Basic Chemistry Chemistry Chemistry is the science of

Basic Chemistry

Chemistry: • Chemistry is the science of matter. – Scientists study chemicals, their properties, and REACTIONS (rxn). • BIOCHEMISTRY is the chemistry of living things.

Water: • most chemical rxns occur only in water (universal solvent) • 90% or more of cells is H 2 O • essential to life • Solutions are made up of the – Solute: – Solvent:

Elements • most simple substance of matter • cannot be broken down • ex. Fe. O, Mg, Na
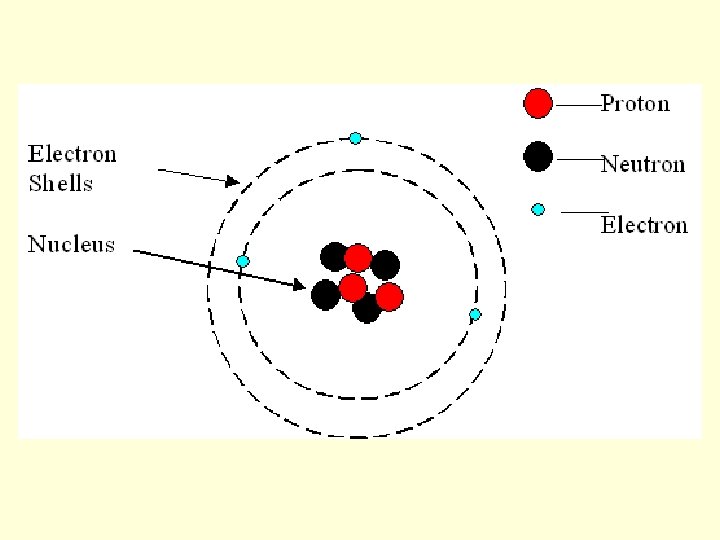

Valence Electrons: • outermost electrons • (carry negative charge) • responsible for bonding • goal: to have a full valence energy level
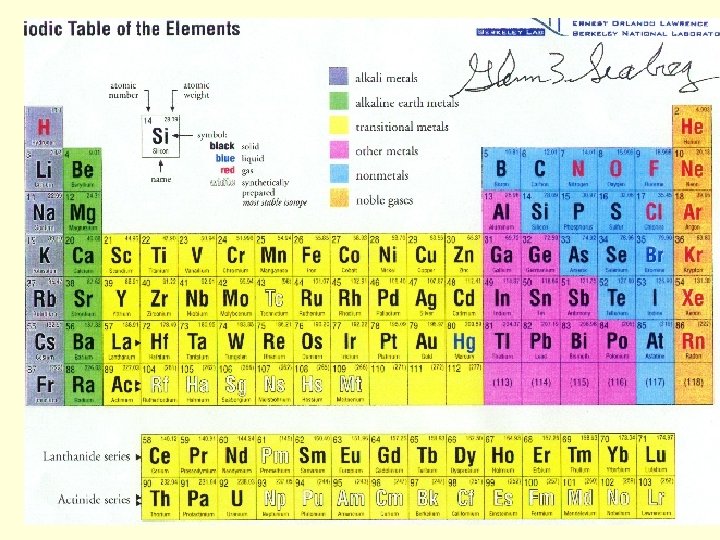

Boxes on the Periodic Table • Each box on the PT contains the following information: – Atomic number = – Atomic Mass = – Atomic Symbol = – # of protons = – #of neutrons = – # of electrons = 6 C 12. 001

Compound • Chemically bonding of 2 or more elements formed by: 1. 2. 3. 4. Ionic Bonds: Covalent Bonds Polar Bonds Hydrogen Bonds
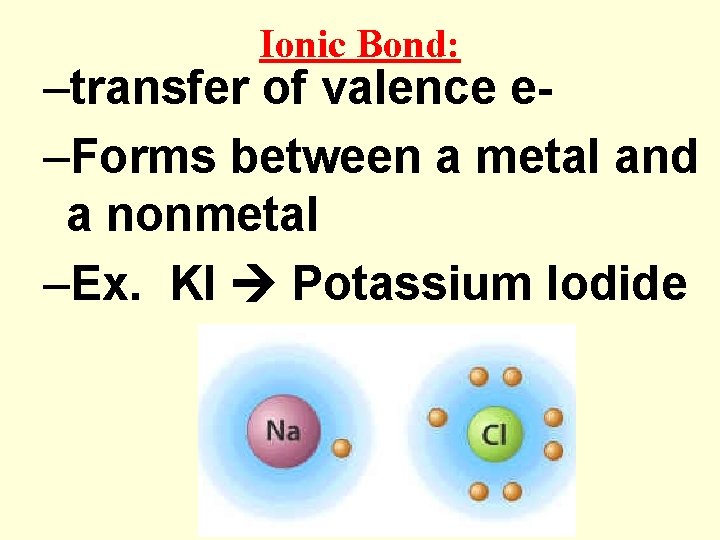
Ionic Bond: –transfer of valence e–Forms between a metal and a nonmetal –Ex. KI Potassium Iodide
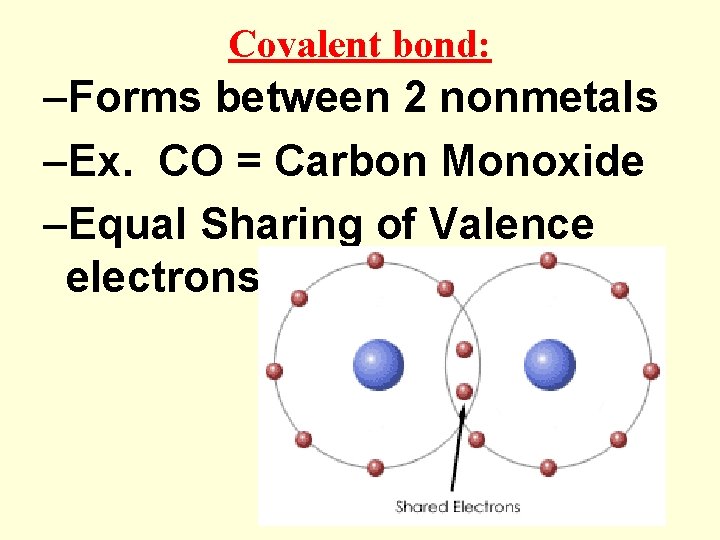
Covalent bond: –Forms between 2 nonmetals –Ex. CO = Carbon Monoxide –Equal Sharing of Valence electrons

Polar Bonds • between covalent and ionic bonds • unequal sharing of the valence electrons • have slightly positive and slightly negative ends • Ex. Water!!

Adhesion and Cohesion • Water is attracted to other water. – This is called cohesion. • Water can also be attracted to other materials. – This is called adhesion.

Surface Tension • the cohesion of water molecules at the surface of a body of water
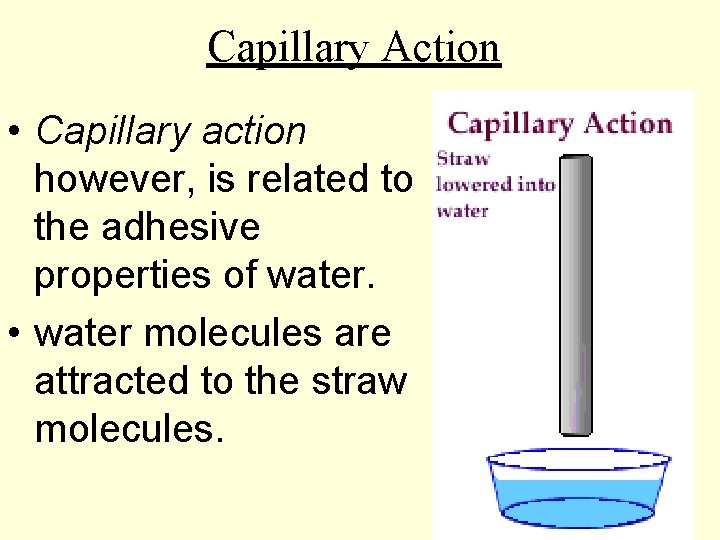
Capillary Action • Capillary action however, is related to the adhesive properties of water. • water molecules are attracted to the straw molecules.

Hydrogen Bonds : –Weaker bonds than ionic and covalent –Allow for large biological molecules • Ex. Proteins • DNA –Bonds water molecules together

Acid and Bases: • Are ionized • Can conduct electrical current • Located throughout the body – Ex. Mouth, stomach

Acid : Acids: • Have extra H+ ions • Have a sour taste • Have a low p. H (between 1 – 5)

Bases: • Have OH- ions • Have a slippery feel • Have a high p. H (b/w 8 -12)
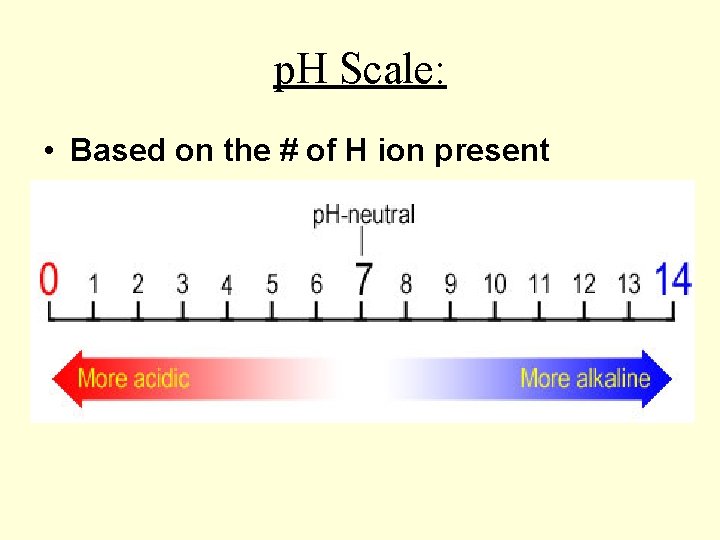
p. H Scale: • Based on the # of H ion present

Buffers: • keep its p. H constant • resist changes in p. H when acids or bases are added to them. • In the body proteins act as a buffer, ex. Blood

Neutralization: • Combing an Acids with a base • results is neither an acid or a base. • H+ form the acid and the OH- form the base combine to form water • Ex. HCl + Na. OH Na. Cl +H 2 O acid + base salt + water

Acid , Base Indicators: • Phenolphthalein (base only) • p. H paper (both) • Universal Indicator Solution (Both) • Litmus paper (either) – Red – Blue

Litmus paper:

p. H paper:

Universal Indicator Solution:

Phenolphthalein: Clear in the presence of an acid Pink in a base

Inorganic chemistry: • compound usually does not contain carbon or hydrogen

Organic Chemistry: • the compounds usually found in Living or once living things • contains carbon and hydrogen • most common elements in living things are: • C, H, O, N, P • Carbon is often referred to as the element of LIFE ***CHANGES ON THIS SLIDE***
- Slides: 29