can be disabling. ÷ Are rarely life threatening. ÷ Include seizures and allergic reactions caused by the body's reaction to a particular component in a vaccine. | 11
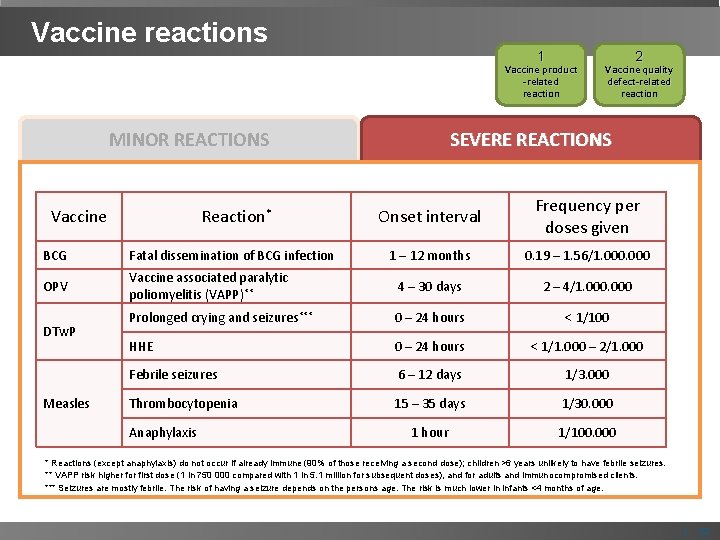
Vaccine reactions 1 Vaccine product -related reaction MINOR REACTIONS 2 Vaccine quality defect-related reaction SEVERE REACTIONS Severe vaccine reactions, treatments and rates associated with childhood vaccines Vaccine Reaction* Onset interval Frequency per doses given 1 – 12 months 0. 19 – 1. 56/1. 000 BCG Fatal dissemination of BCG infection OPV Vaccine associated paralytic poliomyelitis (VAPP)** 4 – 30 days 2 – 4/1. 000 Prolonged crying and seizures*** 0 – 24 hours < 1/100 HHE 0 – 24 hours < 1/1. 000 – 2/1. 000 Febrile seizures 6 – 12 days 1/3. 000 Thrombocytopenia 15 – 35 days 1/30. 000 1 hour 1/100. 000 DTw. P Measles Anaphylaxis * Reactions (except anaphylaxis) do not occur if already immune (90% of those receiving a second dose); children >6 years unlikely to have febrile seizures. ** VAPP risk higher for first dose (1 in 750 000 compared with 1 in 5. 1 million for subsequent doses), and for adults and immunocompromised clients. *** Seizures are mostly febrile. The risk of having a seizure depends on the persons age. The risk is much lower in infants <4 months of age. | 12

Difference between serious and severe reactions Severe reactions (Not regulatory term) • Can be disabling and, rarely, life threatening. • Must be reported. • Most do not lead to long-term problems. • Severe reactions include serious reactions but also include other severe reactions. Any untoward medical occurrence that Serious reactions at any dose: (Regulatory term) Results in death. Requires inpatient hospitalization. Results in persistent or significant disability. Is life-threatening. Congenital Anomaly | 13

Serious AEFI • Death Serious AEFI • Hospitalization or prolongation of existing hospitalization (e. g. , encephalopathy, seizures, aseptic meningitis) • Persistent or significant disability or incapacity (e. g. , paralysis) • Congenital anomaly/birth defect • Life-threatening ‘Serious’ is not synonymous with ‘severe’ (i. e. , intensity or severity of the event) Source: report of CIOMS/WHO Working Group on Vaccine Pharmacovigilance, 2012 | 14
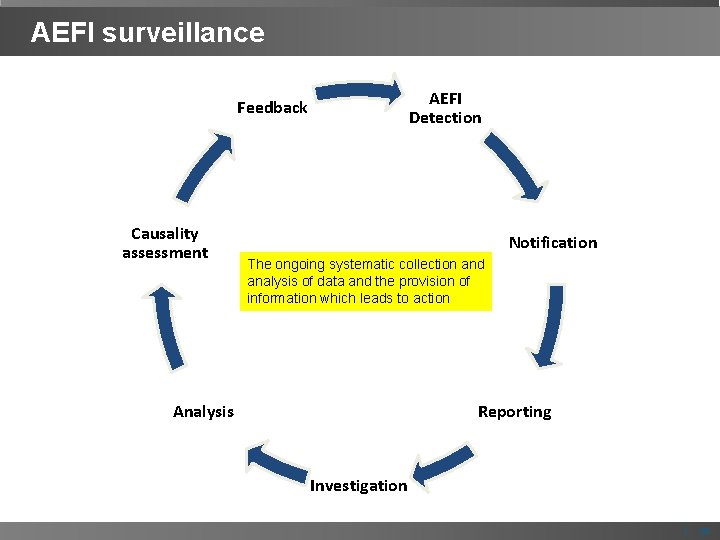
AEFI surveillance AEFI Detection Feedback Causality assessment Notification The ongoing systematic collection and analysis of data and the provision of information which leads to action Analysis Reporting Investigation | 15
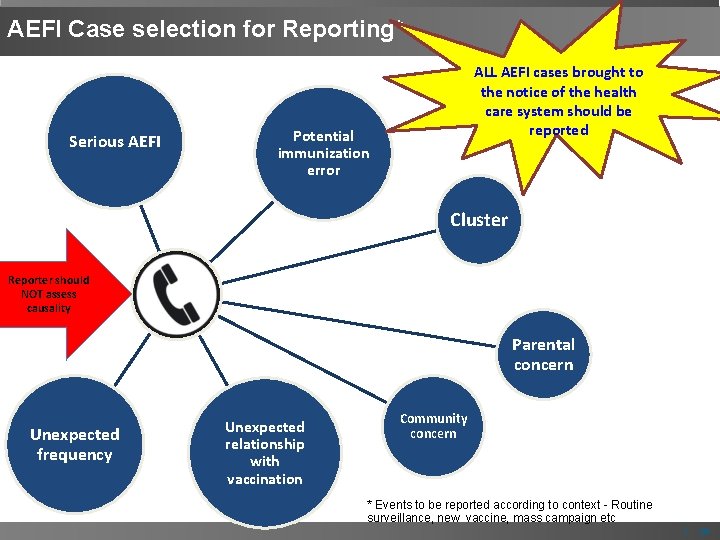
AEFI Case selection for Reporting* Serious AEFI ALL AEFI cases brought to the notice of the health care system should be reported Potential immunization error Cluster Reporter should NOT assess causality Parental concern Unexpected frequency Unexpected relationship with vaccination Community concern * Events to be reported according to context - Routine surveillance, new vaccine, mass campaign etc | 16

Cluster "A cluster of AEFIs is two or more cases of the same adverse event related in time, place or vaccine administered". * Aggregation of relatively uncommon events or diseases in space and/or time in frequency that are believed or perceived to be greater than could be expected by chance. ** * WHO Aide Memoire: AEFI Investigation, 2004 ** Last, John M. 2001. A Dictionary of Epidemiology, 4 th ed, | 17

Signal Reported information on possible causal relationship between an adverse event and vaccine Previously unknown Or Incompletely documented Usually more than a single report required Important considerations • seriousness of the event • quality of the information. Source: WHO International Drug Monitoring Programme, 1991 | 18

AEFIs needing special attention If immunization error is suspected • e. g. bacterial abscess, severe local reaction, high fever or sepsis, BCG lymphadenitis, toxic shock syndrome, clusters of AEFIs) Significant events of unexplained cause occurring within 30 days after a vaccination Events causing parental or community concern | 19

Causality and Causality assessment Causality* • Is the relationship between two events (the cause and the effect), where the second event is a consequence of the first Causality Assessment • Determining if such a relationship exists and if so to what extent *A direct cause is a factor in absence of which the effect would not occur (necessary cause). *Sometimes, there are multiple factors that can precipitate or function as co-factors for the effect (event) to occur. | 20
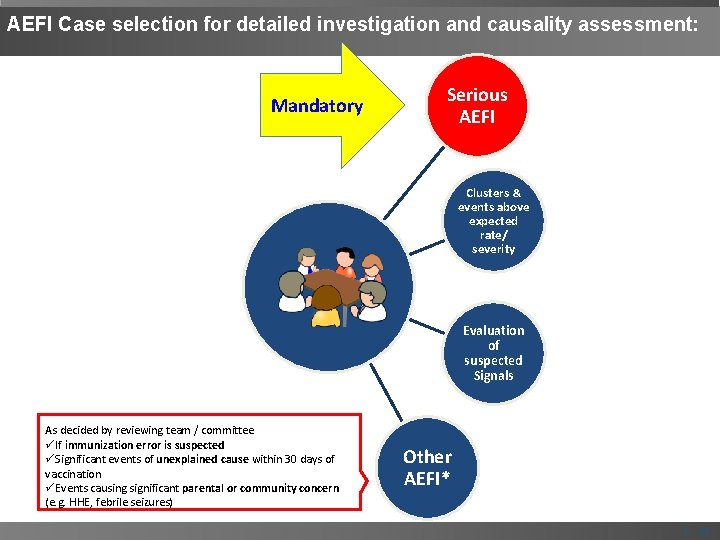
AEFI Case selection for detailed investigation and causality assessment: Mandatory Serious AEFI Clusters & events above expected rate/ severity Evaluation of suspected Signals As decided by reviewing team / committee üIf immunization error is suspected üSignificant events of unexplained cause within 30 days of vaccination üEvents causing significant parental or community concern (e. g. HHE, febrile seizures) Other AEFI* | 21

Why apply these concepts & terminology to AEFI? Different immunization schedules Different vaccines Batch related problems Cold chain problems Immunization errors Different background rates Coincidental events Genetic differences Globalization of vaccine safety | 22
- Slides: 22