Basic Chemistry in the Human Body Matter matter
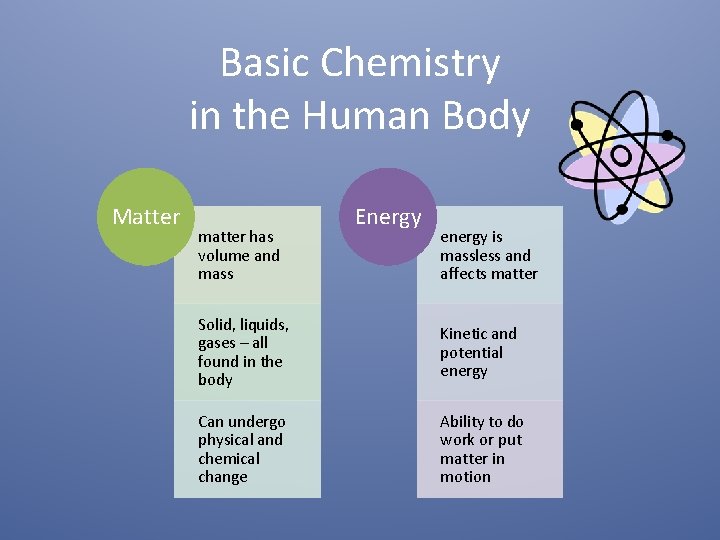
Basic Chemistry in the Human Body Matter matter has volume and mass Energy energy is massless and affects matter Solid, liquids, gases – all found in the body Kinetic and potential energy Can undergo physical and chemical change Ability to do work or put matter in motion

Forms of Energy Chemical: energy stored in chemical bonds • body uses chemical energy in foods Electrical: energy resulting from movement of charged particles (electrons) • body generates electrical current across charged particles Mechanical: energy involved in moving matter • muscles shorten pulling on bones causing limbs to move Radiant: energy travelling in waves • light energy stimulates eyes, UV rays stimulate body to make vitamin D
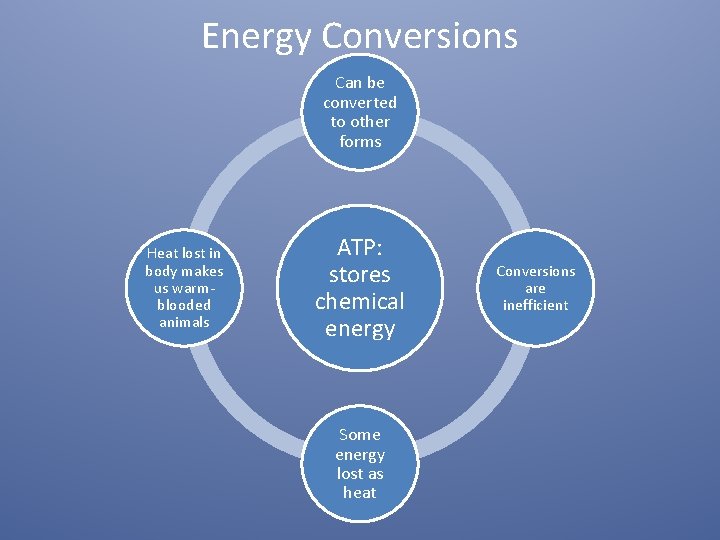
Energy Conversions Can be converted to other forms Heat lost in body makes us warmblooded animals ATP: stores chemical energy Some energy lost as heat Conversions are inefficient

Common Elements in the Body Oxygen • 65% of body mass • In organic and inorganic molecules • Needed to oxidize glucose during ATP production Carbon • 18. 5% of body mass • Primary component of all organic molecules such as carbohydrates, lipids, proteins, and nucleic acids Hydrogen • 9. 5% of body mass • In most organic molecules • Influences p. H of body fluids Nitrogen • 3. 2% of body mass • Component of proteins and nucleic acids Other Elements • Calcium: in bones and teeth, muscle contraction, neural transmission, & blood clots • Phosphorus: in bones and teeth, nucleic acids, and proteins, needed for ATP • Potassium: needed for conduction of nerve impulses and muscle contraction
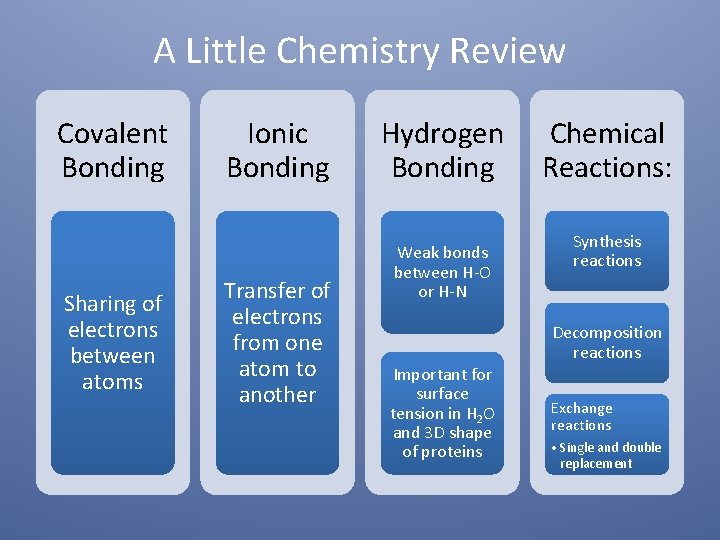
A Little Chemistry Review Covalent Bonding Sharing of electrons between atoms Ionic Bonding Transfer of electrons from one atom to another Hydrogen Bonding Weak bonds between H-O or H-N Important for surface tension in H 2 O and 3 D shape of proteins Chemical Reactions: Synthesis reactions Decomposition reactions Exchange reactions • Single and double replacement

Two Types of Compounds Organic Compounds Inorganic Compounds • Contain carbon • Carbohydrates • Lipids • Proteins • Nucleic Acids • Lack carbon and tend to be simpler & smaller • Water • Salts • Acids • Bases

Inorganic Compounds Water & its functions Most abundant inorganic compound in the body • Approx. 2/3 of entire body weight High heat capacity • Absorbs and releases lots of heat without changing temperature drastically • This helps prevent sudden changes in body temperature Is Polar • Polarity makes it an excellent solvent • allows water to act as transport medium within the body Chemical Reactivity • Water is important to many chemical reactions within the body Cushioning • Acts as a protective cushion in the body • Cerebrospi nal fluid • Amniotic fluid

Inorganic Compounds Cont. Salts & their functions Calcium and phosphorus salts are most common in the body Salts are vital to body function Salts are electrolytes • Sodium and potassium ions from salts are necessary nerve impulses • Iron needed to form hemoglobin in the blood • Conduct electrical current in solution • When electrolytes out of balance the body struggles to function properly

Inorganic Compounds Cont. ACIDS • Substance that can release H+ ions (proton donors) • Have a sour taste BASES • Proton acceptors • Hydroxides and bicarbonates are common in the body • Have a bitter taste Acids and Bases react with each other to form water and a salt – neutralization Cells are very sensitive to slight p. H changes Kidneys, lungs, and buffers are important to maintaining acid-base balance Ex. Normal blood p. H 7. 35 -7. 45, if dips too much hemoglobin unable to carry enough oxygen and can be fatal (condition known as acidosis)

Organic Compounds Carbohydrates Includes sugars and starches • Contain C, H, O usually following Cn. H 2 n. On ratio • Provide easy to use source of food energy for body’s cells • Energy released from breakdown of carbs can be stored in ATP or as fat Monosaccharides Disaccharides Polysaccharides • Simple sugars with 3 -7 carbons • Includes glucose, fructose, galactose, ribose, & deoxyribose • Made of 2 simple sugars • Includes sucrose, lactose, & maltose • Must be broken down by hydrolysis before being absorbed into the body • Made of long chains of simple sugars • Includes starches and glycogen

Organic Compounds cont. Lipids Contain C, H, O with C & H far more numerous than O (ex. C 57 H 110 O 6 ) • Found in meat, egg yolk, milk products, and oils • Insoluble in water Neutral Fats (triglycerides) • made of 3 fatty acids attached to glycerol • Found in fat deposits around body • Protect and insulate body organs • Major source of stored energy Phospholipids • Similar to neutral fats but contain phosphorous • Found in cell membranes allowing membrane to be selectively permeable • Help form white matter of brain and nervous tissue Steroids • Cholesterol • In cell membranes, used to make other steroids and hormones • Bile salts • From liver, needed in digestion • Vitamin D • Normal bone growth • Sex hormones • Estrogen/progesterone • Testosterone

Organic Compounds cont. Proteins Contain C, H, O, N, and sometimes S • Made of building blocks called amino acids • Classified as fibrous or globular Fibrous Proteins Globular Proteins • Important for binding structures together and giving strength • Ex. Collagen in bones, cartilage, and tendons • Ex. Keratin in hair, nails, and skin • Also called functional proteins • Antibodies • aka: immunoglobulins • Function in immune response to protect body from invading substances • Hormones • Regulate growth and development • Ex. Growth hormone, insulin, nerve growth factor • Transport proteins • Ex. Hemoglobin in blood transports oxygen • Enzymes • Act as catalysts to almost all reactions within the body

Organic Compounds cont. Nucleic Acids Make up genes in chromosomes Deoxyribonucleic Acid (DNA) • Direct growth and development by dictating the structure of proteins • Contain C, H, O, N, P • Made up of nucleotides containing a nitrogenous base, a pentose sugar, and a phosphate group • Nitrogenous bases include adenine, guanine, cytosine, thymine, and uracil • Genetic material in cell nucleus • 2 major functions • Replicates itself before cell division to ensure all genetic information in all cells is identical • Provides instructions for building every protein in the body • Double strand of nucleotides known as a double helix • Contains deoxyribose sugar Ribonucleic Acid (RNA) • Found outside the nucleus • “molecular slave” of DNA • Carries out protein synthesis as ordered by DNA • Single strand of nucleotides • Messenger RNA • Carries DNA’s instructions from nucleus to ribosomes • Ribosomal RNA • Oversees binding together of amino acids in ribosomes during protein synthesis • Transfer RNA • Brings amino acids to ribosomes
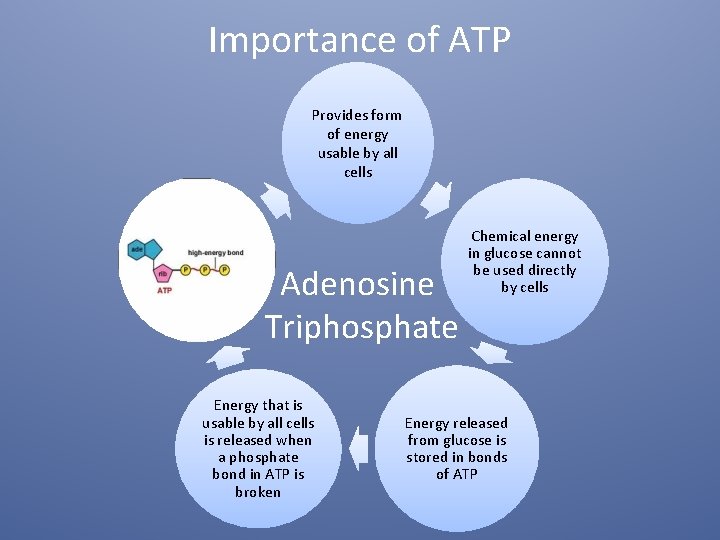
Importance of ATP Provides form of energy usable by all cells Adenosine Triphosphate Energy that is usable by all cells is released when a phosphate bond in ATP is broken Chemical energy in glucose cannot be used directly by cells Energy released from glucose is stored in bonds of ATP
- Slides: 14