BASIC CONCEPTS IN INFECTION CONTROL Decontamination of equipment

BASIC CONCEPTS IN INFECTION CONTROL Decontamination of equipment International Federation of Infection Control

Decontamination n n Decontamination is a process which removes or destroys microorganisms to render an object safe for use It includes n n n Cleaning Disinfection Sterilisation © IFIC: 2005

Cleaning n A process that removes foreign material (e. g. soil, organic material, microorganisms) from an object © IFIC: 2005

Disinfection n A process that reduces the number of pathogenic micro-organisms from inanimate objects or skin to a level which is not harmful to health © IFIC: 2005

High level disinfection n A process that kills Mycobacterium tuberculosis & enteroviruses, in addition to other vegetative bacteria, fungi & more sensitive viruses © IFIC: 2005

Sterilisation n A process that destroys all microorganisms including bacterial spores n Sterilisation cannot be proved except by culturing. This is not routinely done, so normally an object is said to have been sterilized if it has gone through a controlled process of sterilisation © IFIC: 2005

n n Choice of decontamination method The level of decontamination should be such that there is no risk of infection when using the equipment The choice of the method depends on the type of material, number & type of organisms involved & risk of infection to patients or staff © IFIC: 2005

Classification of infection risk n Infection risk from equipment or environment can be classified into three categories that indicate the level of decontamination required 1. Low risk 2. Intermediate risk 3. High risk © IFIC: 2005

1. Low risk n n Items in contact with normal & intact skin, or the inanimate environment not in contact with the patient (e. g. walls, floors, ceilings, furniture, sinks & drains) Cleaning & drying is usually adequate for decontamination © IFIC: 2005

2. Intermediate risk n n Equipment that does not penetrate the skin, or enter sterile areas of the body, but is in contact with mucous membranes or non-intact skin, Other items contaminated with virulent or transmissible organisms n n e. g. respiratory equipment, gastrointestinal endoscopes, vaginal instruments, thermometers Cleaning followed by disinfection is usually adequate decontamination © IFIC: 2005

3. High risk n Items penetrating sterile tissues, including body cavities & the vascular system, n n e. g. surgical instruments, intra-uterine devices, vascular catheters Cleaning followed by sterilisation is required for safe decontamination © IFIC: 2005

The need for cleaning before disinfection n The presence of organic material impedes disinfection and sterilization by providing protection for microorganisms In addition, these decontamination processes may damage equipment by fixing protein residues to their surfaces For these reasons, thorough cleaning of used equipment before disinfection or sterilization is essential © IFIC: 2005

Protection of cleaning staff n Staff responsible for cleaning contaminated equipment shall: n n n Have received adequate training Wear appropriate protective clothing, such as gloves, gowns and aprons, masks, and goggles Be immunised against hepatitis A and hepatitis B viruses © IFIC: 2005

Manual Cleaning n n n Staff handling contaminated items should wear good quality gloves for personal protection All items requiring disinfection or sterilisation should be dismantled before cleaning Cold water is preferred for cleaning as it hot water or disinfectants will coagulate protein materials (blood, sputum, etc. ), making them more difficult to remove (Cont. ) © IFIC: 2005

Manual cleaning (Cont. ) n The most simple, cost effective method is brushing n n n Keep the brush below the surface of the water to prevent the release of aerosols Decontaminated & dry the brush after use Finally rinse items in clean, warm water & dry. Items are then ready for use or disinfection or sterilisation © IFIC: 2005

Methods of disinfection n n Disinfection can be carried out by using heat or chemicals Thermal disinfection is preferred. n It is generally more reliable than chemicals, leaves no residues, is more easily controlled & is non-toxic © IFIC: 2005

Steam sterilisation n n Moist heat as saturated steam under pressure is the most widely used method of sterilisation It is dependable, non-toxic, has good penetrating ability, is relatively inexpensive, is sporidicidal, and can easily be monitored for efficacy © IFIC: 2005

Autoclaves n n n In modern pressurised autoclaves, dry steam under pressure is raised to temperatures above boiling Large quantities of heat energy are released during the conversion of steam to water This is transferred to any microorganisms present and is an efficient method of killing of heat resistant organisms and spores © IFIC: 2005

There are two types of steam steriliser n n Gravity displacement autoclaves admit steam at the top of the chamber, forcing air out the bottom through a drain High-speed pre-vacuum sterilisers are similar except that they are fitted with a pump to remove air from the chamber before steam is admitted, improving the efficiency of the process © IFIC: 2005
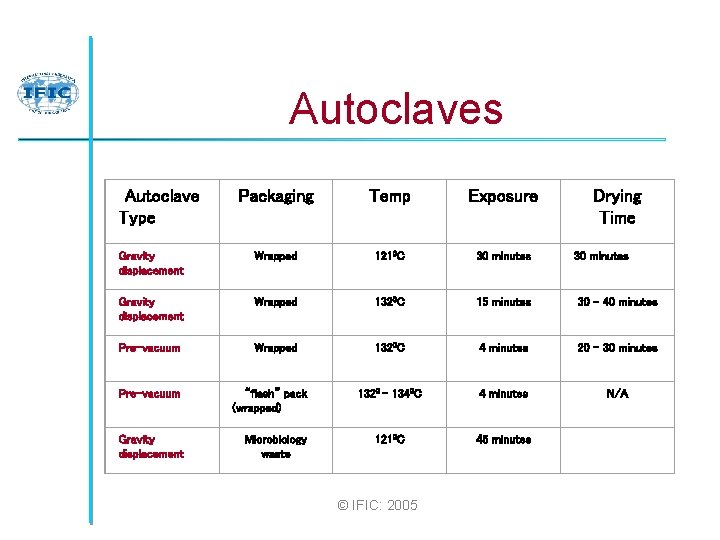
Autoclaves Autoclave Type Packaging Temp Exposure Drying Time Gravity displacement Wrapped Pre-vacuum Wrapped 1210 C 30 minutes 1320 C 15 minutes 30 – 40 minutes 4 minutes 20 – 30 minutes 4 minutes N/A 1320 C Pre-vacuum Gravity displacement “flash” pack (wrapped) Microbiology waste 1320 – 1340 C 1210 C 45 minutes © IFIC: 2005

Benchtop sterilizers n Small steam sterilisers that are useful in dentist's offices and outpatient clinics n n n They will sterilise wrapped & unwrapped instruments, rubber and plastic. They must be maintained & operated according to the manufacturers' instructions and operators must be trained in their use Written records of maintenance and operating parameters must be kept and audited © IFIC: 2005

Steam sterilisers must be maintained regularly to ensure they are working effectively n n A written record of maintenance must be kept and audited The maintenance of correct parameters of steam, pressure, temperature, and time are essential to ensure sterilisation. Regular biological testing is done with Bacillus stearothermophilus A written record of test results must be kept and audited © IFIC: 2005

The use of pressure cookers n n n Pressure cookers have been used to reprocess items While pressure cookers are relatively inexpensive and simple to use, it is difficult to ensure and evaluate their efficacy It is also difficult to ensure that the items and their wrappings are dry before storage © IFIC: 2005

Boiling n n n Boiling is not a reliable method of sterilisation, but it is useful in parts of the world where resources are sparse To obtain the best results, ensure that heat -resistant items are absolutely clean and that items are boiled for at least five minutes The boiler must be well maintained © IFIC: 2005

Disadvantages of boiling n These include: n n Lack of ability to destroy bacterial spores and some viruses Since it is impossible to package items to be boiled, there are difficulties with storage n Instruments may be stored in stainless steel trays that have been sterilised or boiled; they should be transferred to the trays with transfer forceps that have been similarly treated © IFIC: 2005

Disinfection by boiling n n Clean the item thoroughly Place in a container & cover with water Heat the water to boiling & start timing the process Boiling should continue for at least 5 minutes (Cont. ) © IFIC: 2005

Disinfection by boiling (Cont. ) n n n Addition of 2% sodium bicarbonate helps to prevent corrosion of the instruments & utensils If cheatle forceps are used for handling of items, they should be boiled (or autoclaved) with the holder at least daily & stored dry The boiler should be emptied & dried daily © IFIC: 2005

Disinfection by water at lower temperatures n This can be used for items damaged by higher temperatures n e. g. 80°C for 5 min, using suitable temperature controlling equipment © IFIC: 2005

n Chemicals for high-level disinfection A limited number of disinfectants can be used for this purpose n n n e. g. glutaraldehyde 2%, 6% hydrogen peroxide, 0. 2 -0. 3% peracetic acid If a sporicidal action is required, immersion in 2% glutaraldehyde for at least 3 hours is required Gluteraldehyde vapour is hazardous & appropriate extraction & respiratory protection should be provided if this compound is used © IFIC: 2005

Storage & dilution of chemical disinfectants n n Chemical disinfectants must be made up freshly to the correct concentrations according to the manufactures’ instructions & discarded after the correct period of time or number of uses They should be stored in clean bottles with plastic stoppers. When bottles are empty they should be thoroughly cleaned before re-filling © IFIC: 2005 (Cont. )

Storage & dilution of chemical disinfectants n Partially empty bottles should not be topped up since this will encourage contamination with, & multiplication of, disinfectant resistant organisms © IFIC: 2005

After chemical disinfection: n Thoroughly rinse the item with sterile water n n If sterile water is not available, freshly boiled water can be used. After rinsing, keep the items dry & protected from recontamination © IFIC: 2005

References & bibliography n n n Mc. Farland LV et al. Nosocomial acquisition of Clostridium difficile infection. New Engl J Med 1989; 320: 204 -8. Canada Communicable Disease Report Supplement Infection Control Guidelines: Handwashing, Cleaning Disinfection and Sterilisation in Healthcare, Health Canada, Laboratory Centre for Disease Control, Bureau of Infectious Disease, Nosocomial and Occupational Infections. 1998. Bennett JV, Brachman PS, Eds. . Hospital Infections, 4 th ed. Lippincott - Raven Publishers, Philadelphia and New York, 1998. Wenzel RP, Ed. Prevention and Control of Nosocomial Infections. Williams & Wilkins, Baltimore, 1994. Reichert M, Young JH. Sterilization Technology for the Health Care Facility, 2 nd ed. , Jones and Bartlett, Publishers, 1997. Boston, Toronto, London, Singapore. © IFIC: 2005
- Slides: 33