Basic Concepts of Electrochemistry ELECTROCHEMISTRY Electricitydriven Chemistry or

Basic Concepts of Electrochemistry ELECTROCHEMISTRY Electricity-driven Chemistry or Chemistry-driven Electricity: charge flow (electrons, holes, ions) Chemistry (redox): reduction = electron uptake oxidation = electron loss Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
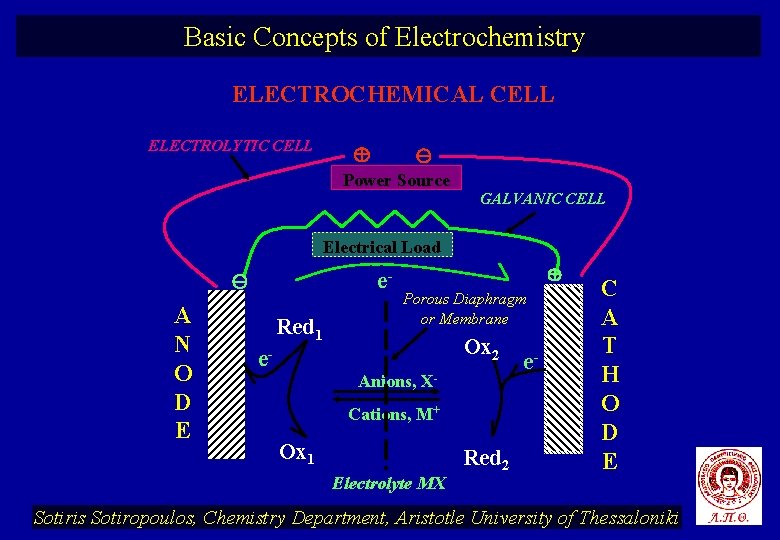
Basic Concepts of Electrochemistry ELECTROCHEMICAL CELL ELECTROLYTIC CELL Power Source GALVANIC CELL Electrical Load A N O D E e- Red 1 Porous Diaphragm or Membrane Ox 2 e- Anions, XCations, M+ Ox 1 Electrolyte MX Red 2 e- C A T H O D E Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
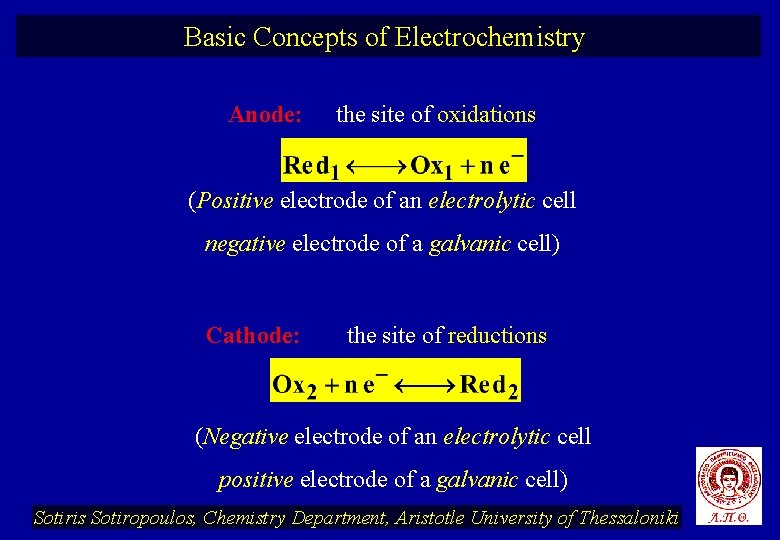
Basic Concepts of Electrochemistry Anode: the site of oxidations (Positive electrode of an electrolytic cell negative electrode of a galvanic cell) Cathode: the site of reductions (Negative electrode of an electrolytic cell positive electrode of a galvanic cell) Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki

Basic Concepts of Electrochemistry ELECTROCHEMICAL APPLICATIONS (Conversion of Chemical to Electrical Energy) • Batteries (for electronic devices, automotion etc) • Fuel Cells (for automotion, power stations etc) • Electroanalysis (potentiometric electronalytical techniques and sensors, e. g. ion selective electrodes, gas sensors etc) Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki

Basic Concepts of Electrochemistry ELECTROCHEMICAL APPLICATIONS (Conversion of Electrical to Chemical Energy) • Electrolysis (e. g. chloralkali industry, hydrogen production) • Electrosynthesis (e. g. adiponitrile production Νylon 66) • Electroplating and Metal Processing (e. g. decorative metal plating, elctrochemical machining) • Cathodic corrosion protection of metals and metal composites (e. g. bridge and ship protection) • Waste treatment (e. g. metal ion removal and recovery, organics oxidation etc) • Electroanalysis and Elecrochemical Sensors (e. g. determination of heavy metals, organic contaminants and biological compounds; glucose, oxygen, ethanol sensors) Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki

Basic Concepts of Electrochemistry PARAMETERS OF AN ELECTROCHEMICAL PROCESS • Cell or electrode potential: E • • Current or current density: Ι or i=Ι/Α • Concentration of electroactive species in the bulk (homogeneous) solution: C b time: t i=f(E, Cb, t) or E=g(i, Cb, t ) Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
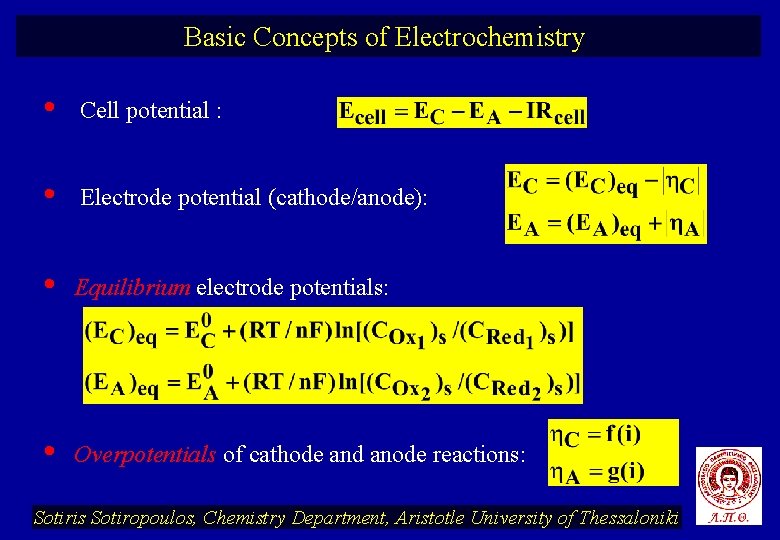
Basic Concepts of Electrochemistry • Cell potential : • Electrode potential (cathode/anode): • Equilibrium electrode potentials: • Overpotentials of cathode and anode reactions: Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki

Basic Concepts of Electrochemistry GENERAL STEPS OF AN ELECTRODE PROCESS • Mass transfer of reactants/products to/from the electrode. • Surface reactions (e. g. adsorption, phase transitions etc). • Charge transfer (heterogeneous electron or hole exchange) at the electrode surface. • Homogeneous chemical reactions in the bulk solution. Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
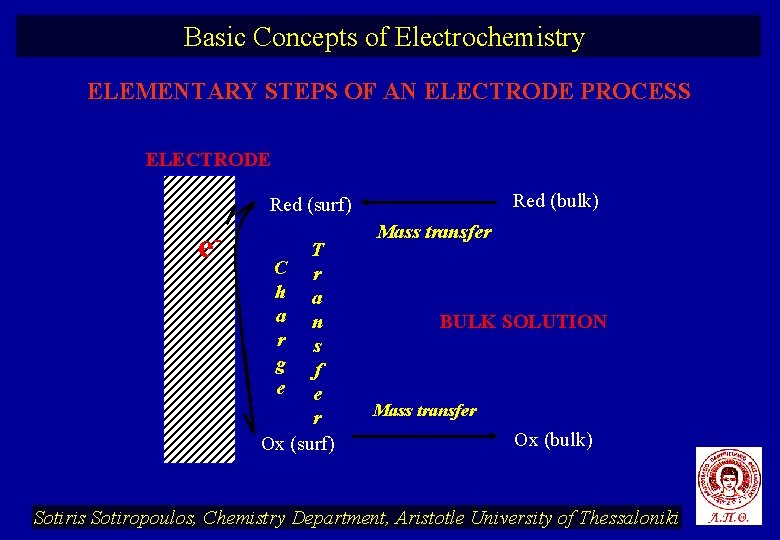
Basic Concepts of Electrochemistry ELEMENTARY STEPS OF AN ELECTRODE PROCESS ELECTRODE Red (bulk) Red (surf) e- T C r h a a n r s g f e e r Ox (surf) Mass transfer BULK SOLUTION Mass transfer Ox (bulk) Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki

Basic Concepts of Electrochemistry CURRENT DENSITY-ELECTRODE REACTION RATE where: • • km = mass transfer coefficient ke = f(diffusion/flow rate and cell geometry ) = charge transfer coefficient = f(electrode reaction, electrode material, electrode potential) Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
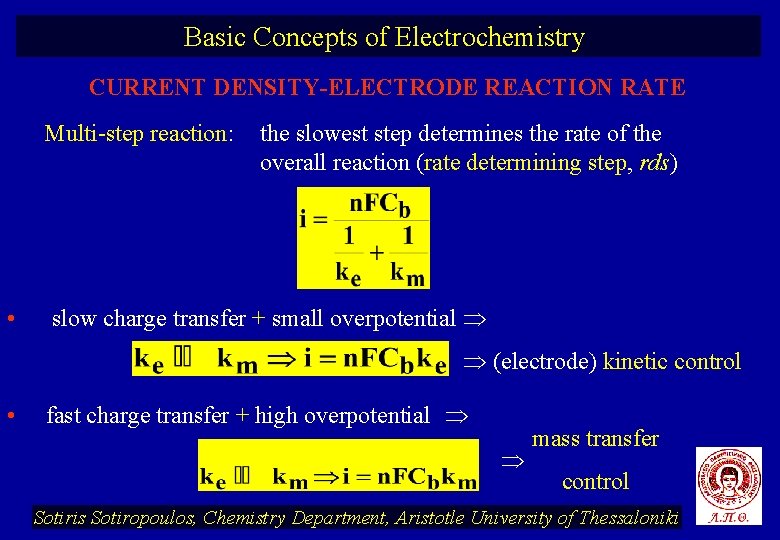
Basic Concepts of Electrochemistry CURRENT DENSITY-ELECTRODE REACTION RATE Multi-step reaction: • the slowest step determines the rate of the overall reaction (rate determining step, rds) slow charge transfer + small overpotential (electrode) kinetic control • fast charge transfer + high overpotential mass transfer control Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
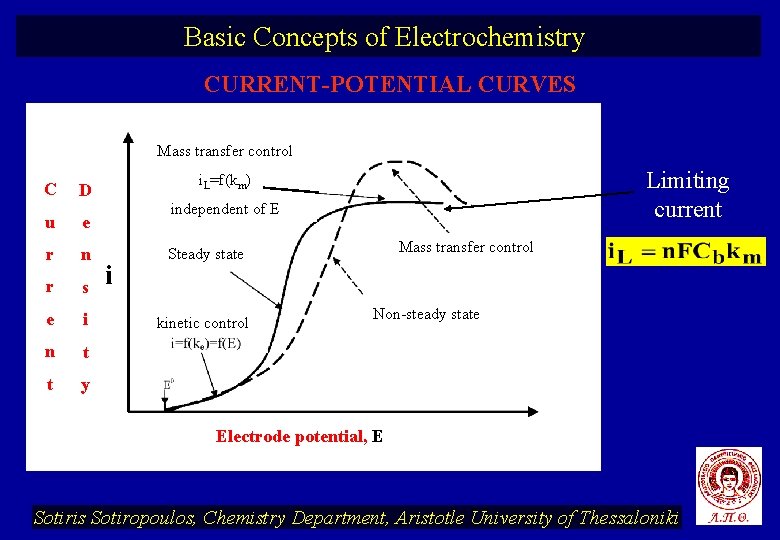
Basic Concepts of Electrochemistry CURRENT-POTENTIAL CURVES Mass transfer control C u e r n r s e i n t t y Limiting current i. L=f(km) D independent of E i Masstransfercontrol Steady state kinetic control Non-steady state Electrode potential, E Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
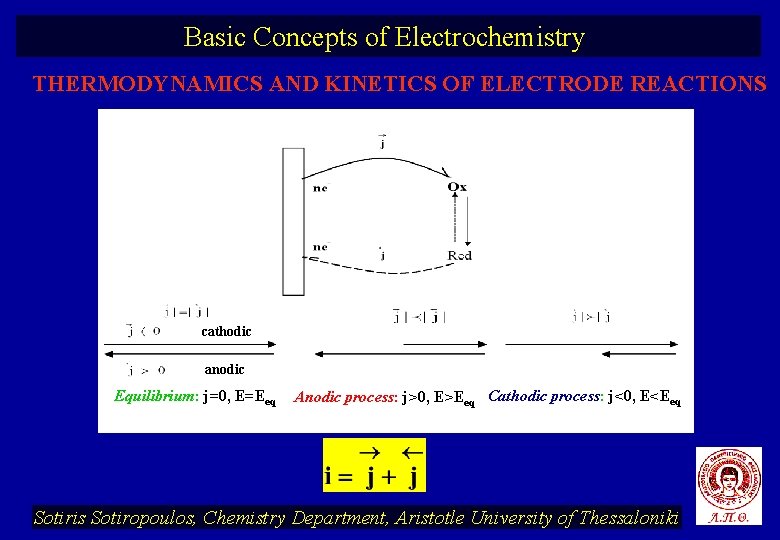
Basic Concepts of Electrochemistry THERMODYNAMICS AND KINETICS OF ELECTRODE REACTIONS cathodic anodic Equilibrium: j=0, E=Eeq Anodic process: j>0, E>Eeq Cathodic process: j<0, E<Eeq Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
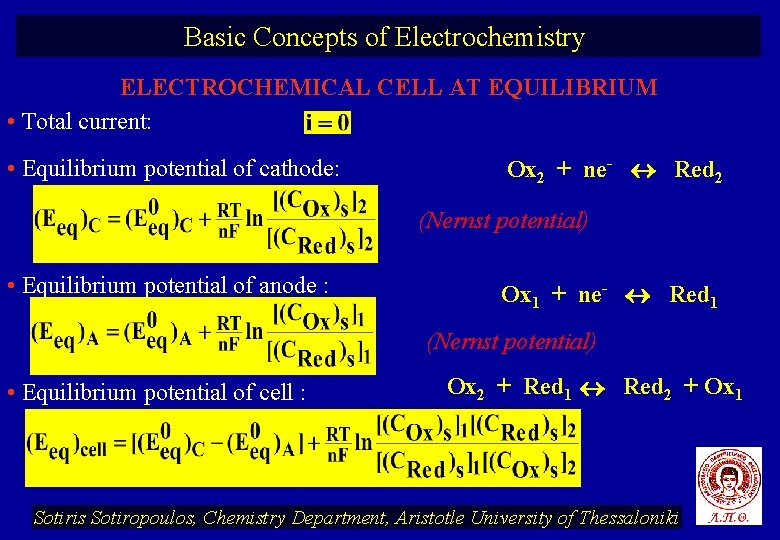
Basic Concepts of Electrochemistry ELECTROCHEMICAL CELL AT EQUILIBRIUM • Total current: • Equilibrium potential of cathode: Ox 2 + ne- Red 2 (Nernst potential) • Equilibrium potential of anode : Ox 1 + ne- Red 1 (Nernst potential) • Equilibrium potential of cell : Ox 2 + Red 1 Red 2 + Ox 1 Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki

Basic Concepts of Electrochemistry ELECTROCHEMICAL CELL AT EQUILIBRIUM Free-Gibbs Energy (of the overall reaction Ox 2 + Red 1 Red 2 + Ox 1 in the electrochemical cell): • (Eeq)cell > 0 ΔG < 0 spontaneous process (galvanic cell) • (Eeq)cell < 0 ΔG > 0 non spontaneous process (electrolytic cell) Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki

Basic Concepts of Electrochemistry KINETICS OF AN IRREVERSIBLE-SLOW ELECTRODE REACTION • • slow charge transfer: kinetically controlled current: (η=Ε-Εeq) Butler-Volmer equation Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
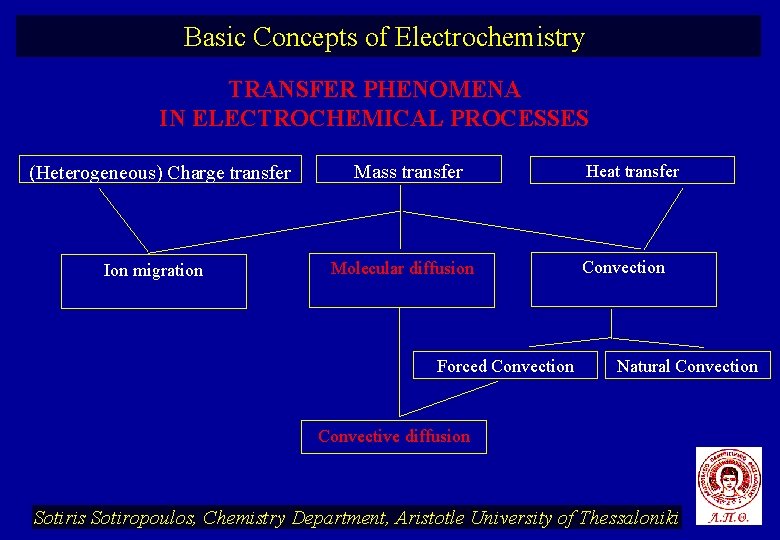
Basic Concepts of Electrochemistry TRANSFER PHENOMENA IN ELECTROCHEMICAL PROCESSES (Heterogeneous) Charge transfer Ion migration Mass transfer Molecular diffusion Forced Convection Heat transfer Convection Natural Convection Convective diffusion Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
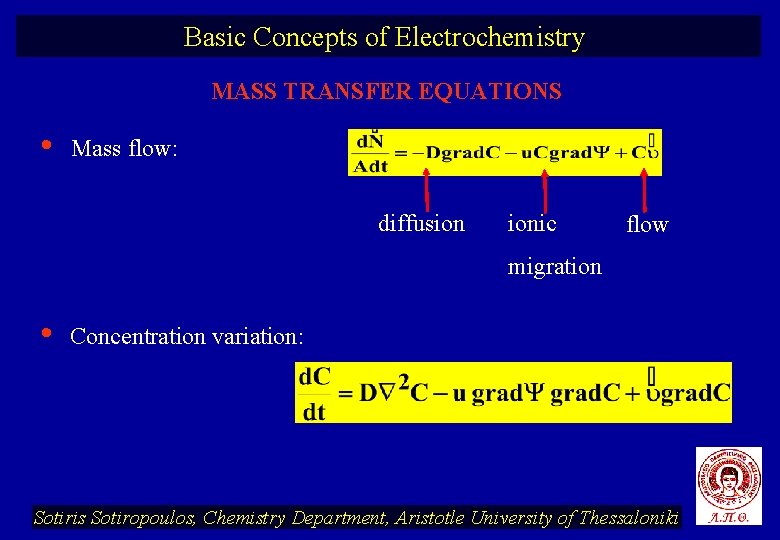
Basic Concepts of Electrochemistry MASS TRANSFER EQUATIONS • Mass flow: diffusion ionic flow migration • Concentration variation: Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
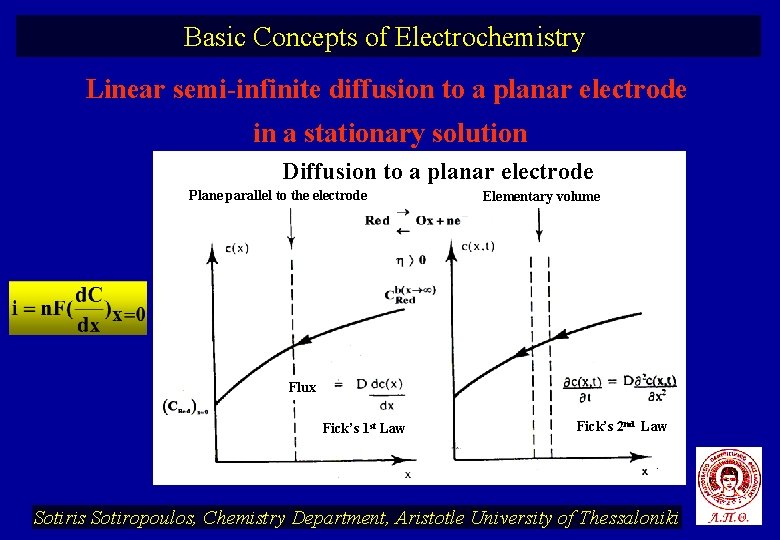
Basic Concepts of Electrochemistry Linear semi-infinite diffusion to a planar electrode in a stationary solution Diffusion to a planar electrode Plane parallel to the electrode Elementary volume Flux Fick’s 1 st Law Fick’s 2 nd Law Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
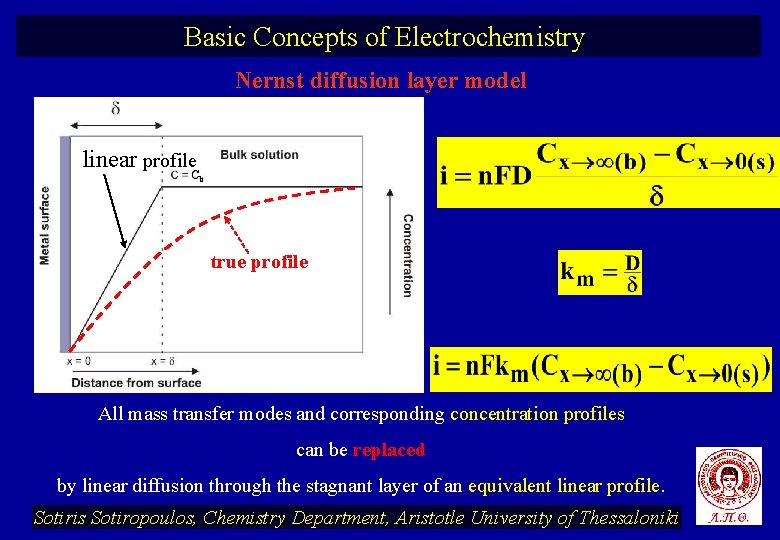
Basic Concepts of Electrochemistry Nernst diffusion layer model linear profile. C b true profile All mass transfer modes and corresponding concentration profiles can be replaced by linear diffusion through the stagnant layer of an equivalent linear profile. Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki
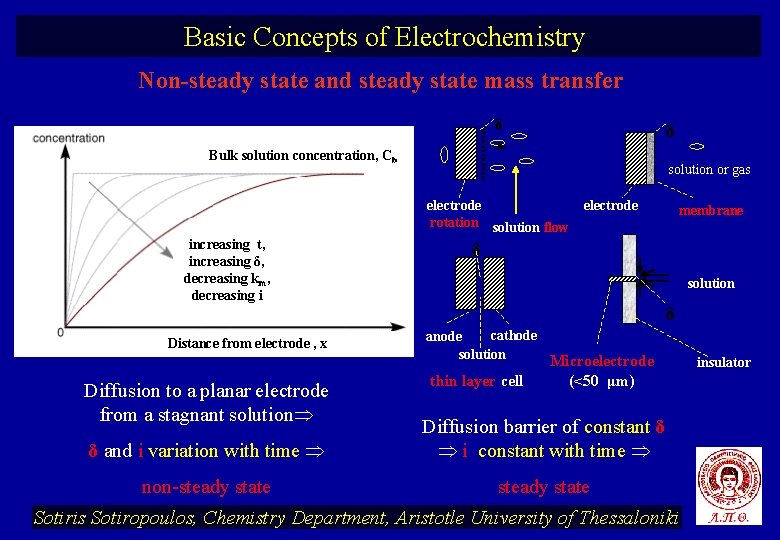
Basic Concepts of Electrochemistry Non-steady state and steady state mass transfer δ δ Bulk solution concentration, Cb solution or gas electrode rotation solution flow increasing t, increasing δ, decreasing km, decreasing i Distance from electrode , x Diffusion to a planar electrode from a stagnant solution electrode membrane δ solution δ cathode anode solution thin layer cell Microelectrode (<50 μm) δ and i variation with time Diffusion barrier of constant δ i constant with time non-steady state Sotiris Sotiropoulos, Chemistry Department, Aristotle University of Thessaloniki insulator
- Slides: 21