Basic Chemistry A basic understanding of chemistry is
Basic Chemistry A basic understanding of chemistry is extremely important to understanding biology and biological systems -- organisms, their tissues and their cells are ultimately composed of atoms and molecules and many important biological processes are chemical reactions -- e. g. metabolism, photosynthesis, growth.

Chemistry organization levels • Molecule - 2 or more atoms bonded together • Atom - smallest unit of an element • Subatomic Particle - proton, electron, neutron Go to PBS Try It, you will need Shockwave Atom builder http: //www. pbs. org/wgbh/aso/tryit/atom/

terms I am going to assume that you have had no chemistry -- we will begin at the beginning and just cover some very basic chemistry. • The atom is the basic chemical unit. Composed of 2 parts: 1. nucleus -- contains subatomic protons (+ charge) and neutrons (no charge). 2. electrons contains subatomic protons (+ charge) and neutrons (no charge). 3. Protons (+ charge)- located in the nucleus

2. 1 Atoms • Organisms are chemical machines – one must know chemistry in order to understand biology • Any substance in the universe that has mass and occupies space is comprised of matter – all matter is made up of atoms

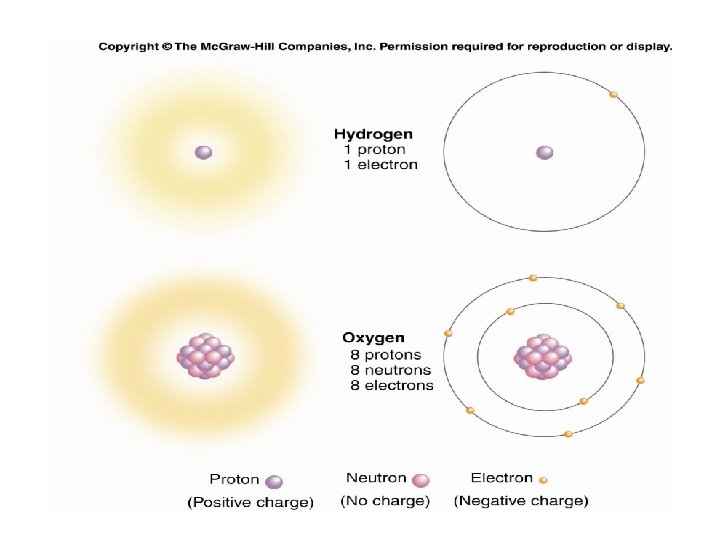
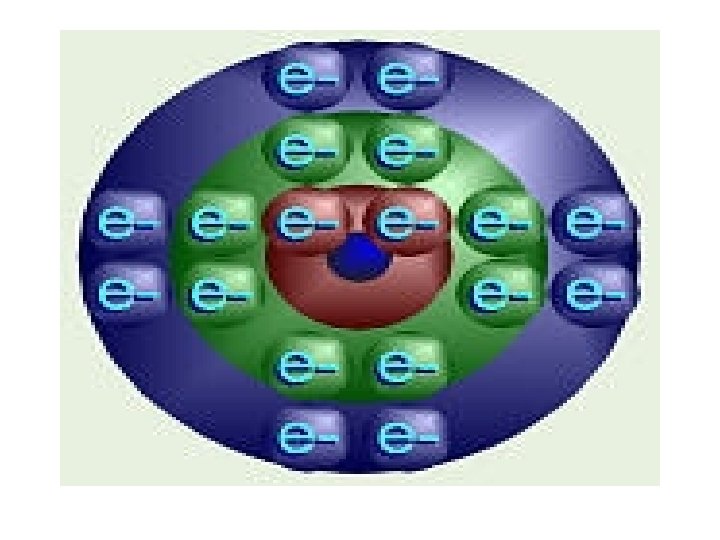
2. 1 Atoms • All atoms have the same structure – at the core is a dense nucleus comprised of two subatomic particles • protons (positively charged) • neutrons (no associated charge) – orbiting the nucleus is a cloud of another subatomic particles • electrons (negatively charged)
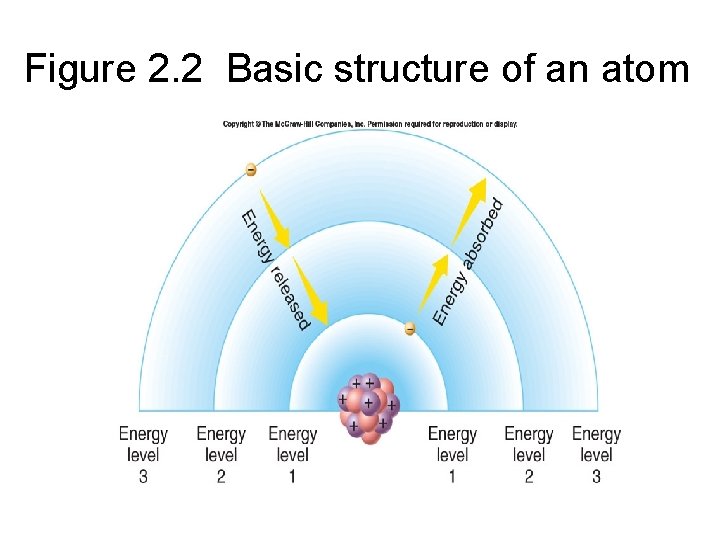
Figure 2. 2 Basic structure of an atom

2. 1 Atoms • An atom can be characterized by the number of protons it has or by its overall mass – atomic number • the number of protons in the nucleus • atoms with the same atomic number exhibit the same chemical properties and are considered to belong to the same element – mass number • the number of protons plus neutrons in the nucleus • electrons have negligible mass

2. 1 Atoms • Electrons determine the chemical behavior of atoms – these subatomic components are the parts of the atom that come close enough to each other in nature to interact

2. 1 Atoms • Electrons are associated with energy – electrons have energy of position, called potential energy – Electrons occupy energy levels, or electron shells, of an atom, which are actually complex, threedimensional volumes of space called orbitals • orbitals are where electrons are most likely to be found

2. 1 Atoms • Electron shells have specific numbers of orbitals/valances/ energy levels that may be filled with electrons. Bohr Model innermost shell holds 2 electrons, all other outer shells hold 8. must fill inner shells before moving to next shell. – atoms that have incomplete electron orbitals tend to be more reactive – atoms will lose, gain, or share electrons in order to fill completely their outermost electron shell – these actions are the basis of chemical bonding

2. 1 Atoms • as electrons move to a lower energy level, closer to the nucleus, energy is released • moving electrons to energy levels farther out from the nucleus requires energy Figure 2. 3 The electrons of atoms possess potential energy
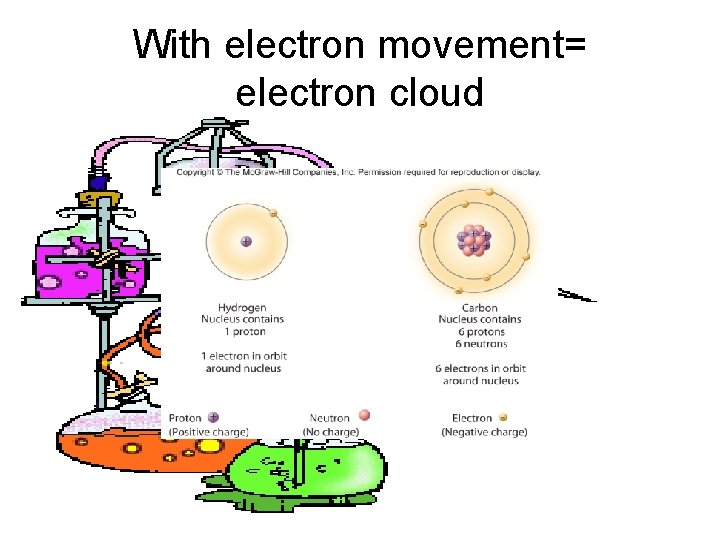
With electron movement= electron cloud
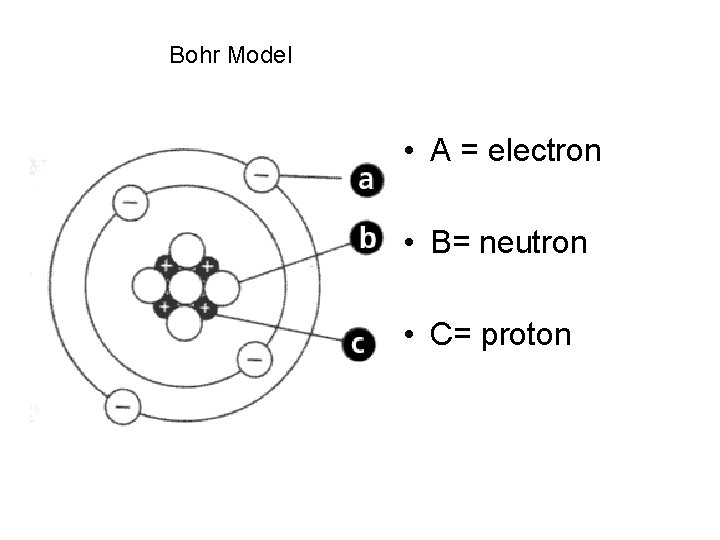
Bohr Model • A = electron • B= neutron • C= proton
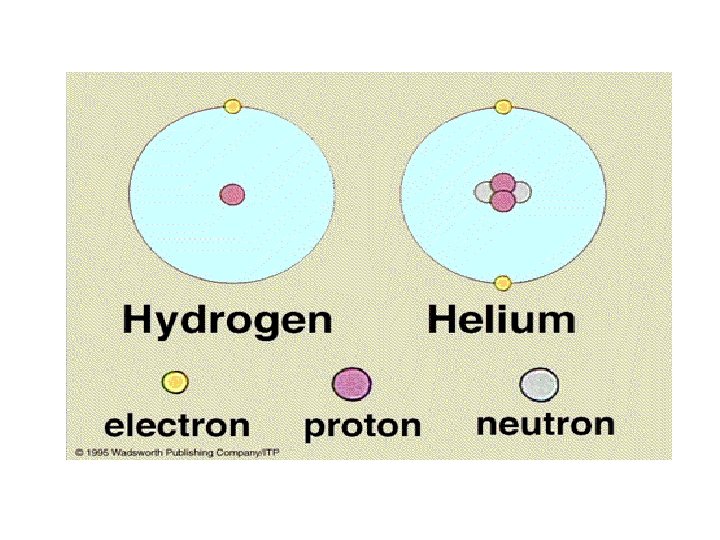

Elements are star stuff • There are 92 naturally occurring kinds of atoms or elements -- differ from one another by having different number of protons and neutrons in the nucleus and different numbers of electrons revolving around the nucleus. • They are formed inside of stars • 1 -6 of the periodic table are in all stars the rest are formed when a star goes supernova
Go to handouts!

95% of living material is made of only 4 different atoms: 1. 20% carbon (C) 2. 62% oxygen (0) 3. 10% hydrogen (H) 4. 3% nitrogen (N) • Remaining 5% made up of 30 different elements. e. g. magnesium, sodium, calcium, see back of a multiple mineral/vitamin bottle

2. 2 Ions and Isotopes • Ions – atoms that have gained or lost one or more electrons • Isotopes – atoms that have the same number of protons but different numbers of neutrons – most elements in nature exist as mixtures of different isotopes

Figure 2. 6 Isotopes of the element carbon

2. 2 Ions and Isotopes • Some isotopes are unstable and break up into particles with lower atomic numbers – this process is known as radioactive decay • Radioactive isotopes have multiple uses – dating fossils – medical procedures
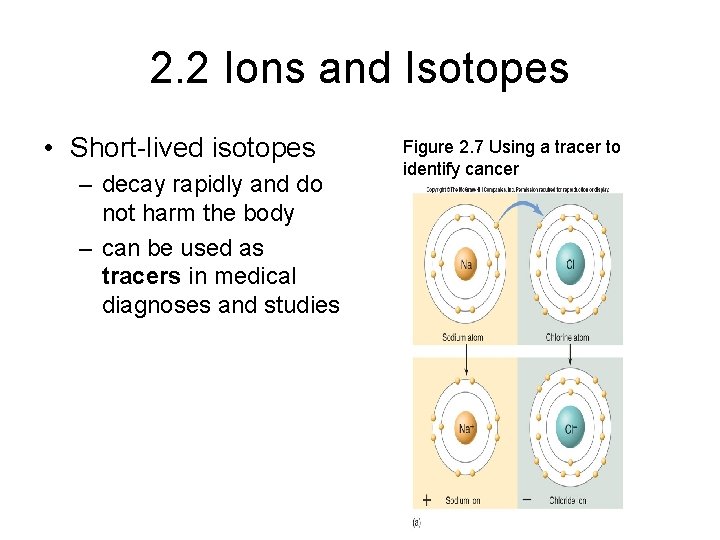
2. 2 Ions and Isotopes • Short-lived isotopes – decay rapidly and do not harm the body – can be used as tracers in medical diagnoses and studies Figure 2. 7 Using a tracer to identify cancer

MOLECULES • Atoms are not usually found by themselves; they combine with other atoms to form molecules -- e. g. O atoms combine with one another to form O 2 -- this is the form that oxygen generally is found - when different atoms combine a compound is formed -- e. g. H 2 O, NH 3, CO 2, Na. Cl.

• The ability of particular atoms to combine or react with other atoms is determined by the atom’s electron configuration -- electrons move around nucleus in levels called shells, orbits, or valence levels which representing different energy levels. + or – Example oxygen has 6 outer elections so it is a +6

Shells/Orbits/Levels and electrons • The inner most shell/orbit/valence level= energy level can hold 2 electrons. The second shell/orbit etc and all other outer shells/orbits etc can hold a maximum of 8 electrons
• Atoms that have outer electron shells that are filled are inert = nonreactive • Other atoms, including H, C, O, and N, do not have filled outer shells and they are reactive - they readily combine with other atoms.

• All atoms, while having a matching number of protons and electrons, want to have a full outer shell/orbit. This leads to atoms combining, sharing outer shell electrons, to be happy. But since their outer shell is not full all the time they stay in balance and stable. Creating a stable molecule

2. 3 Molecules Form Bonds • A molecule is a group of atoms held together by energy in the form of a chemical bond There are 3 principal types of chemical bonds • 1. Ionic 2. Covalent 3. Hydrogen • van der Waals forces are a kind of weak chemical attraction (not a bond) that come into play when atoms are very close to each other
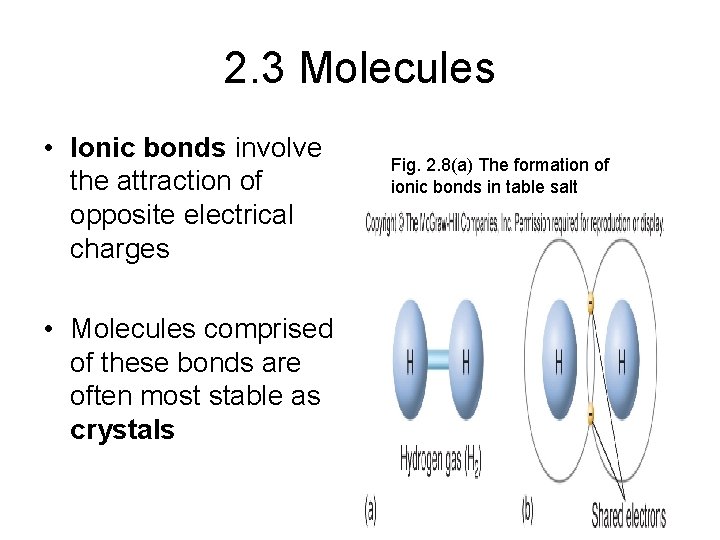
2. 3 Molecules • Ionic bonds involve the attraction of opposite electrical charges • Molecules comprised of these bonds are often most stable as crystals Fig. 2. 8(a) The formation of ionic bonds in table salt

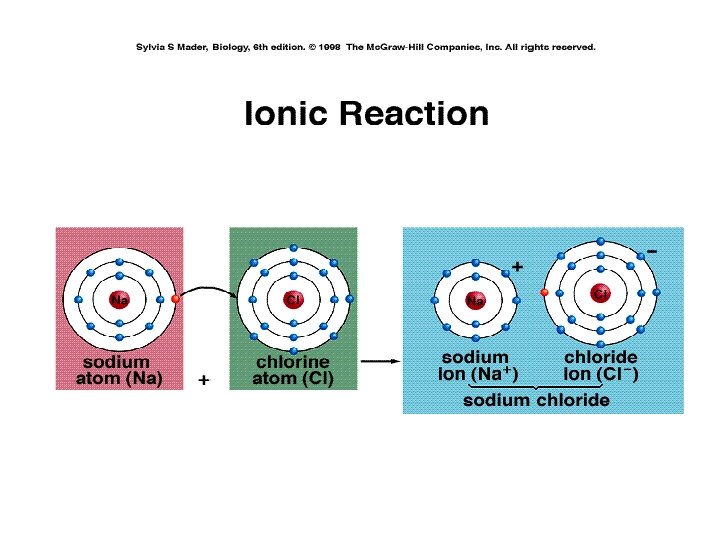
Ionic Bonds • Ionic bonding: One way that atoms may achieve electron stability in their outer shell is by losing or gaining electrons and becoming ions. • Example: sodium (Na) and chlorine (Cl) often form ions (Na+, Cl-) -- sodium losses an electron and chlorine picks up an electron -- since they are ions with opposite charges, they are attracted to one another and an ionic bond is formed -- the resulting molecule is a compound called sodium chloride (Na. Cl) (table salt). • Ionic bonds are strong when the compound exists as a dry solid -- in water, these compounds dissolve readily back into ions.
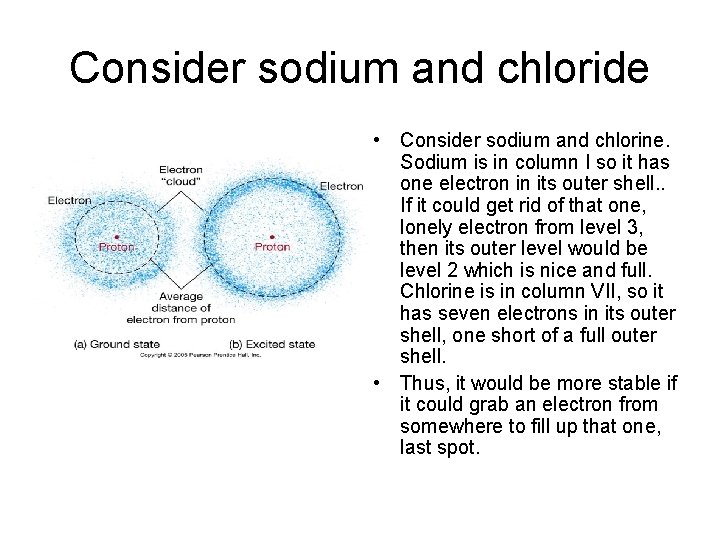
Consider sodium and chloride • Consider sodium and chlorine. Sodium is in column I so it has one electron in its outer shell. . If it could get rid of that one, lonely electron from level 3, then its outer level would be level 2 which is nice and full. Chlorine is in column VII, so it has seven electrons in its outer shell, one short of a full outer shell. • Thus, it would be more stable if it could grab an electron from somewhere to fill up that one, last spot.

2. 3 Molecules • Covalent bonds form between two atoms when they share electrons – the number of electrons shared varies depending on how many the atom needs to fill its outermost electron shell – covalent bonds are stronger than ionic bonds are directional
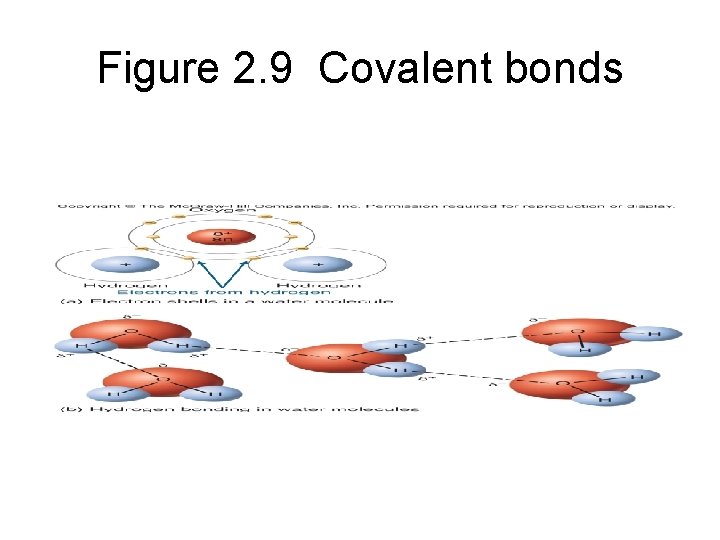
Figure 2. 9 Covalent bonds

Covalent Bonds • Covalent bonding: rather than gaining or losing electrons, atoms may share electrons with other atoms to fill both their outer shells and give them stability. • Two types of covalent bonds: nonpolar and polar covalent bonds.
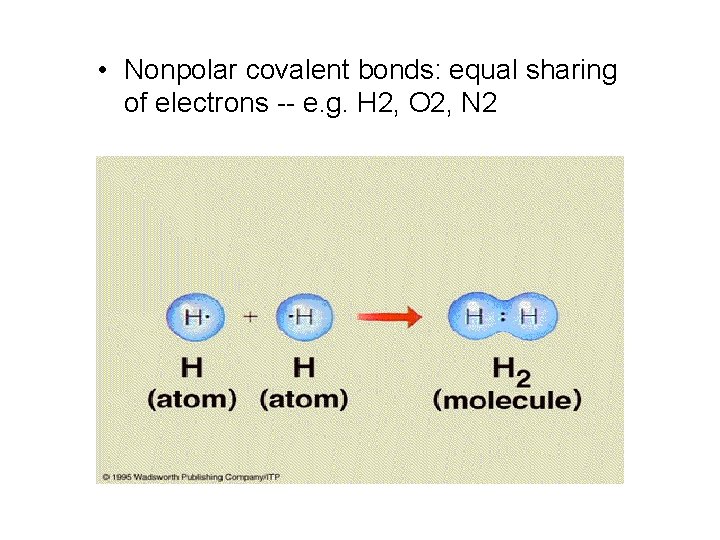
• Nonpolar covalent bonds: equal sharing of electrons -- e. g. H 2, O 2, N 2

2. 3 Molecules • Some atoms may be better at attracting the shared electrons of a covalent bond – this creates tiny partial negative and positive charges within the molecule, now called a polar molecule – polar covalent bonds form when the shared electrons of a covalent bond spend more time in the vicinity of a particular atoms
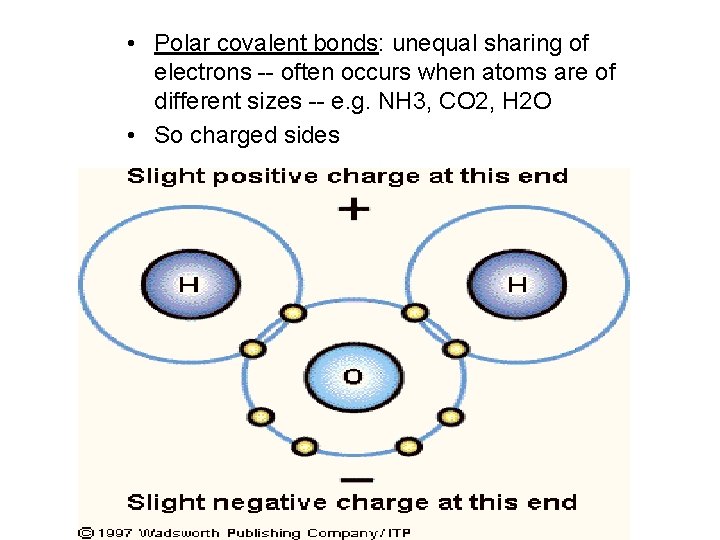
• Polar covalent bonds: unequal sharing of electrons -- often occurs when atoms are of different sizes -- e. g. NH 3, CO 2, H 2 O • So charged sides
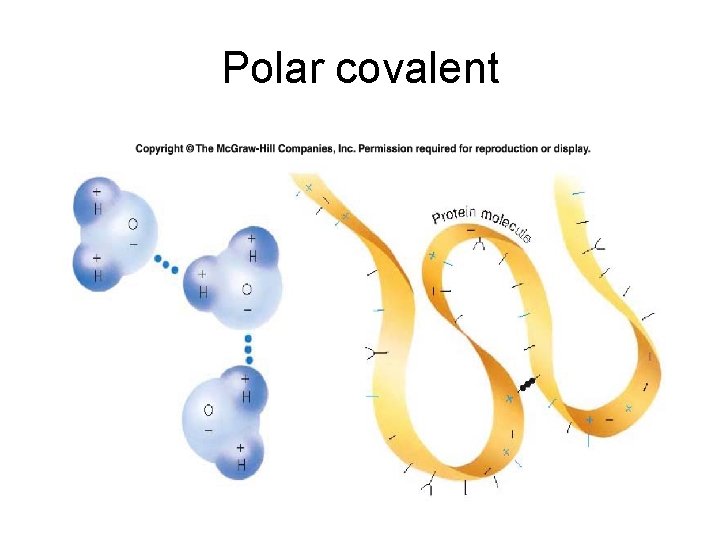
Polar covalent

2. 3 Molecules • Water molecules are polar and can form hydrogen bonds with each other Figure 2. 11 Hydrogen bonding in water molecules

• Hydrogen bonds -- differ from ionic and covalent bonds. • Ionic and covalent bonds are bond between atoms to form molecules -but, hydrogen bonds form between molecules.

2. 3 Molecules • Hydrogen bonds are weak electrical attractions between the positive end of one polar molecule and the negative end of another – each atom with a partial charge acts like a magnet to bond weakly to another polar atom with an opposite charge – the additive effects of many hydrogen bonding interactions can add collective strength to the bonds

• Hydrogen bonding gives water many of its biologically important characteristics -- e. g. high specific heat, surface tension, cohesiveness. • Now a message about water yes, is to science dihydrogenoxide or H 2 O

2. 4 Unique Properties of Water • Water is essential for life – the chemistry of life is water chemistry • Water is a polar molecule – water can form hydrogen bonds – hydrogen bonding confers on water many different special properties

2. 4 Unique Properties of Water • Heat Storage – water temperature changes slowly and holds temperature well • Ice Formation – few hydrogen bonds break at low temperatures • water becomes less dense as it freezes because hydrogen bonds stabilize and hold water molecules farther apart • High Heat of Vaporization – water requires tremendous energy to vaporize because of all the hydrogen bonds that must be broken – when water vaporizes, it takes this heat energy with it, allowing for evaporative cooling

Figure 2. 12 Ice formation

2. 4 Unique Properties of Water • Water molecules are attracted to other polar molecules – cohesion – when one water molecule is attracted to another water molecule – adhesion – when polar molecules other than water stick to a water molecule Figure 2. 13 Cohesion

2. 4 Unique Properties of Water • High polarity – in solution, water molecules tend to form the maximum number of hydrogen bonds • hydrophilic molecules are attracted to water and dissolve easily in it – these molecules are also polar and can form hydrogen bonds • hydrophobic molecules are repelled by water and do not dissolve – these molecules are nonpolar and do not form hydrogen bonds

Figure 2. 14 How salt dissolves in water

2. 4 Unique Properties of Water • Water is essential for life – the chemistry of life is water chemistry • Water is a polar molecule – water can form hydrogen bonds – hydrogen bonding confers on water many different special properties

2. 4 Unique Properties of Water • Heat Storage – water temperature changes slowly and holds temperature well • Ice Formation – few hydrogen bonds break at low temperatures • water becomes less dense as it freezes because hydrogen bonds stabilize and hold water molecules farther apart • High Heat of Vaporization – water requires tremendous energy to vaporize because of all the hydrogen bonds that must be broken – when water vaporizes, it takes this heat energy with it, allowing for evaporative cooling

Figure 2. 12 Ice formation

2. 4 Unique Properties of Water • Water molecules are attracted to other polar molecules – cohesion – when one water molecule is attracted to another water molecule – adhesion – when polar molecules other than water stick to a water molecule Figure 2. 13 Cohesion

2. 4 Unique Properties of Water • High polarity – in solution, water molecules tend to form the maximum number of hydrogen bonds • hydrophilic molecules are attracted to water and dissolve easily in it – these molecules are also polar and can form hydrogen bonds • hydrophobic molecules are repelled by water and do not dissolve – these molecules are nonpolar and do not form hydrogen bonds

Figure 2. 14 How salt dissolves in water
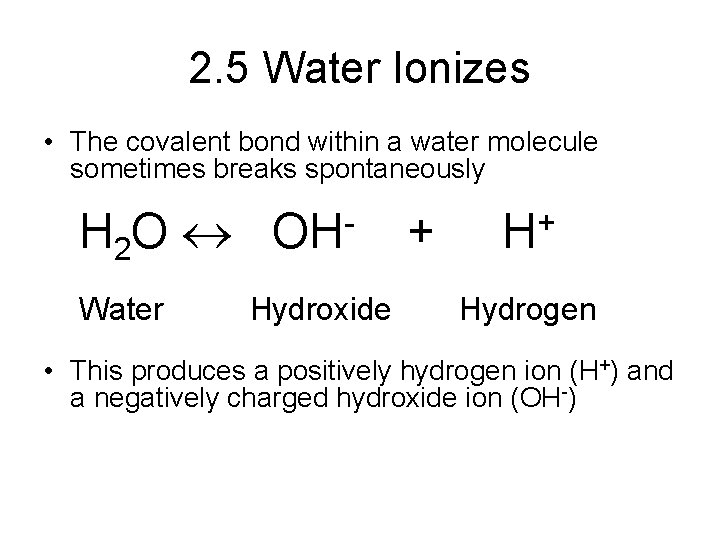
2. 5 Water Ionizes • The covalent bond within a water molecule sometimes breaks spontaneously H 2 O Water OH Hydroxide + + H Hydrogen • This produces a positively hydrogen ion (H+) and a negatively charged hydroxide ion (OH-)

2. 5 Water Ionizes • The amount of ionized hydrogen from water in a solution can be measured as p. H • The p. H scale is logarithmic, which means that a p. H scale difference of 1 unit actually represents a 10 -fold change in hydrogen ion concentration

Figure 2. 15 The p. H scale

2. 5 Water Ionizes • Pure water has a p. H of 7 – there are equal amounts of [H+] relative to [OH-] • Acid – any substance that dissociates in water and increases the [H+] – acidic solutions have p. H values below 7 • Base – any substance that combines with [H+] when dissolved in water – basic solutions have p. H values above 7

2. 5 Water Ionizes • The p. H in most living cells and their environments is fairly close to 7 – proteins involved in metabolism are sensitive to any p. H changes • Organisms use buffers to minimize p. H disturbances – a buffer is a chemical substance that takes up or releases hydrogen ions

Review of water and p. H

Water constitutes two-thirds the mass of most organisms & 75% earth's surface. • • Importance of Water to Life 1. Coolant: Has high heat of vaporization, aids body cooling. 2. High specific heat: ( H 20 = 1 cal / g / degree C ) - bodies of water stay constant temp. 3. Transport: polarity - dissolves many substances. 4. Habitat: Major component of internal (organism) & external environments

Properties of Water


Concepts of acids and bases • Some compounds release large numbers of H+ ions when they are in water -- these are called acids -- e. g. HCl, H 2 SO 4 are acids. • On the other hand, some compounds release OH- ions when they are in water -- these compounds are called bases -- e. g. Na. OH, KOH are bases. • Strengths of acids and bases are measured by the p. H scale --ranges from 0 to 14 -- measures relative concentrations of H+ and OH- ions -- pure water is neutral and has p. H = 7. 0 -- anything with a p. H less than 7 is an acid; p. H greater than 7 is a base.
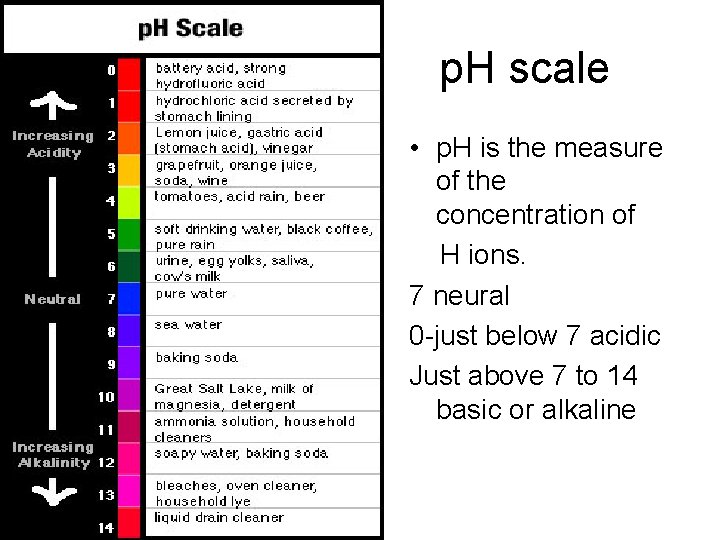
p. H scale • p. H is the measure of the concentration of H ions. 7 neural 0 -just below 7 acidic Just above 7 to 14 basic or alkaline
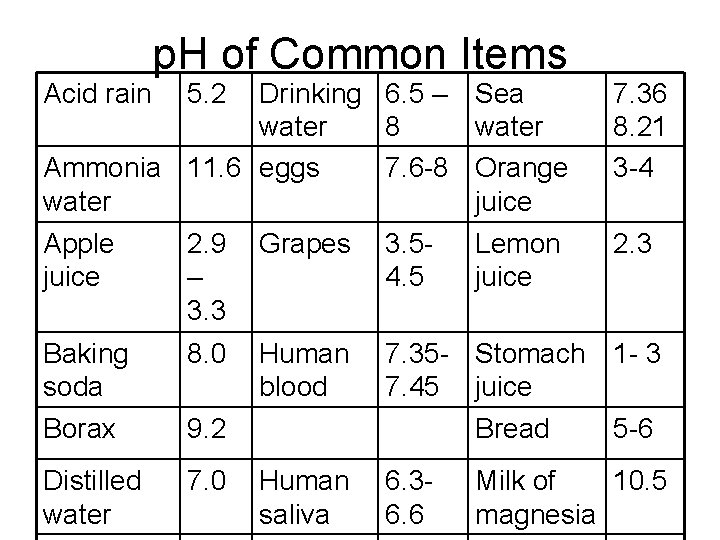
Acid rain p. H of Common Items 5. 2 Drinking 6. 5 – Sea water 8 water Ammonia 11. 6 eggs 7. 6 -8 Orange water juice 7. 36 8. 21 3 -4 Apple juice 2. 9 – 3. 3 Grapes 3. 54. 5 2. 3 Baking soda 8. 0 Human blood 7. 35 - Stomach 1 - 3 7. 45 juice Borax 9. 2 Distilled water 7. 0 Lemon juice Bread Human saliva 6. 36. 6 5 -6 Milk of 10. 5 magnesia

• p. H is important biologically because chemical reactions in cells are p. H specific -- disruptions in p. H will adversely affect organisms -- may effect entire groups of organisms -- e. g. acid rain and effect on lakes. • Organisms must maintain relatively constant internal p. H (homeostasis) to live.

• p. H of blood = 7. 4 -- slightly basic --serious illness or death may result if this p. H isn't maintained within narrow limits -- -- yet H+ and OH- may be added to blood through various metabolic processes -- how is "p. H" balance maintained? • Blood also contains special chemical compounds called "buffers" -- buffers pick up excess H+ or OH- ions or release them if they are in short supply -- by doing this, they act to keep p. H in blood (and cells) from changing.

In the bonds we store energy • We speak of energy as kinetic or potental • Kinetic is the energy of movemen While potential is energy in storage for later use. Energy is the ability to move things or do work Energy can not be created or destroyed it can however be changed between forms.

• First law of thermodynamics In its simplest form, the First Law of Thermodynamics states that neither matter nor energy can be created or destroyed. The amount of energy in the universe is constant – energy can be changed, moved, controlled, stored, or dissipated. However, this energy cannot be created from nothing or reduced to nothing. Every natural process transforms energy and moves energy, but cannot create or eliminate it. This principle forms a foundation for many of the physical sciences

Just in case you were wondering • They is a second and even a third law • Second Law of Thermodynamics - Increased Entropy The Second Law of Thermodynamics is commonly known as the Law of Increased Entropy. While quantity remains the same (First Law), the quality of matter/energy deteriorates gradually over time. How so? Usable energy is inevitably used for productivity, growth and repair. In the process, usable energy is converted into unusable energy. Thus, usable energy is irretrievably lost in the form of unusable energy. - See more at: http: //www. allaboutscience. org/secondlaw-of-thermodynamics. htm#sthash. n 7 ax. Zkb. S. dpuf
- Slides: 73