Basic Chemistry of Life Matter Energy Matter Anything

Basic Chemistry of Life

Matter & Energy �Matter � Anything that occupies space & has mass � Composes elements � C, H, O & N make up 96% of human mass � C, H, O, N, P & Ca make up 99% of human mass � Can undergo physical & chemical changes �Energy � The ability to do work – puts matter into motion � Kinetic Energy: energy doing work � Potential Energy: stored energy �All living things are built of matter & require energy to grow & function
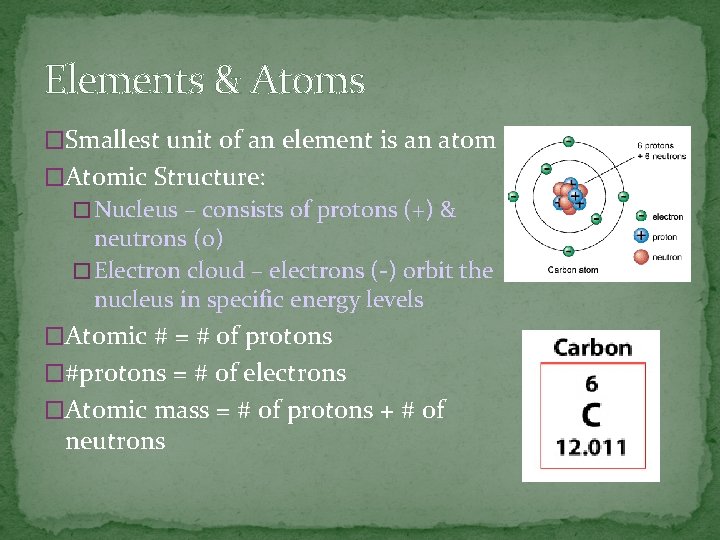
Elements & Atoms �Smallest unit of an element is an atom �Atomic Structure: � Nucleus – consists of protons (+) & neutrons (o) � Electron cloud – electrons (-) orbit the nucleus in specific energy levels �Atomic # = # of protons �#protons = # of electrons �Atomic mass = # of protons + # of neutrons

Molecules & Compounds �Molecule � Smallest unit of two or more combined atoms � Ex: H 2, C 6 H 12 O 6, O 2 �Compound � A molecule containing two or more different kinds of atoms � Ex: CH 4, H 2 O �Compounds have unique properties that are different from the elements that compose them.

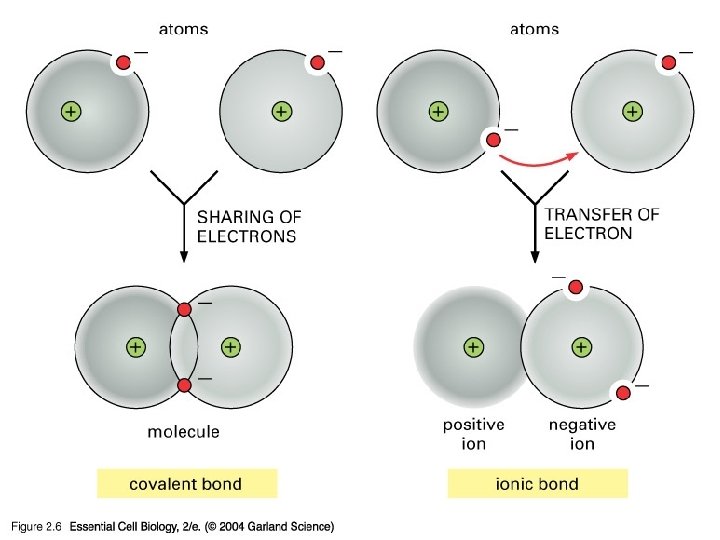
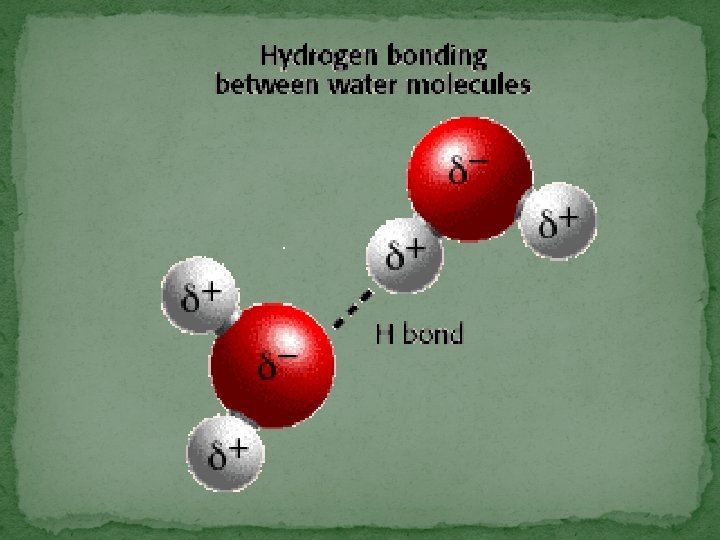
Chemical Bonds �The electrons are the part of the atom involved in bonding. �Types of Bonds: � Ionic Bonds �Electrons are transferred from one atom to another � Covalent Bonds �Pairs of valence electrons are shared between atoms. �Most bonds in living organisms are covalent � Hydrogen Bonds (weak bonds) �Provide temporary bonding between certain atoms in large complex molecules such as proteins & nucleic acids

Chemical Reactions �The formation & breakdown of compounds. � Synthesis Reaction (A + B = AB) � Decomposition Reactions (AB = A + B) � Exchange Reactions (AB + C = AC + B) �Metabolism – sum of all chemical reactions in the body. �Formation of bonds requires energy �Breaking of bonds releases energy

Types of Chemical Compounds �Inorganic Compounds � Lack carbon & contain ionic bonds � Resist decomposition � Ex: water, salts, & many acids & bases �Organic Compounds � Contain carbon & usually hydrogen � Covalently bonded � Ex: Carbohydrates, lipids, proteins, & nucleic acids �Both are equally essential to life!

Inorganic Compounds �Water � Most abundant substance in the body � Excellent solvent & suspension medium � Important in chemical reactions, absorbs & releases heat, lubricates, and protects �Acids, Bases, & Salts (electrolytes) � Form ions in water – Acids (H+) and Bases (OH-) � p. H of different parts of the body must remain constant � Buffer system eliminates excess H+ ions and OH- ions in order to maintain p. H homeostasis
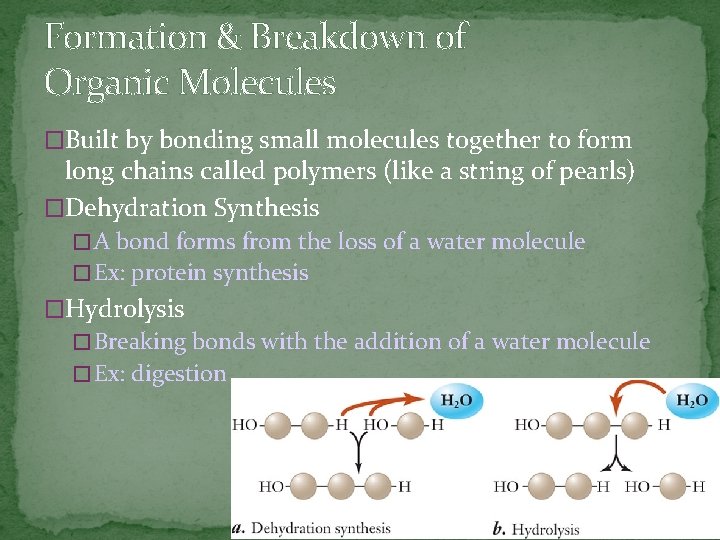
Formation & Breakdown of Organic Molecules �Built by bonding small molecules together to form long chains called polymers (like a string of pearls) �Dehydration Synthesis � A bond forms from the loss of a water molecule � Ex: protein synthesis �Hydrolysis � Breaking bonds with the addition of a water molecule � Ex: digestion

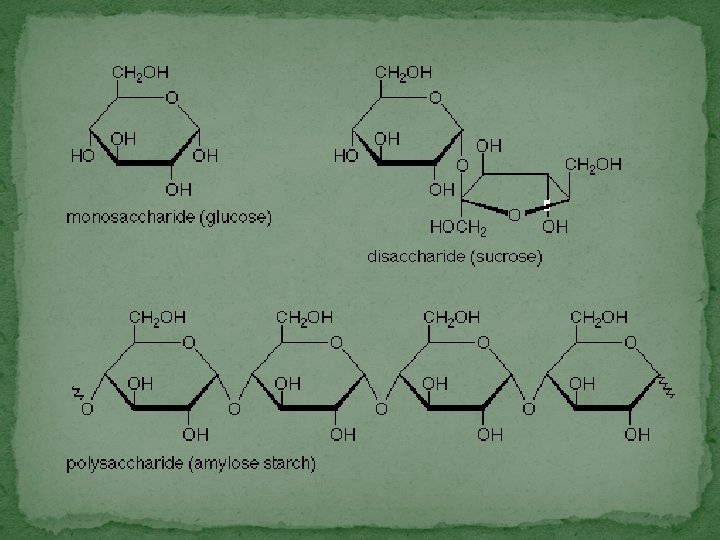
Organic Compounds: Carbohydrates �Sugars & starches composed of carbon, hydrogen, & oxygen �Store & release energy – 3 types � Monosaccharides – simple sugars �Building blocks of carbohydrates �Ex: glucose & fructose � Disaccharides – two simple sugars �Ex: sucrose, lactose, maltose � Polysaccharides – many sugars �Long branching chains of monosaccharides �Ex: starch & glycogen

Organic Compounds: Lipids �Fats, phospholipids, & steroids. �Composed of lots of carbon & hydrogen, with less oxygen. � C 57 H 110 O 6 �Protect, insulate, & provide long-term energy storage �Types: � Triglycerides � 3 fatty acids & glycerol � Phospholipids � 2 fatty acids, glycerol, & a phosphate group � Steroids – 4 rings (cholesterol)
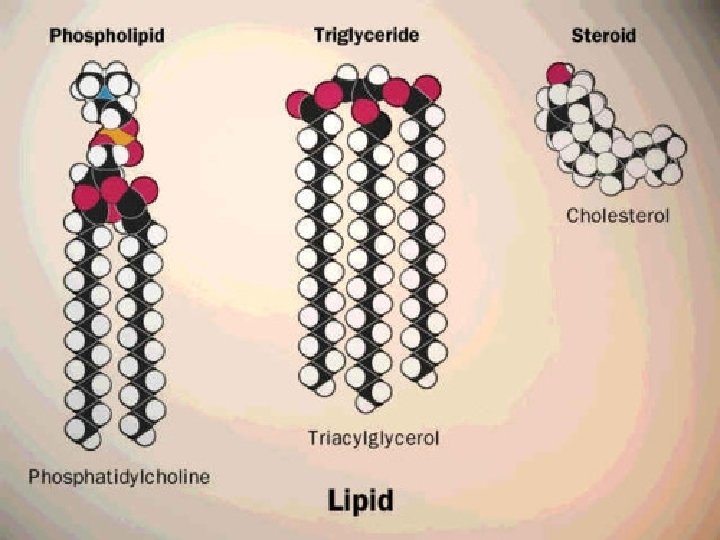

Lipids Continued 1. Saturated Fats – fatty acid chains of carbon with only single bonds � Generally solid at room temperature � Ex: butter & steak fat 2. Unsaturated Fats – fatty acid chains of carbon with double bonds � Generally liquid at room temperature (oils) � Ex: peanut oil, corn oil, olive oil, etc…

Organic Compounds: Proteins � 50% of the organic matter in the body �Contain C, H, O, N, & sometimes S �Composed of long chains of amino acids �Uses: � Make up cell membranes � Build structures (like muscles) � Carry out metabolic processes (enzymes carry out chemical reactions) � Defensive of the body �There are 1000 ths of different proteins each with a unique shape & function. � Ex catalase, lactase, amylase, hemoglobin, actin, etc…

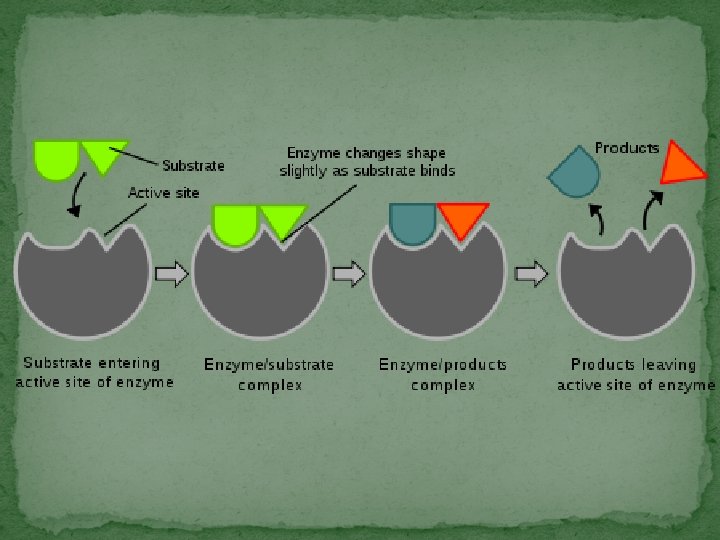
Enzymes & Substrates �Proteins that speed-up chemical reactions � Enzymes are specific for the reactant they work with (like a lock & a key). � Depends on the enzyme’s shape �The reactant that an enzyme works with is called its substrate � Only one type of substrate will fit each enzyme �The substrate fits into a region of the enzyme called the active site.

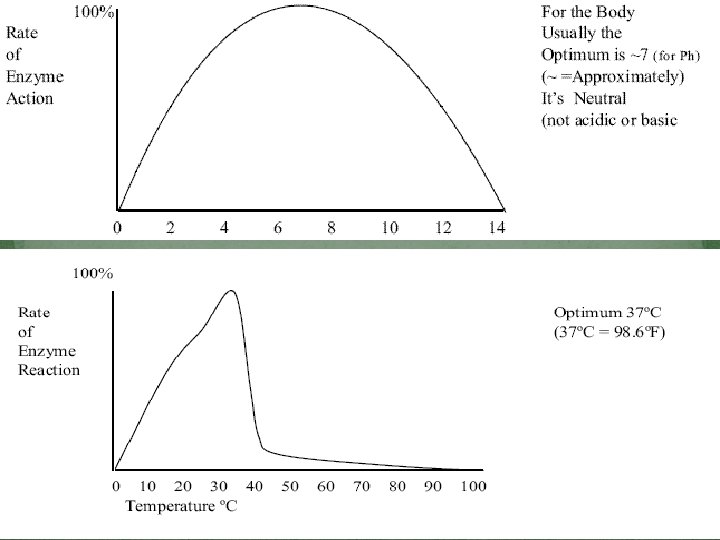
Factors Affecting Enzymes 1. Temperature � 2. p. H � 3. There is an optimum p. H for enzyme reactions Amount of enzymes � 4. There is an optimum temperature for enzyme reactions The more enzymes available the faster the reaction Amount of substrate � The more substrate available the faster the reaction � Changes in temperature or p. H may cause enzymes to become “denatured” � Changes enzymes shape & they can no longer bind to the substrate…will render the enzymes functionless

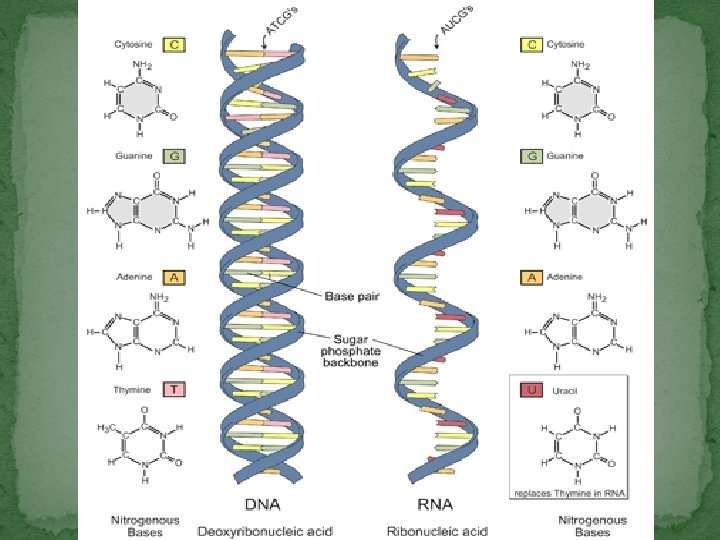
Nucleic Acids �Organic compound composed of Nucleotides � Sugar, phosphate, & nitrogen base � Types: DNA & RNA �DNA – double helix � Organism’s information code �RNA – single helix � Involved in protein synthesis
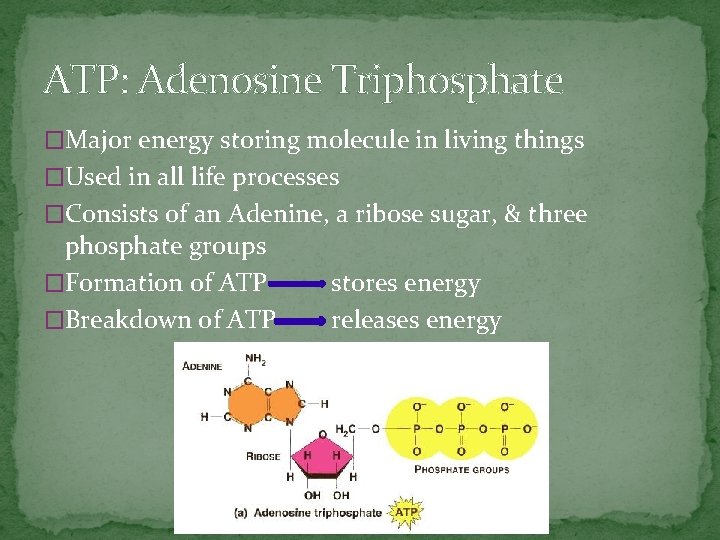
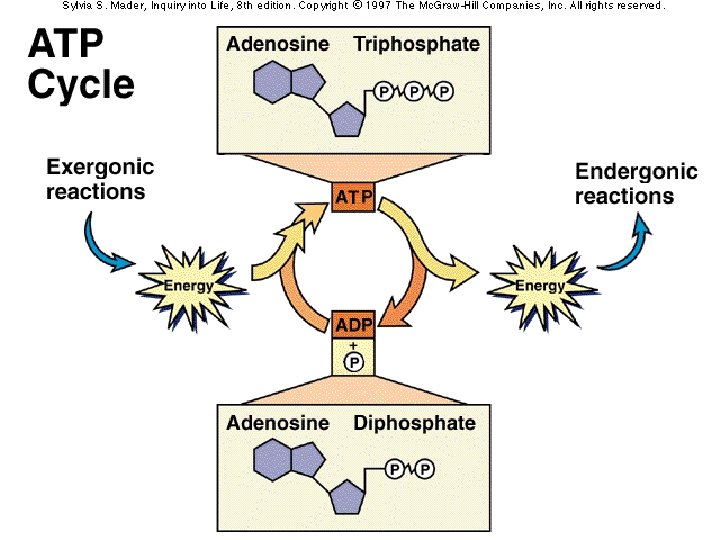
ATP: Adenosine Triphosphate �Major energy storing molecule in living things �Used in all life processes �Consists of an Adenine, a ribose sugar, & three phosphate groups �Formation of ATP – �Breakdown of ATP – stores energy releases energy
- Slides: 25