BASIC CELL BIOLOGY I CHEMISTRY of LIFE BASIC

BASIC CELL BIOLOGY I CHEMISTRY of LIFE BASIC CONCEPTS OF THE METABOLISM

BASIC CONCEPTS OF THE METABOLISM • • Information Turnover Energy Supply Metabolic Pathways; the Turnover of Materials Basic Properties of the Enzymes

Lecture 6 METABOLISM THE TOTALITY OF CHEMICAL REACTIONS IN THE CELL Information turnover – continuation, use and transfer of the hereditary information Energy supply – production of energy for the chemical reactions in the cell Turnover of the materials - biosynthesis and degradation of macromolecules, their constituents and other substances in the cell
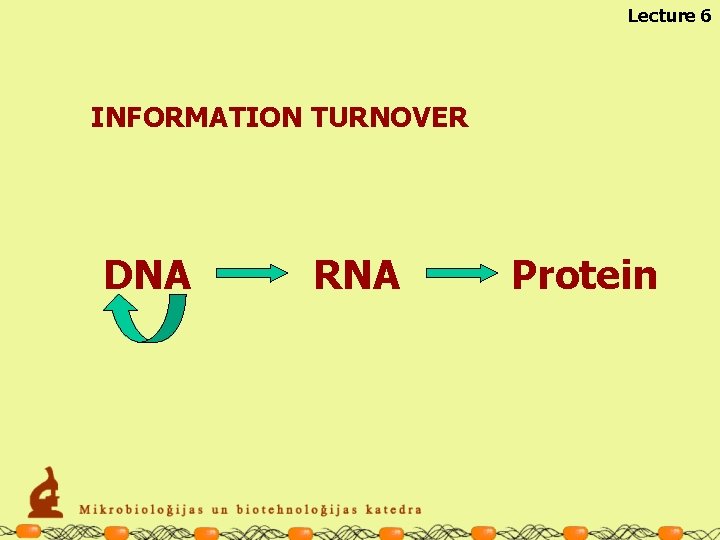
Lecture 6 INFORMATION TURNOVER DNA RNA Protein
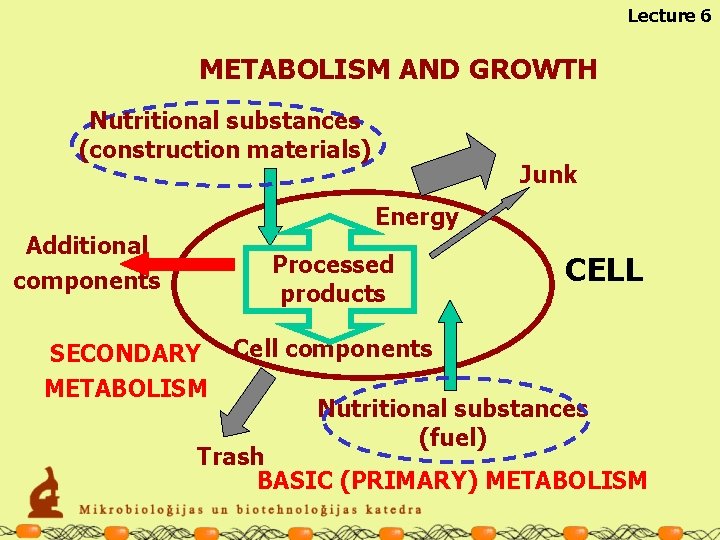
Lecture 6 METABOLISM AND GROWTH Nutritional substances (construction materials) Junk Energy Additional components Processed products SECONDARY METABOLISM CELL Cell components Nutritional substances (fuel) Trash BASIC (PRIMARY) METABOLISM

Lecture 6 Energy supply ENERGY IS SUPPLIED TO THE MOST CELLS BY CHEMICAL REACTIONS - OXIDATION FOR THE PLANTS AND SEVERAL MICROORGANISM SPECIES THE SOURCE OF ENERGY MAY BE LIGHT
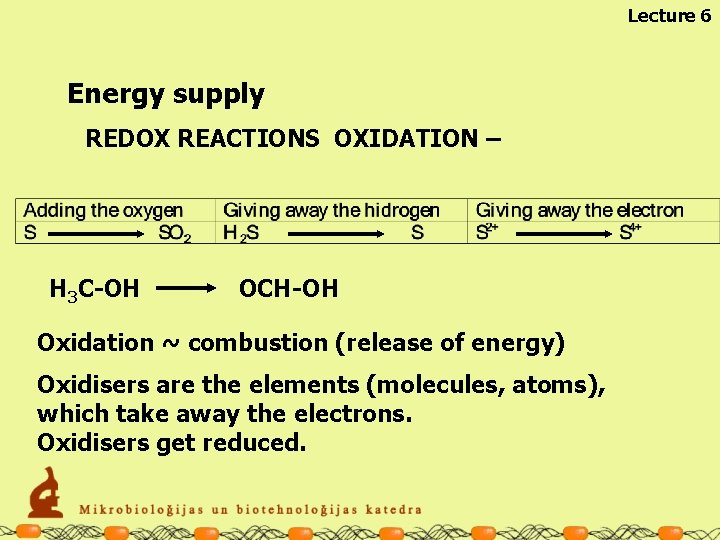
Lecture 6 Energy supply REDOX REACTIONS OXIDATION – H 3 C-OH OCH-OH Oxidation ~ combustion (release of energy) Oxidisers are the elements (molecules, atoms), which take away the electrons. Oxidisers get reduced.

Lecture 6 Energy supply REDOX REACTIONS REDUCTION – Reduction needs the energy Reducers are the elements (molecules, compounds), which donate electrons to other elements (molecules, compounds). Reducers get oxidised.
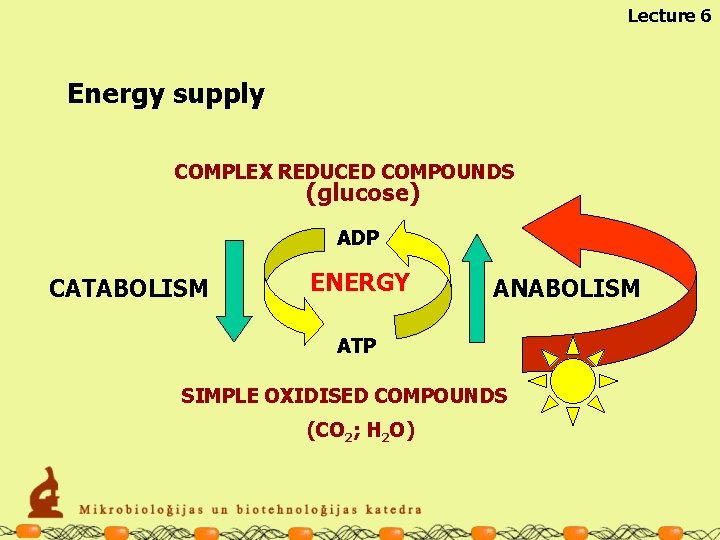
Lecture 6 Energy supply COMPLEX REDUCED COMPOUNDS (glucose) ADP CATABOLISM ENERGY ANABOLISM ATP SIMPLE OXIDISED COMPOUNDS (CO 2; H 2 O)
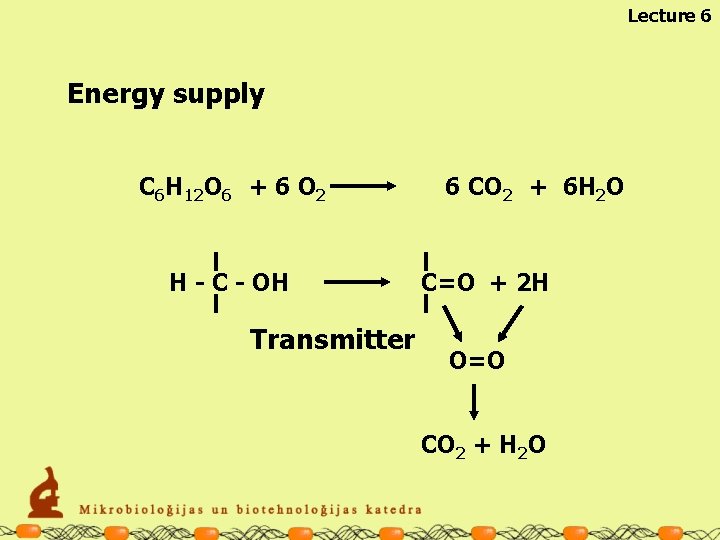
Lecture 6 Energy supply C 6 H 12 O 6 + 6 O 2 H - C - OH Transmitter 6 CO 2 + 6 H 2 O C=O + 2 H O=O CO 2 + H 2 O
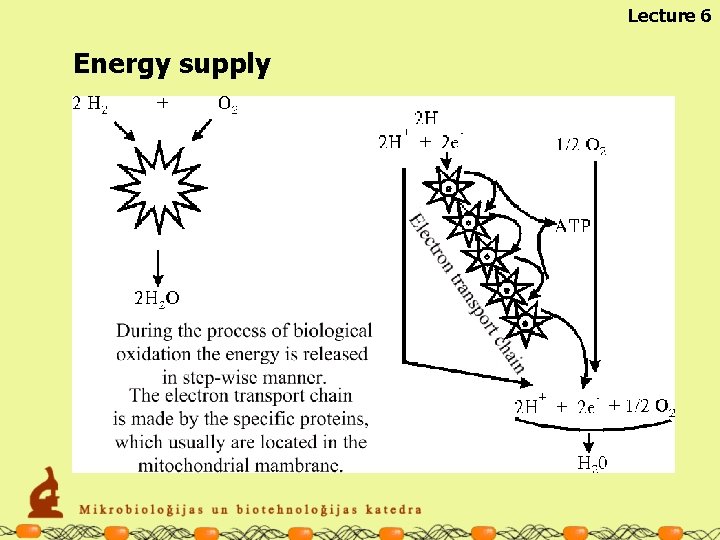
Lecture 6 Energy supply
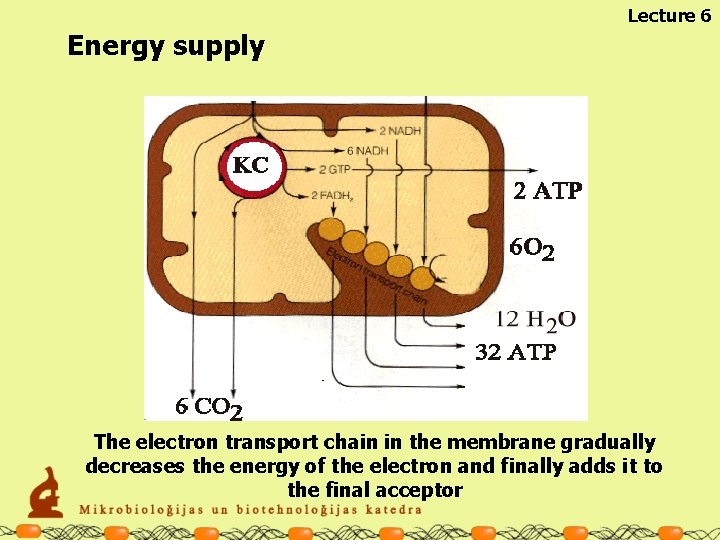
Lecture 6 Energy supply The electron transport chain in the membrane gradually decreases the energy of the electron and finally adds it to the final acceptor
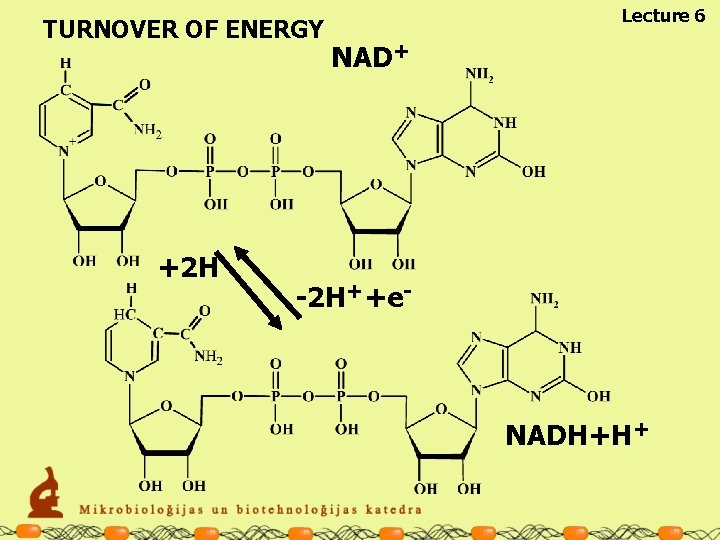
TURNOVER OF ENERGY +2 H Lecture 6 NAD+ -2 H++e- NADH+H+
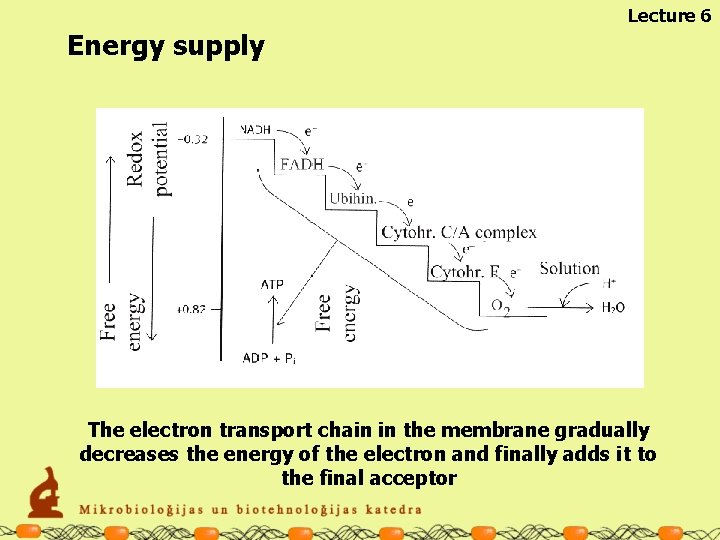
Lecture 6 Energy supply The electron transport chain in the membrane gradually decreases the energy of the electron and finally adds it to the final acceptor
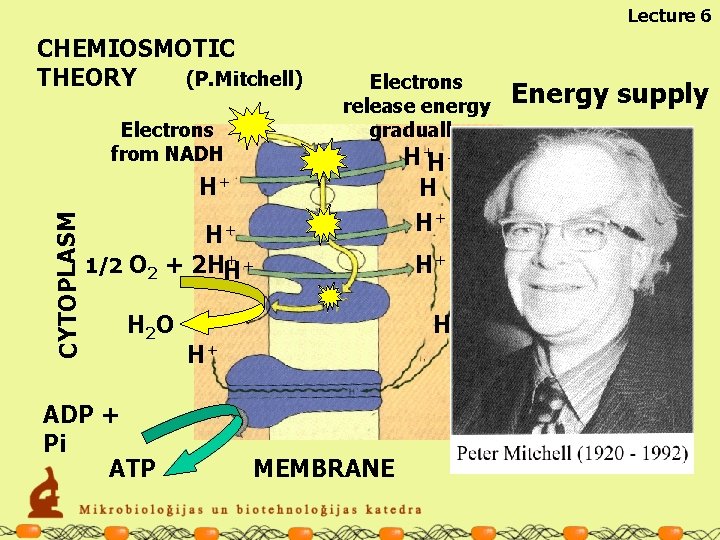
Lecture 6 Electrons from NADH Electrons release energy gradually CYTOPLASM H+ H+ ++ 1/2 O 2 + 2 HH H 2 O ADP + Pi ATP H+H+ H HH++ ++ H H+ H+ H+ MEMBRANE Energy supply OUTERMEMBRANE SPACE CHEMIOSMOTIC THEORY (P. Mitchell)

Lecture 6 Energy supply

Lecture 6 Biosythesis and degradation of the macromolecules and other chemical compounds in the cell occurs in the result of elementary chemical reactions, which are catalysed by special proteins - enzymes.
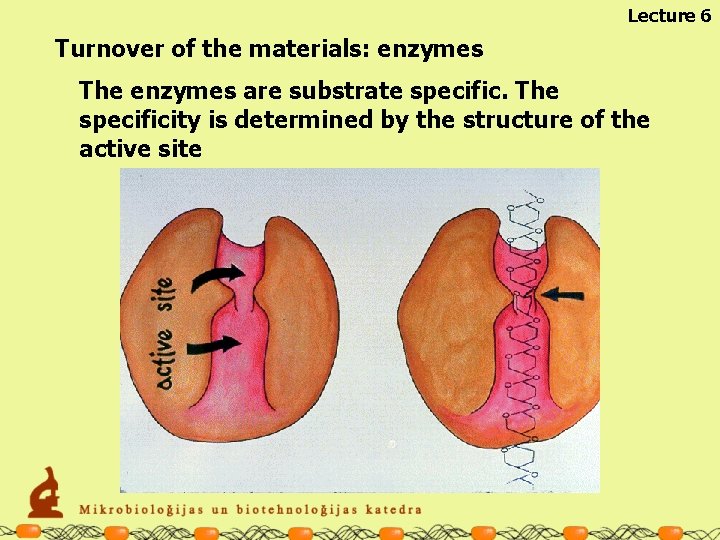
Lecture 6 Turnover of the materials: enzymes The enzymes are substrate specific. The specificity is determined by the structure of the active site
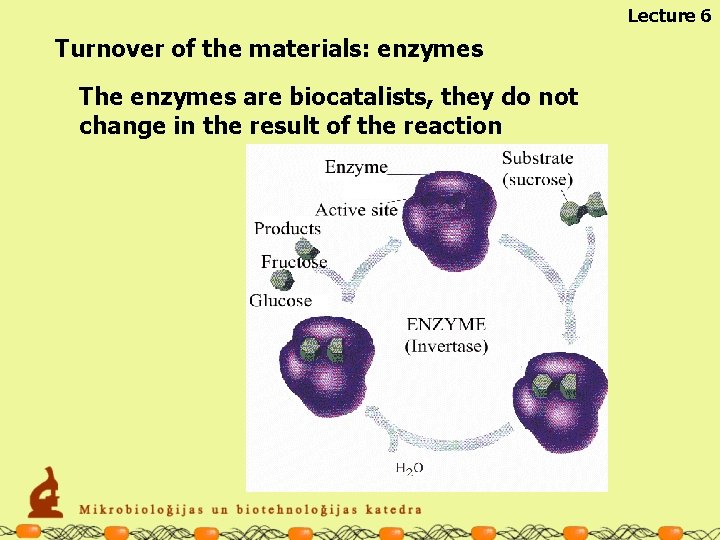
Lecture 6 Turnover of the materials: enzymes The enzymes are biocatalists, they do not change in the result of the reaction
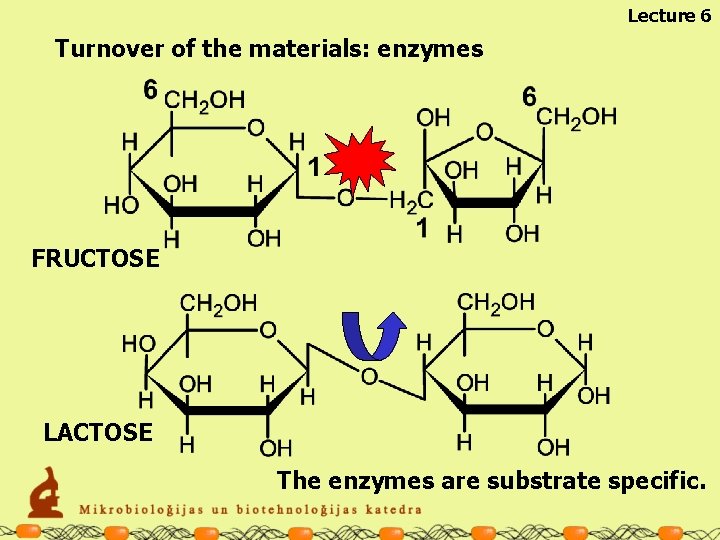
Lecture 6 Turnover of the materials: enzymes FRUCTOSE LACTOSE The enzymes are substrate specific.

Lecture 6 Turnover of the materials: enzymes The enzymes do not supply energy to the reactions they catalyse
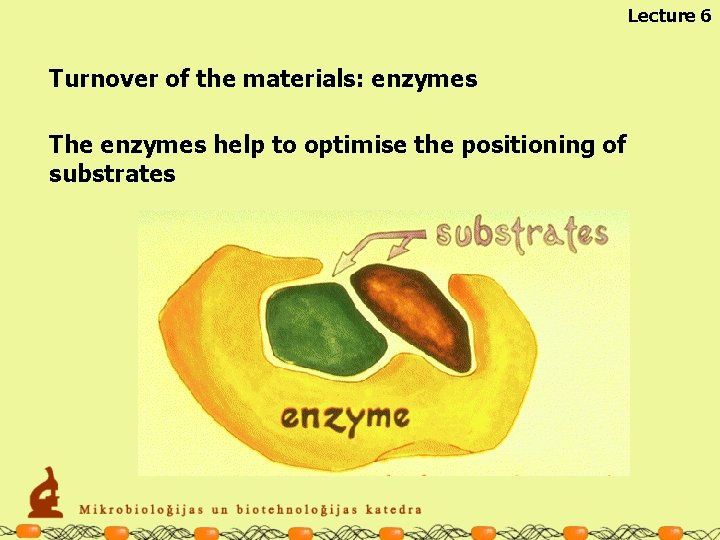
Lecture 6 Turnover of the materials: enzymes The enzymes help to optimise the positioning of substrates
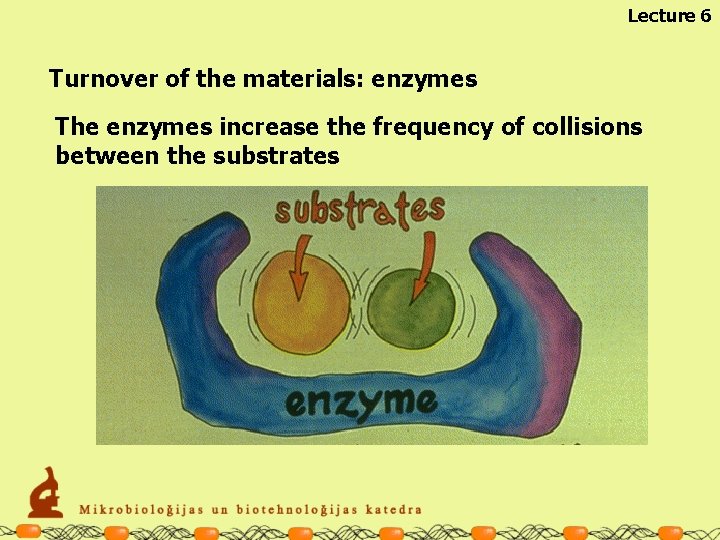
Lecture 6 Turnover of the materials: enzymes The enzymes increase the frequency of collisions between the substrates
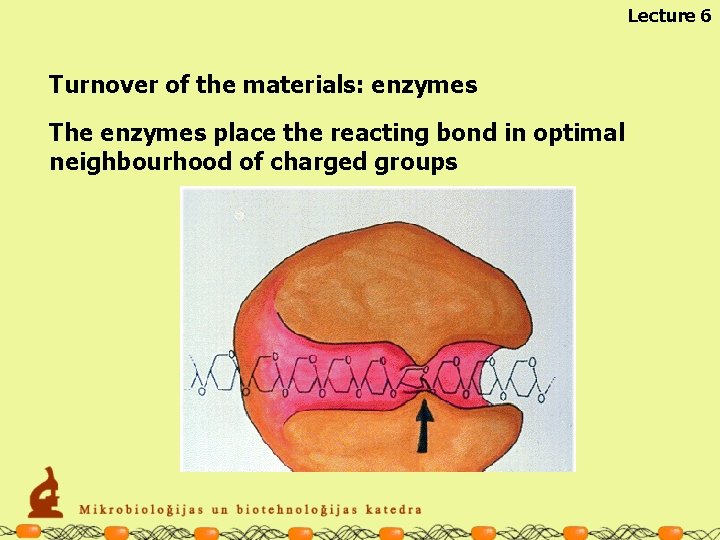
Lecture 6 Turnover of the materials: enzymes The enzymes place the reacting bond in optimal neighbourhood of charged groups
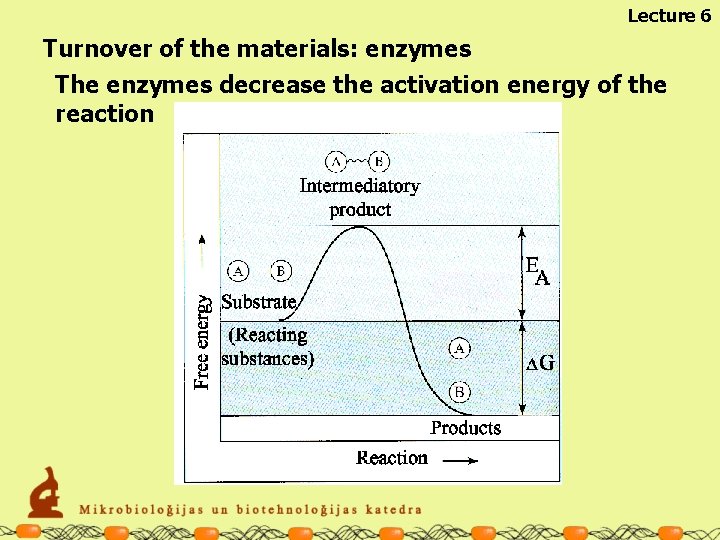
Lecture 6 Turnover of the materials: enzymes The enzymes decrease the activation energy of the reaction
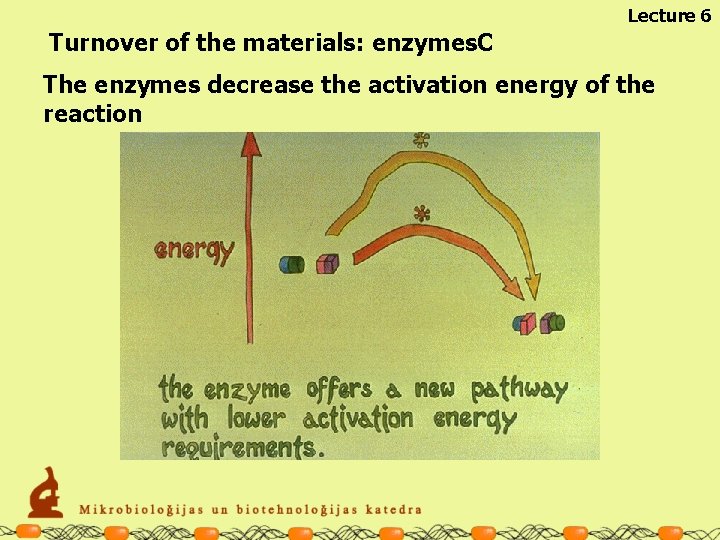
Lecture 6 Turnover of the materials: enzymes. C The enzymes decrease the activation energy of the reaction
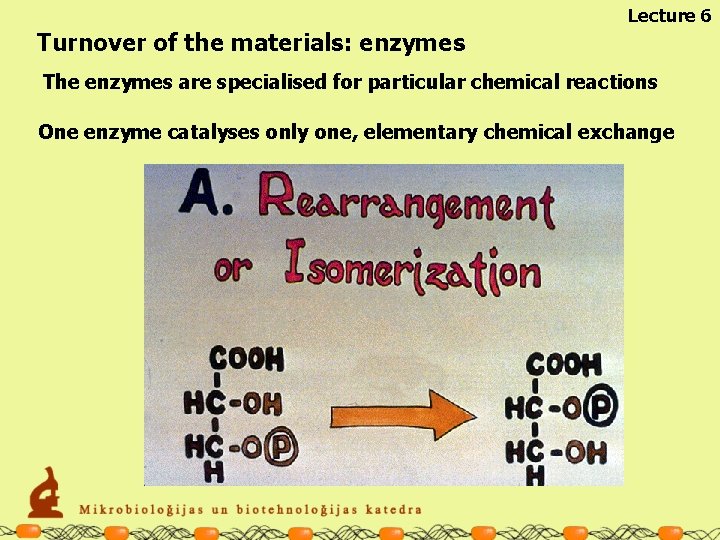
Lecture 6 Turnover of the materials: enzymes The enzymes are specialised for particular chemical reactions One enzyme catalyses only one, elementary chemical exchange
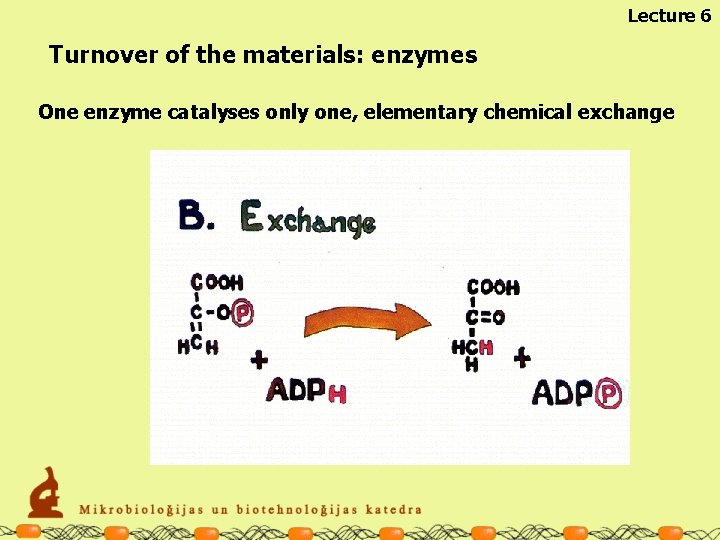
Lecture 6 Turnover of the materials: enzymes One enzyme catalyses only one, elementary chemical exchange
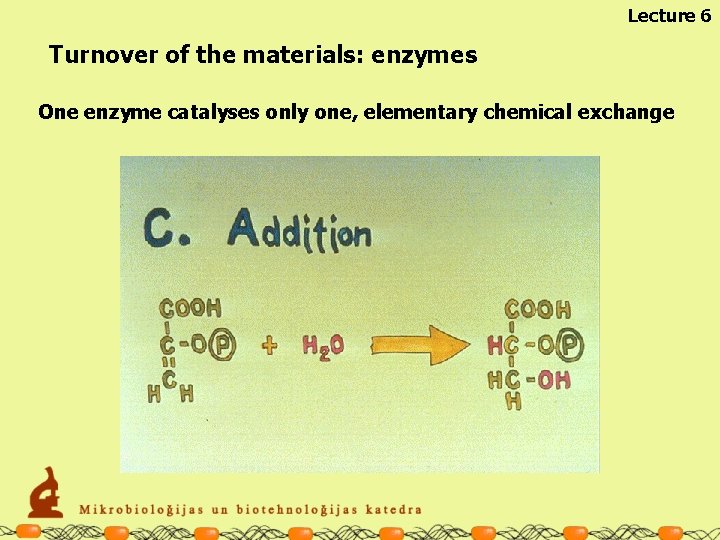
Lecture 6 Turnover of the materials: enzymes One enzyme catalyses only one, elementary chemical exchange
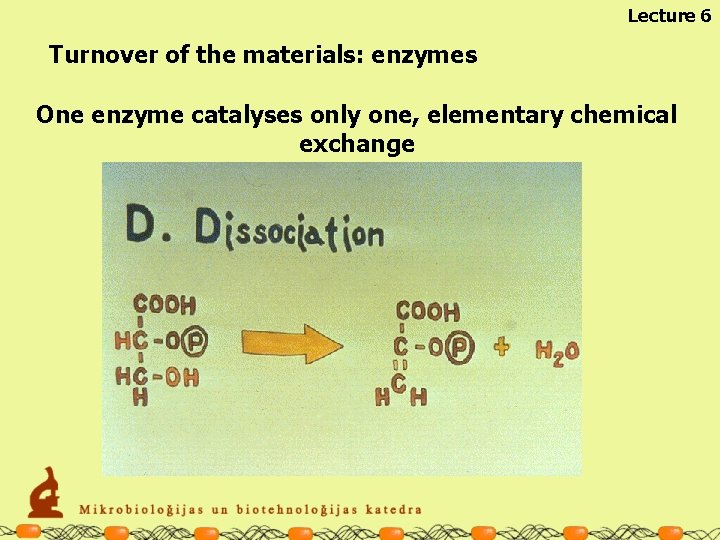
Lecture 6 Turnover of the materials: enzymes One enzyme catalyses only one, elementary chemical exchange

Lecture 6 Turnover of the materials: enzymes The enzymatic reactions are reversible K 1 S +E K 2 SE E +P
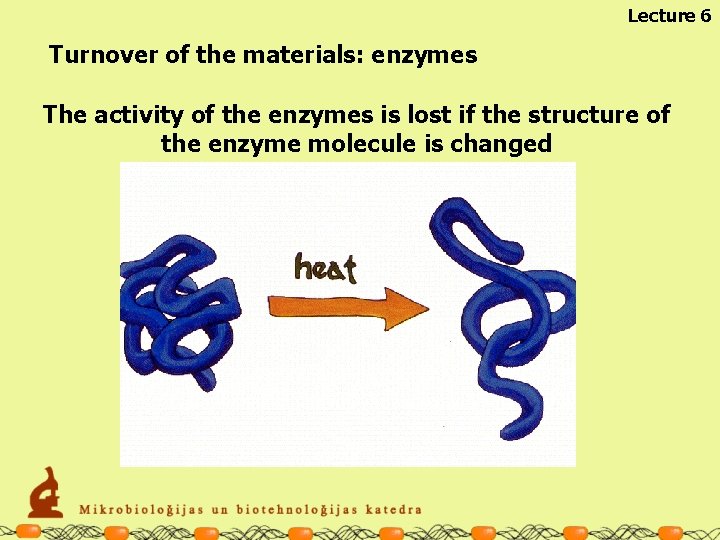
Lecture 6 Turnover of the materials: enzymes The activity of the enzymes is lost if the structure of the enzyme molecule is changed
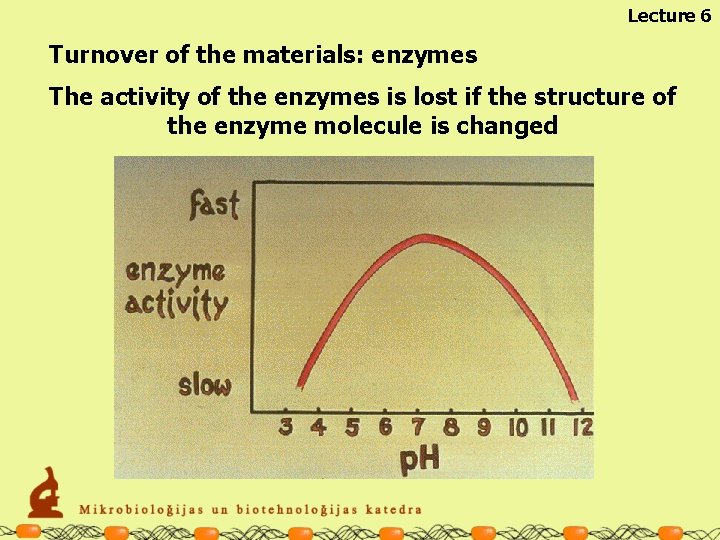
Lecture 6 Turnover of the materials: enzymes The activity of the enzymes is lost if the structure of the enzyme molecule is changed
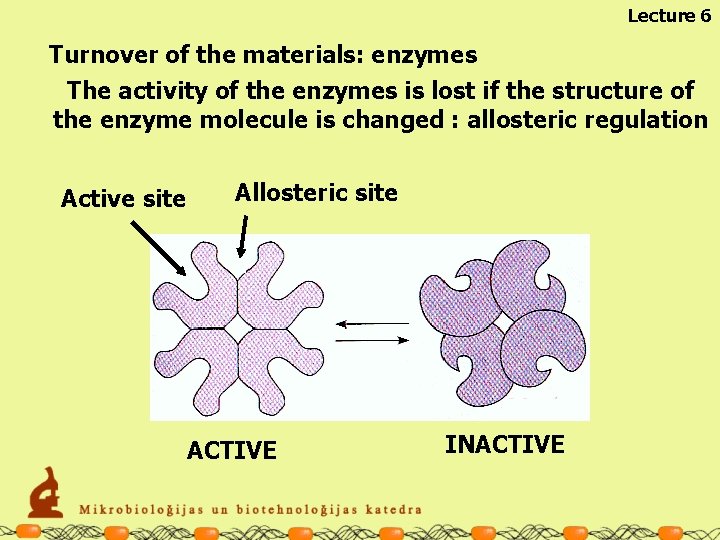
Lecture 6 Turnover of the materials: enzymes The activity of the enzymes is lost if the structure of the enzyme molecule is changed : allosteric regulation Active site Allosteric site ACTIVE INACTIVE
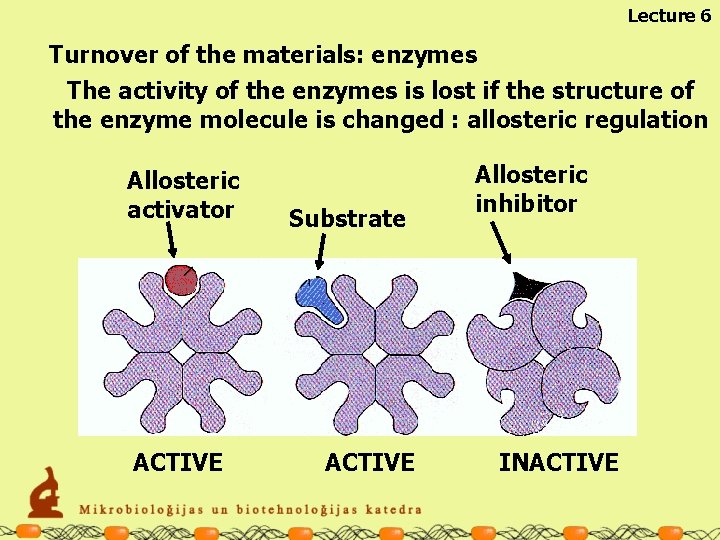
Lecture 6 Turnover of the materials: enzymes The activity of the enzymes is lost if the structure of the enzyme molecule is changed : allosteric regulation Allosteric activator ACTIVE Substrate ACTIVE Allosteric inhibitor INACTIVE
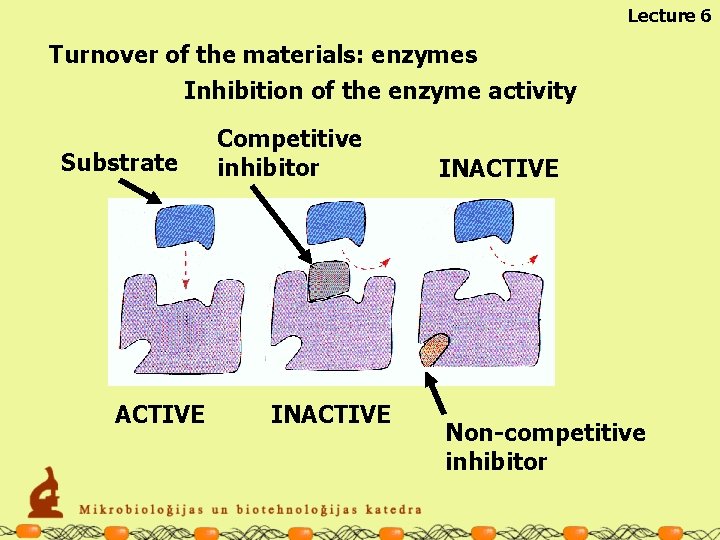
Lecture 6 Turnover of the materials: enzymes Inhibition of the enzyme activity Substrate ACTIVE Competitive inhibitor INACTIVE Non-competitive inhibitor
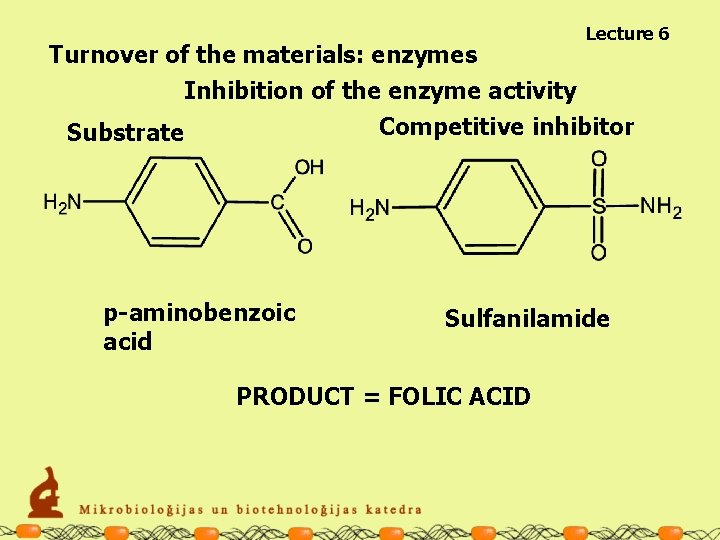
Lecture 6 Turnover of the materials: enzymes Inhibition of the enzyme activity Competitive inhibitor Substrate p-aminobenzoic acid Sulfanilamide PRODUCT = FOLIC ACID
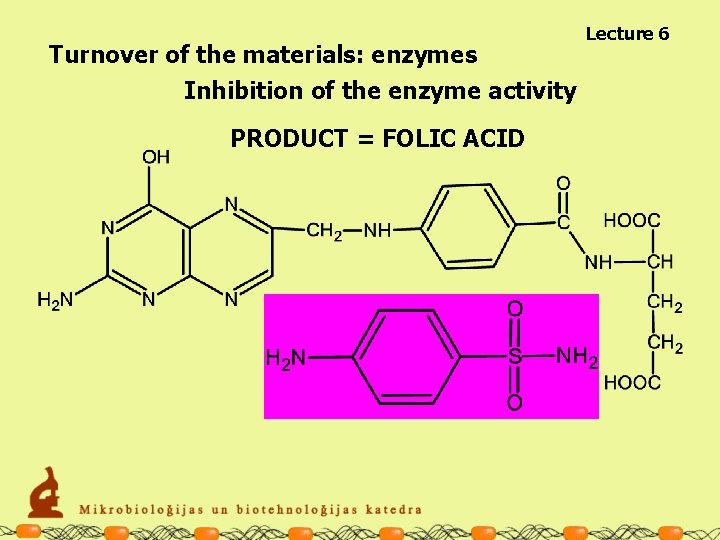
Turnover of the materials: enzymes Inhibition of the enzyme activity PRODUCT = FOLIC ACID Lecture 6

Lecture 6 Turnover of the materials: metabolic pathways METABOLIC REACTIONS ARE ORGANISED IN METABOLIC PATHWAYS Linear

Lecture 6 Turnover of the materials: metabolic pathways METABOLIC REACTIONS ARE ORGANISED IN METABOLIC PATHWAYS Cyclic
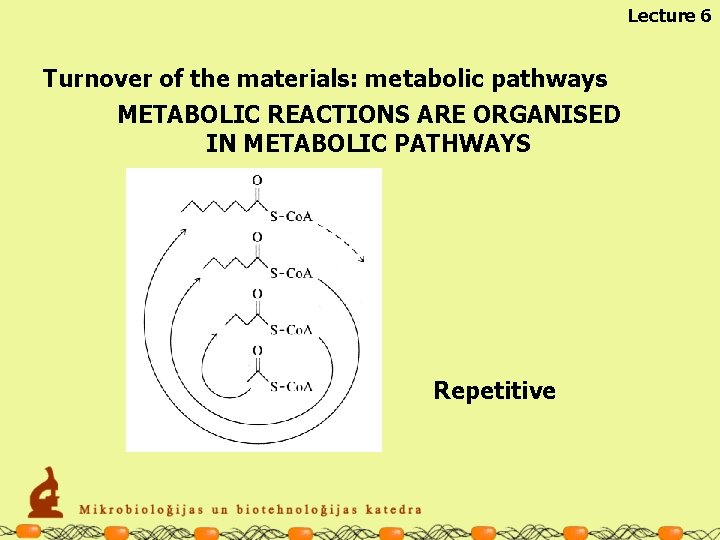
Lecture 6 Turnover of the materials: metabolic pathways METABOLIC REACTIONS ARE ORGANISED IN METABOLIC PATHWAYS Repetitive

Turnover of the materials: metabolic pathways METABOLIC REACTIONS ARE ORGANISED IN METABOLIC PATHWAYS Lecture 6
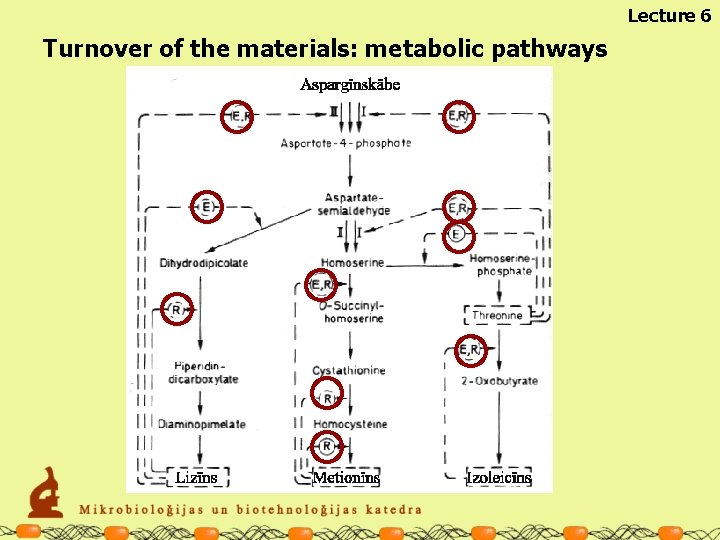
Lecture 6 Turnover of the materials: metabolic pathways

Lecture 6 CHEMISTRY OF LIFE ORGANISMS ARE CHEMICAL FACTORIES
Lecture 6 CHEMISTRY OF LIFE We’ve been here
- Slides: 45