Basic Chemistry and Microbiology Review of Basic Chemistry

Basic Chemistry and Microbiology

Review of Basic Chemistry � smallest basic particle is the atom › Electrons- negatively charged › Protons- positively charged in nucleus › Neutrons- uncharged in nucleus � when electrons are lost or gained, a charge occurs � substances containing only one kind of atom are called elements

� approximately 20 elements are found in all living things � carbon, hydrogen, oxygen, and nitrogen make up 97% � the other 16 are called trace elements � Molecules are when two or more atoms are joined together (Ex: O , CO ) 2 2

Compounds and Solutions � Two or more atoms or molecules joined in a definite proportion by weight is called a compound � Compounds have different characteristics from elements they are made from � Represented by a formula

Compounds and Solutions � Two or more atoms or molecules joined in a definite proportion by weight is called a compound � Compounds have different characteristics from elements they are made from � Represented by a formula

Compounds and Solutions � Types of Compounds › Inorganic �Do not contain carbon �Often has a metal as a positive ion › Organic �Found in living things �Always contain carbon �When with hydrogen they are called hydrocarbons (usually gases) �When with other carbons, they bond in chains

Compounds and Solutions � Solutions › Chemical process take place in solutions › A solution is when one substance dissolves into another › The solute is dissolved into the solvent › If it dissolves it is soluble, if not, insoluble › Note: in microbiology, a tincture is alcohol and some other substance

Carbohydrates � All have carbon (C), hydrogen (H), and oxygen (O) � Occur in ration of 1: 2: 1
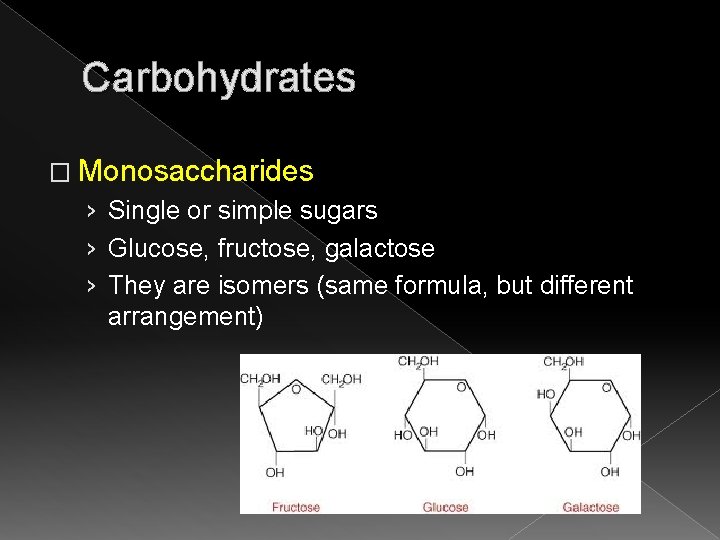
Carbohydrates � Monosaccharides › Single or simple sugars › Glucose, fructose, galactose › They are isomers (same formula, but different arrangement)

Carbohydrates � Glucose (also known as dextrose) › Carried in bloodstream › Combines with oxygen (oxidation) and produces adenosine triphosphate (ATP) � Fructose (found in fruits and honey) › Sweetest of all monosaccharides � Galactose › Found in small amounts in agar, flaxseed, and milk NOTE: “ose” means sugar

Carbohydrates � Disaccharides › Known as a double sugar › Examples: sucrose(table sugar), lactose (milk sugar)and maltose (malt sugar) � Chemical reaction to join: › Dehydration synthesis (opposite to break apart is hydrolysis)
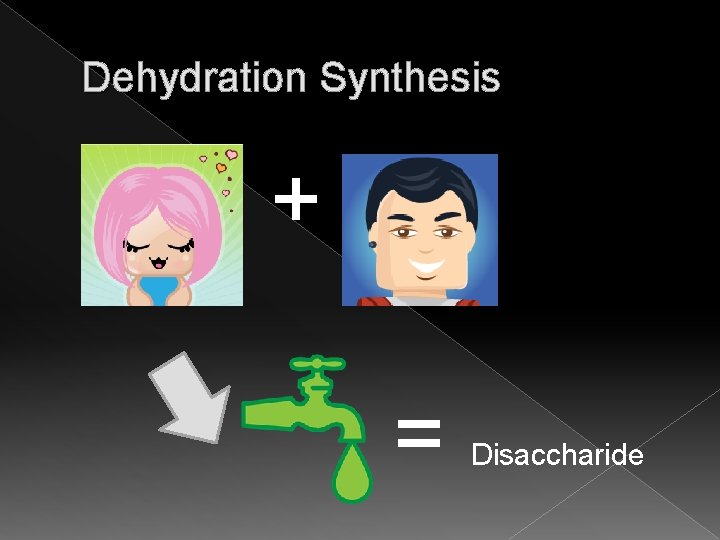
Dehydration Synthesis + = Disaccharide
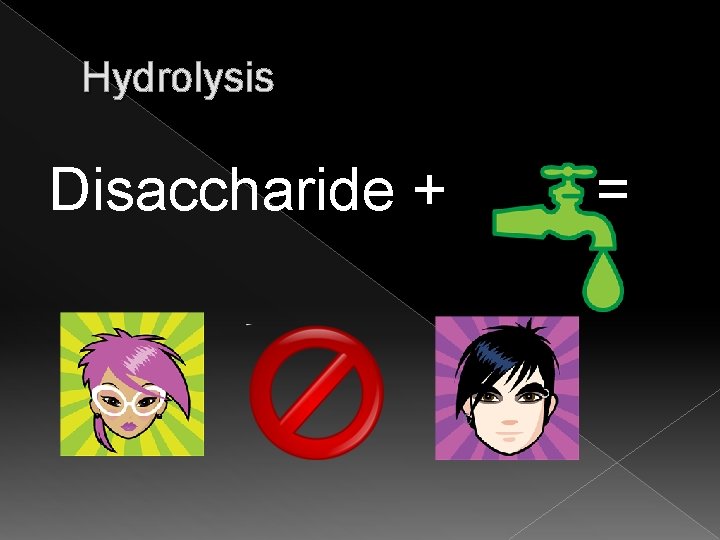
Hydrolysis Disaccharide + =

Carbohydrates � Oligosaccharides › Form chains called polymers › Small chains with only 2 -10 monosaccharides › Ex: insulin � Polysaccharides › Large, complex molecules › Made of hundreds of thousands of glucose › Have very long polymer chains › Ex: starch, cellulose, and glycogen

Carbohydrates � https: //www. youtube. com/watch? v=M 6 ZLDJl uj 6 I � http: //www. khanacademy. org/partner- content/crash-course 1/crash-coursebiology/v/crash-course-biology-103

Lipids � Examples are fats, oils, and waxes � Like carbs, they have carbon, hydrogen, and oxygen, but much less oxygen � Fats are solid and oils are liquid at room temperature � Better sources of energy than carbohydrates (yield more energy) but are harder to oxidize � Three groups: simple lipids, compound lipids, and derived lipids
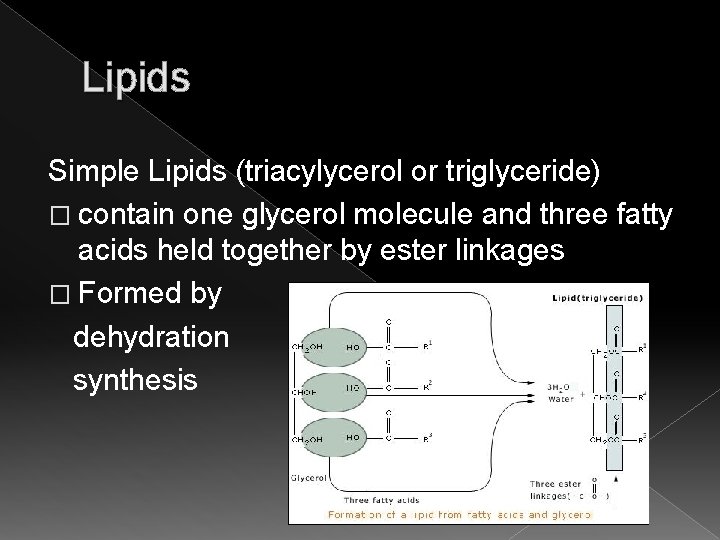
Lipids Simple Lipids (triacylycerol or triglyceride) � contain one glycerol molecule and three fatty acids held together by ester linkages � Formed by dehydration synthesis

Lipids Simple Lipids � Saturated (when all carbon bonds are single and saturated with hydrogen) › › Can block arteries Difficult to break up Raises cholesterol Solid at room temperature

Lipids Simple Lipids � Unsaturated (when two or more hydrogen bonds are replaced with double bonds between carbon atoms) › Liquid at room temperature › Monounsaturated (lacks 2 hydrogen bonds) or Polyunsaturated (lacks 8 or more hydrogen bonds forming 4 or more double bonds) › Lowers Cholesterol › Easier to break up

Lipids � Trans Fats › Unsaturated fat but act like a saturated fat › Has trans arrangement in bonding- the hydrogens are on opposite sides of the double bond. › Typical man-made
Lipids https: //www. youtube. com/watch? v=3 x. F_LK 9 pn L 0 https: //www. youtube. com/watch? v=VGHD 9 e 3 y RIU

Lipids � Compund lipids- lipids which contain an inorganic or organic group in addition to fatty acids and glycerol. › Phosphpholipids- Lipids containing a phosphate group. A phospholipid molecule has a strongly nonpolar and hydrophobic (water insoluble) tail region represented by fatty acid chains and a strongly polar or hydrophilic (water soluble) head region represented by the phosphate group.
Lipids › Glycolipids- These are lipids containing a carbohydrate group, usually galactose. They are found in the nerve cell membranes especially in the myelin sheath.
Lipids › Lipoproteins- These are lipids, usually phospholipids which contain a protein molecule. They occur in the cell membrane. They are also found in milk and egg yolk

Lipids � Derived Lipids (Sterols) › Contain only carbon, hydrogen, and oxygen › Include steroids found in male and female hormones, Vitamin D, cholesterol, and fat soluble vitamins A, E, and K › Classified as lipids only because they are soluble in fat solvents › These are lipids that do not have a straight chain. They are composed of four fused carbon rings and a long hydrocarbon side chain.
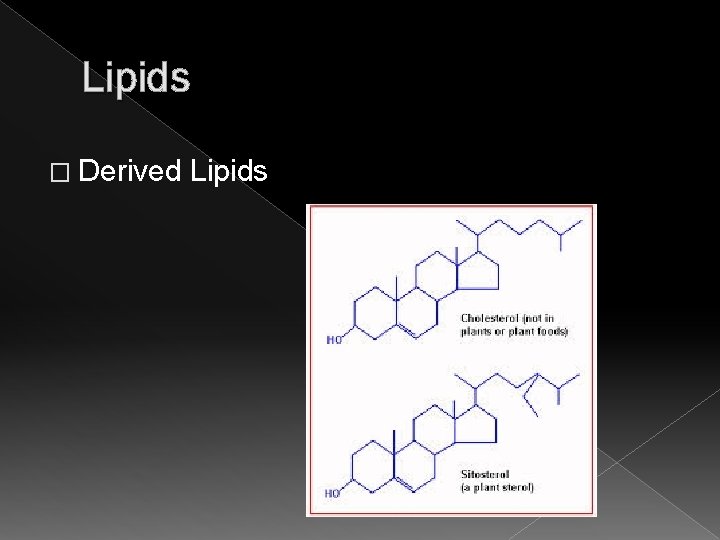
Lipids � Derived Lipids

Proteins � Contain hydrogen, oxygen, and nitrogen and most times phosphorus and sulfur � Found in every part of living cells � Coat viruses � In binding and structural components like fingernails, hair, ligaments, muscles, etc. � Made of AMINO ACIDS
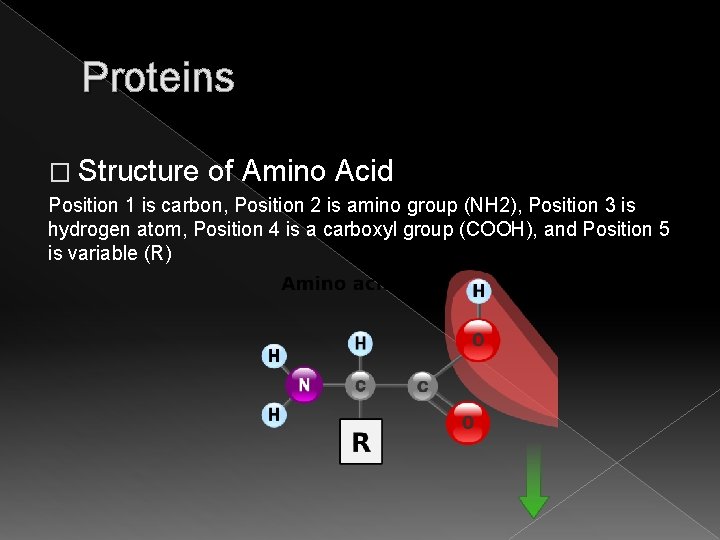
Proteins � Structure of Amino Acid Position 1 is carbon, Position 2 is amino group (NH 2), Position 3 is hydrogen atom, Position 4 is a carboxyl group (COOH), and Position 5 is variable (R)
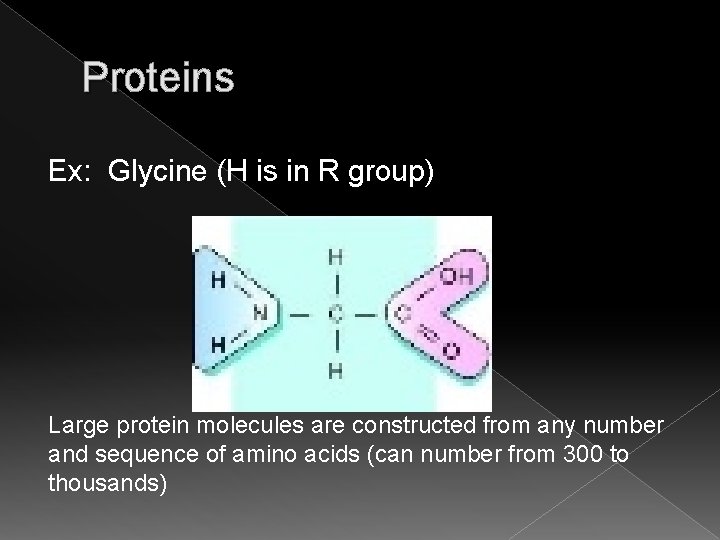
Proteins Ex: Glycine (H is in R group) Large protein molecules are constructed from any number and sequence of amino acids (can number from 300 to thousands)

Proteins � Amino acids linked by dehydration synthesis (C-N) (bond between carboxyl group of one amino acid and amino group of next amino acid). Called a PEPTIDE BOND and a series of linkages is called a POLYPEPTIDE � THIS IS CALLED A PROTEIN

Proteins � Structure › › Primary- straight chain Secondary- helix Tertiary- twisted and folded Quaternary- two or more polypeptide chains are bonded together
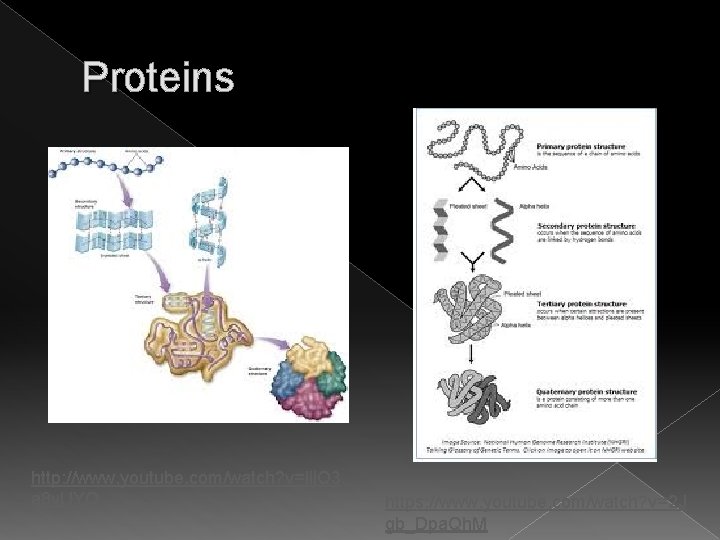
Proteins http: //www. youtube. com/watch? v=lij. Q 3 a 8 y. UYQ https: //www. youtube. com/watch? v=2 J gb_Dpa. Qh. M

Proteins Functions: � Enzymes - proteins that allow chemical reactions to occur in living things � Antibodies – proteins that protect the body from infection � Structure – cytoskeleton, hair, nails, muscles, spider web, silk, feathers , horns, hooves etc…. � Hormones – chemical messengers � Cell membrane – proteins can act as channels through the cell membrane - receptor proteins found on membrane transmit signals to the inside of cells � Hemoglobin – protein found in blood that carries oxygen

Proteins
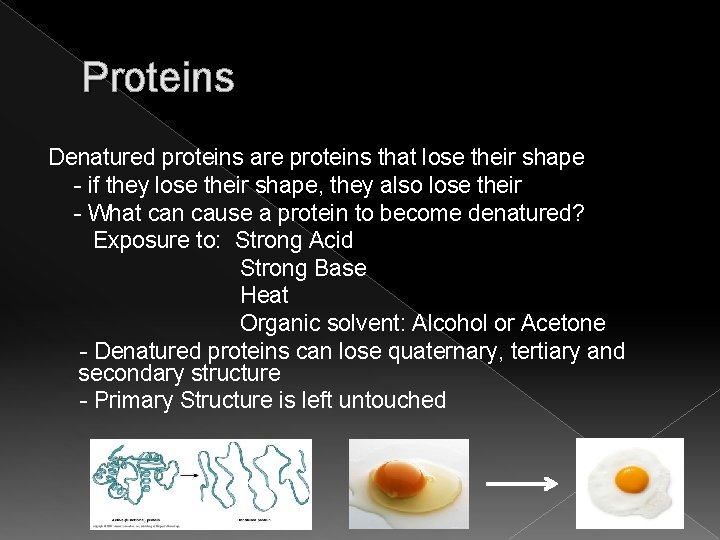
Proteins Denatured proteins are proteins that lose their shape - if they lose their shape, they also lose their - What can cause a protein to become denatured? Exposure to: Strong Acid Strong Base Heat Organic solvent: Alcohol or Acetone - Denatured proteins can lose quaternary, tertiary and secondary structure - Primary Structure is left untouched

Proteins (enzymes) � Specialized proteins � Help provide energy to cell at just the right moment and at just the right speed � Also known as organic catalysts � Highly specific � Very large and complex

Proteins (enzymes) � Made of either all protein or part protein (apoenzyme)attached to a non-protein part (coenzyme) � Coenzymes could be calcium, iron, magnesium, copper, or vitamins like C and B -complex

Proteins (enzymes) � The localized site on the enzyme molecule is called the active site � Each enzyme has its own pattern on the active site (no 2 alike) � An enzyme reacts with a reactant whose molecular pattern fits the enzyme’s molecular pattern. � The molecule that the enzyme reacts with is called a SUBSTRATE molecule
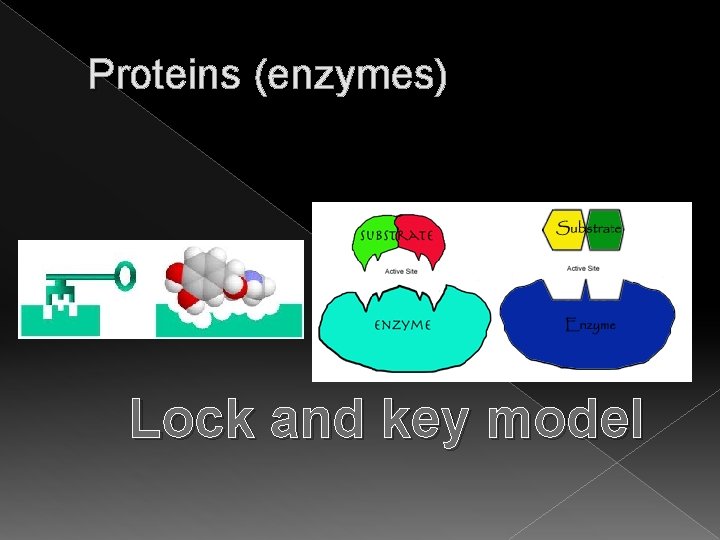
Proteins (enzymes) Lock and key model

Proteins (enzymes) � Temporary physical binding called Enzyme-Substrate Complex

Proteins (enzymes) Enzymes are proteins and if they are exposed to extremes of temp or p. H lose their shape � - if a protein loses its shape, it loses its function � - a protein that loses its shape is said to be denatured � - if an enzyme is denatured, substrate cannot enter the active site
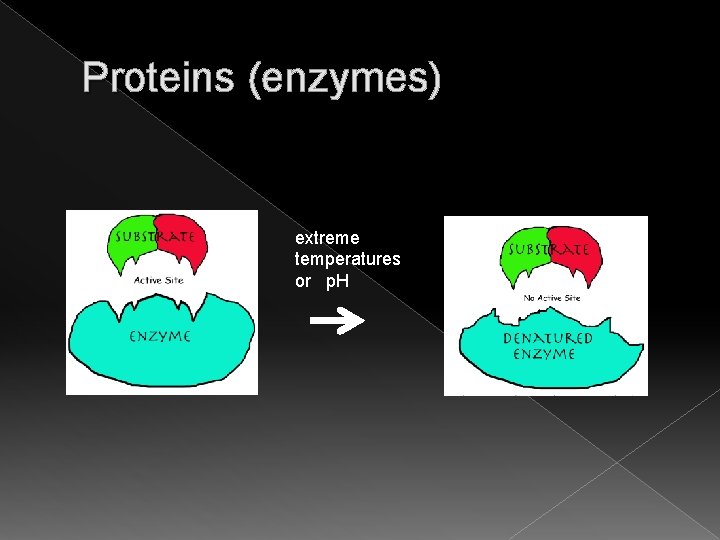
Proteins (enzymes) extreme temperatures or p. H

Proteins (enzymes) � Name usually ends in –ASE � Added to stem word taken from substrate � Examples: › › › Lactase……lactose Lipase……. . . lipids Maltase……maltose Protease…. . . protein Sucrase……. sucrose

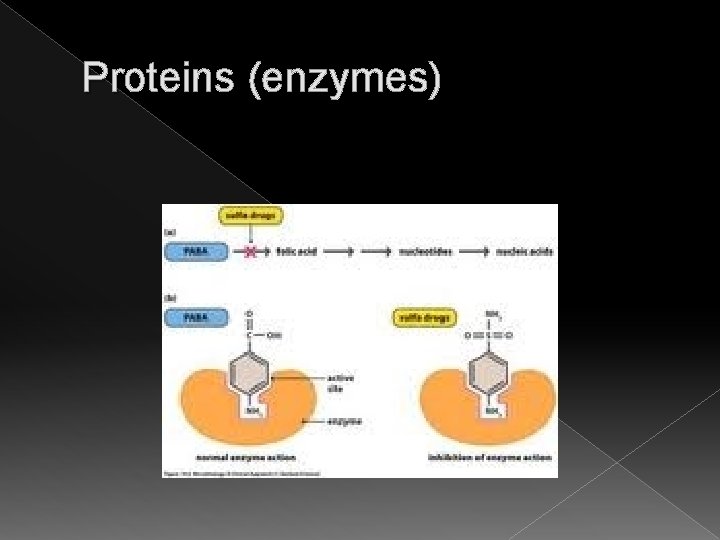
Proteins (enzymes) An example in microbiology: Many bacteria have an enzyme that needs a compound called PABA (para-aminobenzoic acid). PABA helps bacteria make a vitamin called folic acid, which the bacteria need to grow.
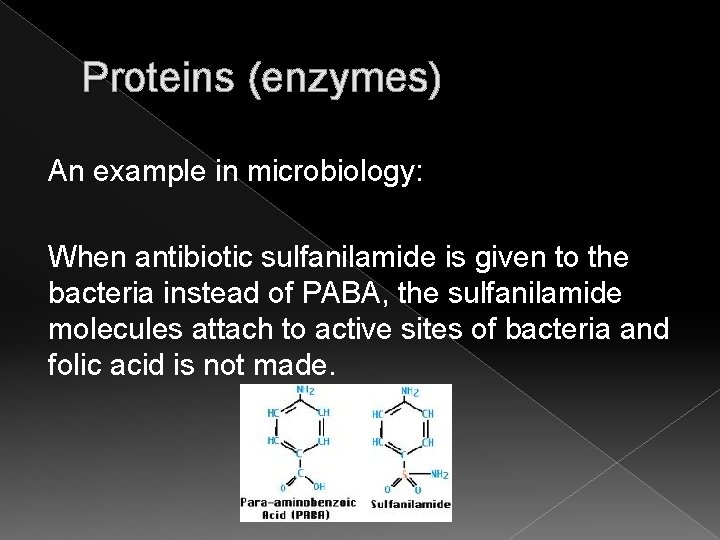
Proteins (enzymes) An example in microbiology: When antibiotic sulfanilamide is given to the bacteria instead of PABA, the sulfanilamide molecules attach to active sites of bacteria and folic acid is not made.
Proteins (enzymes)

Nucleic Acids � Contain carbon, oxygen, hydrogen, nitrogen, and phosphorous � Two types- DNA and RNA

Nucleic Acids � DNA › Found in chromosomes and genes, plasma membrane, mitochondria, and chloroplasts › Consists of a phosphate group, deoxyribose sugar and one of four nitrogenous bases: adenine, thymine, cytosine, and guanine

Nucleic Acids

Nucleic Acids � RNA › Consists of a phosphate group (ribose sugar), and any of the following nitrogenous bases: adenine, cytosine, guanine, and uracil › Single stranded › Found in cytoplasm, nucleoli, and ribosomes › Two kinds: m. RNA and t. RNA
- Slides: 50