Basic Chemistry Review for APES What is MATTER

Basic Chemistry Review for APES What is MATTER? What is Energy?

Key Concepts Ø Science as a process for understanding Ø Components and regulation of systems Ø Matter: forms, quality, and how it changes; laws of matter Ø Energy: forms, quality, and how it changes; laws of energy Ø Nuclear changes and radioactivity
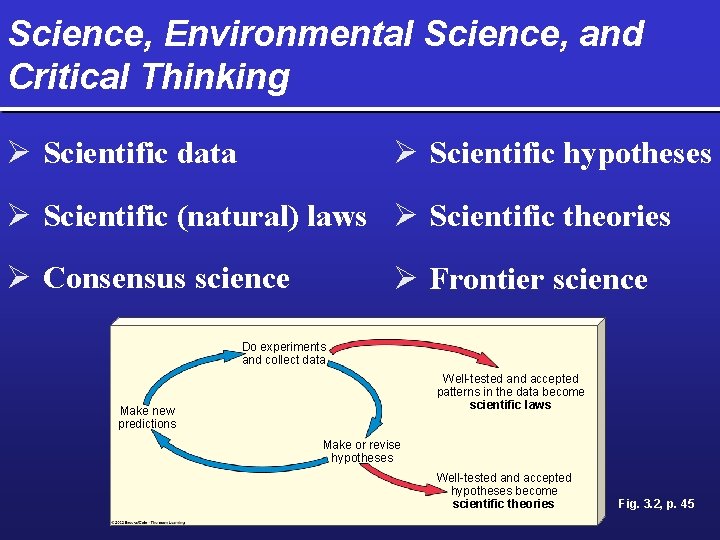
Science, Environmental Science, and Critical Thinking Ø Scientific data Ø Scientific hypotheses Ø Scientific (natural) laws Ø Scientific theories Ø Consensus science Ø Frontier science Do experiments and collect data Well-tested and accepted patterns in the data become scientific laws Make new predictions Make or revise hypotheses Well-tested and accepted hypotheses become scientific theories Fig. 3. 2, p. 45

Models and Behavior of Systems Ø Inputs Ø Flows (throughputs) Ø Stores (storage areas) Ø Outputs
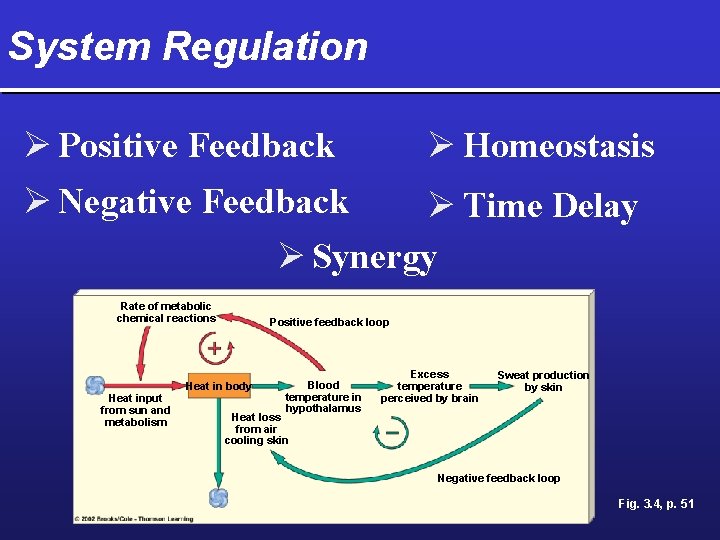
System Regulation Ø Homeostasis Ø Positive Feedback Ø Negative Feedback Ø Time Delay Ø Synergy Rate of metabolic chemical reactions Heat input from sun and metabolism Positive feedback loop Heat in body Blood temperature in hypothalamus Excess temperature perceived by brain Sweat production by skin Heat loss from air cooling skin Negative feedback loop Fig. 3. 4, p. 51

Matter: Forms, Structure, and Quality Ø Elements Ø Compounds Ø Molecules Ø Mixtures

Atoms Subatomic Particles Ø Protons Ø Neutrons Ø Electrons Atomic Characteristics Ø Atomic number Ø Ions Ø Atomic mass Ø Isotopes

Examples of Atoms Hydrogen (H) 0 n 1 p 1 e Mass number = 0 + 1 = 1 Hydrogen-1 (99. 98%) 2 n 1 p 1 e Mass number = 1 + 1 = 2 Hydrogen-2 or deuterium (0. 015%) 1 e Mass number = 2 + 1 = 3 Hydrogen-3 or tritium (T) (trace) Uranium (U) 143 n 92 p 92 e Mass number = 143 + 92 = 235 Uranium-235 (0. 7%) 146 143 n 92 p 92 e Mass number = 146 + 92 = 238 Uranium-238 (99. 3%) Fig. 3. 6, p. 55
Chemical Bonds Ø Chemical formulas Ø Ionic bonds Ø Covalent bonds Ø Hydrogen bonds

Organic Compounds Ø Organic vs. inorganic compounds Ø Hydrocarbons Ø Chlorinated hydrocarbons Ø Chlorofluorocarbons Ø Simple carbohydrates Ø Complex carbohydrates Ø Proteins

Genetic Material Ø Nucleic acids Ø Genes Ø Chromosomes Ø Gene mutations The human body contains about 100 trillion cells. There is a nucleus inside each human cell (except red blood cells). Each nucleus contains 46 chromosomes, arranged in 23 pairs. One chromosome of every pair is from each parent. The chromosomes are filled with tightly coiled strands of DNA. Genes are segments of DNA that contain instructions to make proteins—the building blocks of life. There approximately 140, 000 genes in each cell, each coded by sequences of nucleotides in its DNA molecules. GT AC C G A T Fig. 3. 8, p. 57

Matter Quality and Material Efficiency Ø High-quality matter Ø Low-quality matter Ø Entropy High Quality Low Quality Solid Gas Salt Solution of salt in water Coal Ø Material efficiency (resource productivity) Coal-fired power plant emissions Gasoline Aluminum can Automobile emissions Aluminum ore Fig. 3. 9, p. 57

Review -Acids and Bases

Energy: Forms Ø Kinetic energy Ø Potential energy Ø Heat Sun High energy, short wavelength Low energy, long wavelength Nonionizing radiation Ionizing radiation Cosmic rays Gamma rays 10 -14 Wavelength in meters (not to scale) X rays 10 -12 Visible Far Near ultraviolet waves 10 -8 10 -7 10 -6 Near infrared waves 10 -5 Far infrared waves microwaves 10 -3 TV waves 10 -2 10 -1 Radio waves 1 Fig. 3. 10, p. 58

Energy: Quality Ø High-quality energy Ø Low-quality energy Electricity Very high temperature heat (greater than 2, 500°C) Nuclear fission (uranium) Nuclear fusion (deuterium) Concentrated sunlight High-velocity wind Very high-temperature heat (greater than 2, 500°C) for industrial processes and producing electricity to run electrical devices (lights, motors) High Mechanical motion (to move vehicles and other things) High-temperature heat (1, 000– 2, 500°C) for industrial processes and producing electricity Normal sunlight Moderate-velocity wind High-velocity water flow Concentrated geothermal energy Moderate-temperature heat (100– 1, 000°C) Wood and crop wastes Moderate-temperature heat (100– 1, 000°C) for industrial processes, cooking, producing steam, electricity, and hot water Dispersed geothermal energy Low-temperature heat (100°C or lower) Low High-temperature heat (1, 000– 2, 500°C) Hydrogen gas Natural gas Gasoline Coal Food Source of Energy Relative Energy Quality (usefulness) Low-temperature heat (100°C or less) for space heating Energy tasks Fig. 3. 11, p. 59

Physical and Chemical Changes Energy absorbed solid Melting Evaporation And boiling Freezing Condensation liquid Energy released Fig. 3. 5, p. 54 gas Reactant(s) Products(s) carbon + oxygen carbon dioxide + C + O 2 energy CO 2 + energy O C black solid O colorless gas O C O + energy colorless gas In-text, p. 59

The Law of Conservation of Matter Ø Matter is not consumed Ø Matter only changes form Ø There is no “away”

Matter and Pollution Ø Chemical nature of pollutants Ø Concentration Ø Persistence Ø Degradable (nonpersistent) pollutants Ø Biodegradable pollutants Ø Slowly degradable (persistent) pollutants Ø Nondegradable pollutants

Nuclear Changes Ø Natural radioactive decay Ø Radioactive isotopes (radioisotopes) Ø Gamma rays Ø Alpha particles Ø Beta particles Ø Half life Ø Ionizing radiation Sheet of paper Block of wood Concrete wall Alpha Beta Gamma Fig. 3. 12, p. 62

Nuclear Reactions Fission 92 Kr 36 235 U 92 n 235 U 92 Reaction Conditions Neutron + Hydrogen-2 or deuterium nucleus n n + n Hydrogen-3 or tritium nucleus 235 U 92 + 100 million ˚C + + Proton Neutron Fig. 3. 17, p. 64 + + Helium-3 nucleus Hydrogen-2 or deuterium nucleus + Hydrogen-2 or deuterium nucleus Energy Helium-4 nucleus + n n + D-D Fusion 235 92 U Products D-T Fusion 141 Ba 56 92 Kr n 36 141 Ba 56 Fig. 3. 16, p. 64 Fuel 235 U 92 n 92 Kr 36 n 141 Ba 56 n 141 Ba 56 n n 235 U 92 92 Kr 36 Fusion + 1 billion ˚C Energy Neutron

Fraction of original amount of plutonium-239 left 1 1/2 1/4 1/8 1 st half-life 2 nd half-life 3 rd half-life 0 240, 000 480, 000 Time (years) 720, 000 Fig. 3. 13, p. 62

Other 1% Consumer products 3% Radon 55% Nuclear medicine 4% Medical X rays 10% Space 8% The human body 11% Earth 8% Natural sources 82% Human-generated 18% Fig. 3. 14, p. 63

The First Ironclad Law of Energy First Law of Thermodynamics (Energy) Ø Energy is neither created nor destroyed Ø Energy only changes form Ø You can’t get something for nothing ENERGY IN = ENERGY OUT

Chemical Solar Chemical energy Mechanic energy (food) energy Waste heat (photosynthesis) (moving, thinking, living) Fig. 3. 18, p. 66

The Second Ironclad Law of Energy Second Law of Thermodynamics Ø In every transformation, some energy is converted to heat Ø You cannot break even in terms of energy quality

Connections: Matter and Energy Laws and Environmental Problems Ø High-throughput (waste) economy Ø Matter-recycling economy Ø Low-throughput economy Inputs (from environment) System Throughputs Output (intro environment) Low-quality heat energy High-quality energy Unsustainable high-waste economy Matter Waste matter and pollution Fig. 3. 19, p. 66 See Fig. 3. 20, p. 67
- Slides: 26