Bases Moleculares que Justifican el Tratamiento Dirigido a

Bases Moleculares que Justifican el Tratamiento Dirigido a Diana José Manuel Trigo Hospital Universitario Virgen de la Victoria Málaga Sesión de formación + MIR temas de actualidad Tumores renales: Revisión tratamiento dirigido a diana en el cáncer renal 29 de Octubre de 2010
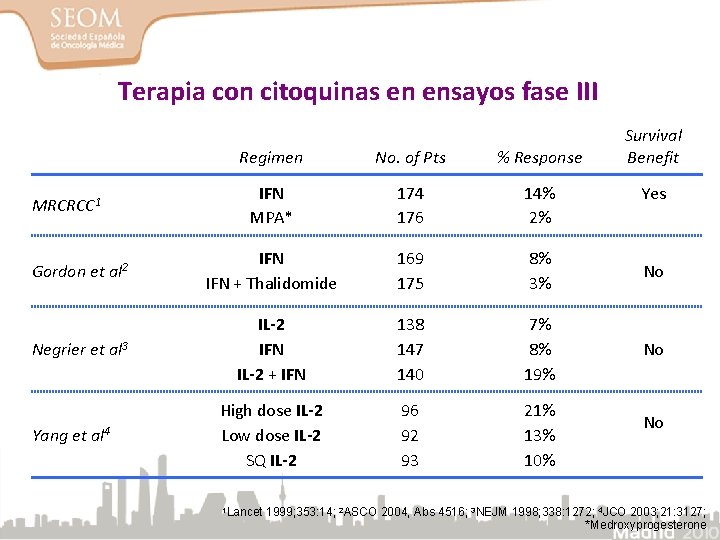
Terapia con citoquinas en ensayos fase III Survival Benefit Regimen No. of Pts % Response IFN MPA* 174 176 14% 2% Gordon et al 2 IFN + Thalidomide 169 175 8% 3% No Negrier et al 3 IL-2 IFN IL-2 + IFN 138 147 140 7% 8% 19% No High dose IL-2 Low dose IL-2 SQ IL-2 96 92 93 21% 13% 10% MRCRCC 1 Yang et al 4 1 Lancet Yes No 1999; 353: 14; 2 ASCO 2004, Abs 4516; 3 NEJM 1998; 338: 1272; 4 JCO 2003; 21: 3127; *Medroxyprogesterone
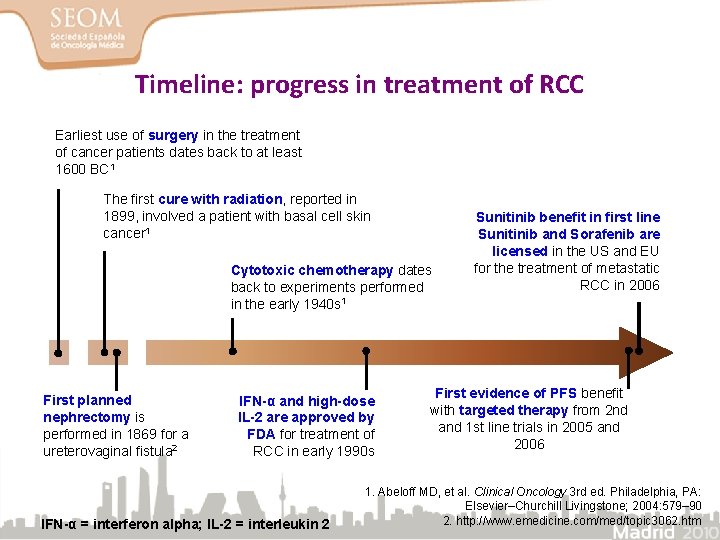
Timeline: progress in treatment of RCC Earliest use of surgery in the treatment of cancer patients dates back to at least 1600 BC 1 The first cure with radiation, reported in 1899, involved a patient with basal cell skin cancer 1 Cytotoxic chemotherapy dates back to experiments performed in the early 1940 s 1 First planned nephrectomy is performed in 1869 for a ureterovaginal fistula 2 IFN-α and high-dose IL-2 are approved by FDA for treatment of RCC in early 1990 s IFN-α = interferon alpha; IL-2 = interleukin 2 Sunitinib benefit in first line Sunitinib and Sorafenib are licensed in the US and EU for the treatment of metastatic RCC in 2006 First evidence of PFS benefit with targeted therapy from 2 nd and 1 st line trials in 2005 and 2006 1. Abeloff MD, et al. Clinical Oncology 3 rd ed. Philadelphia, PA: Elsevier–Churchill Livingstone; 2004: 579– 90 2. http: //www. emedicine. com/med/topic 3062. htm

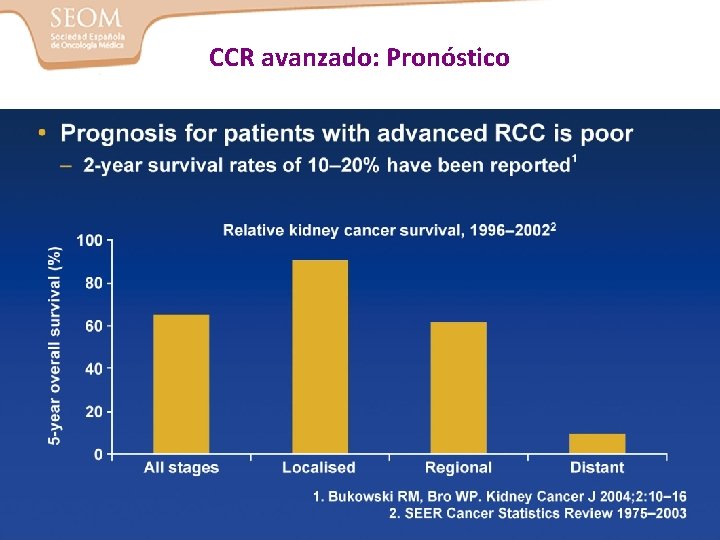
Cáncer renal: Epidemiología • 2% of all cancers in the US (36, 000 incident cases/yr)[1] • Incidence: 11. 3/100, 000 (15. 7 men; 7. 8 women)[2] • Third most common genitourinary cancer after prostate and bladder[2] • 80% to 85% of malignant kidney tumors[2] • 12, 600 people die from RCC yearly, generally due to advanced disease[2] • 5 -year survival has improved – 20. 0% in 1974 -1976; 63. 9% in 1995 -2000[3] • The prognosis for patients with advanced RCC remains poor 1. Motzer RJ, et al. N Engl J Med. 1996; 335: 865 -875. 2 Jemal A, et al. CA Cancer J Clin. 2006; 56: 106 -130. 3. Ries LAG, et al. SEER Cancer Statistics Review, 1975 -2002.
CCR avanzado: Pronóstico
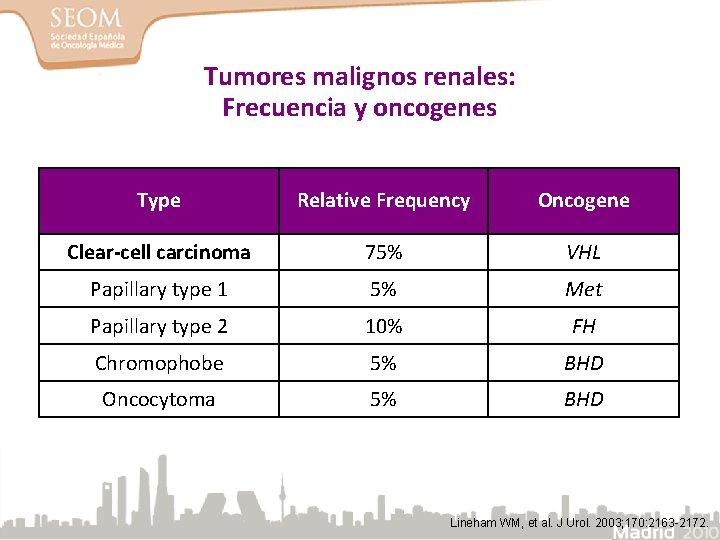
Tumores malignos renales: Frecuencia y oncogenes Type Relative Frequency Oncogene Clear-cell carcinoma 75% VHL Papillary type 1 5% Met Papillary type 2 10% FH Chromophobe 5% BHD Oncocytoma 5% BHD Lineham WM, et al. J Urol. 2003; 170: 2163 -2172.
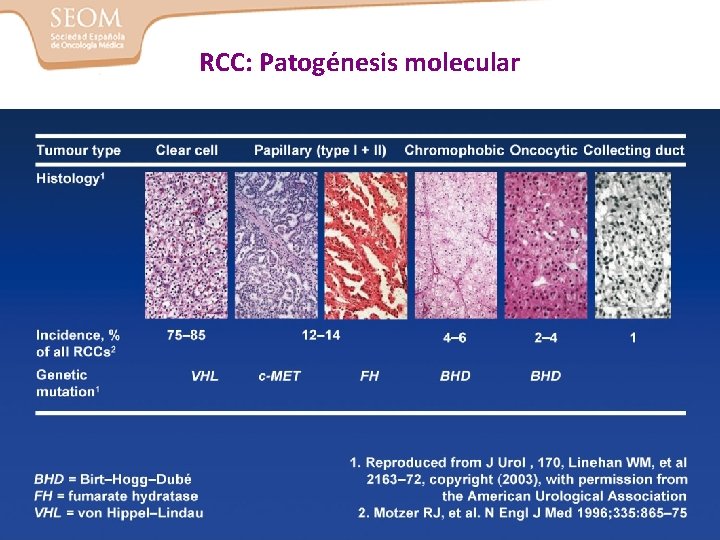
RCC: Patogénesis molecular

Patogenia del carcinoma renal de células claras • Existen dos formas distintas: – Esporádico: 90 -95% – Síndromes hereditarios: 5 -10% • Inactivación del gen supresor tumoral VHL: – 100% de los CR hereditarios – 50 -70% de los CR esporádicos
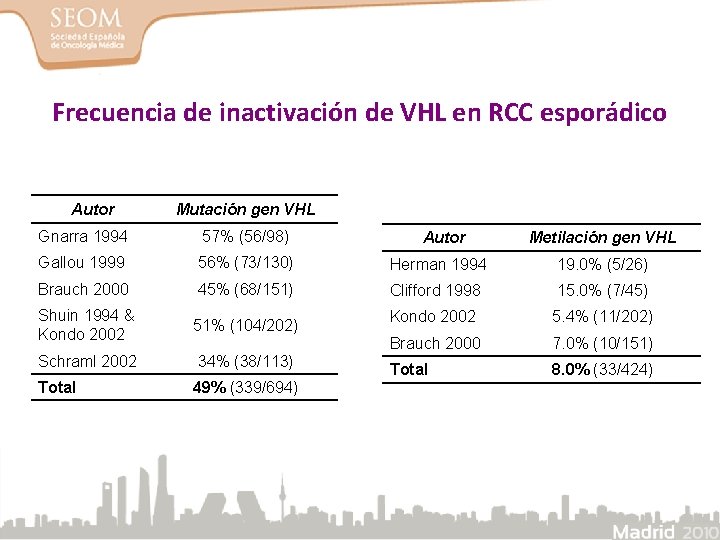
Frecuencia de inactivación de VHL en RCC esporádico Autor Mutación gen VHL Gnarra 1994 57% (56/98) Gallou 1999 56% (73/130) Herman 1994 19. 0% (5/26) Brauch 2000 45% (68/151) Clifford 1998 15. 0% (7/45) Shuin 1994 & Kondo 2002 51% (104/202) Kondo 2002 5. 4% (11/202) Brauch 2000 7. 0% (10/151) Schraml 2002 34% (38/113) Total 8. 0% (33/424) Total 49% (339/694) Autor Metilación gen VHL
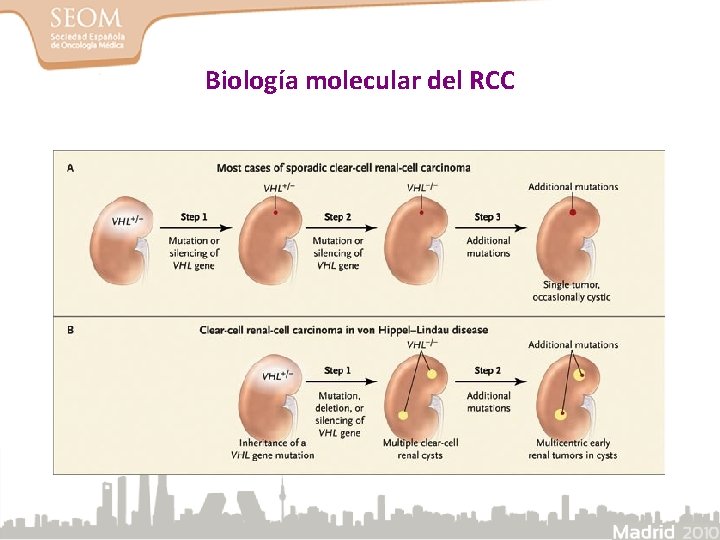
Biología molecular del RCC
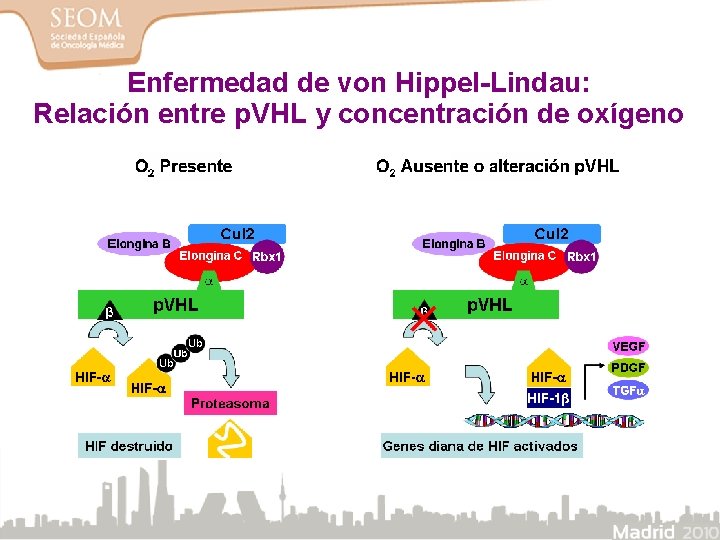
Enfermedad de von Hippel-Lindau: Relación entre p. VHL y concentración de oxígeno
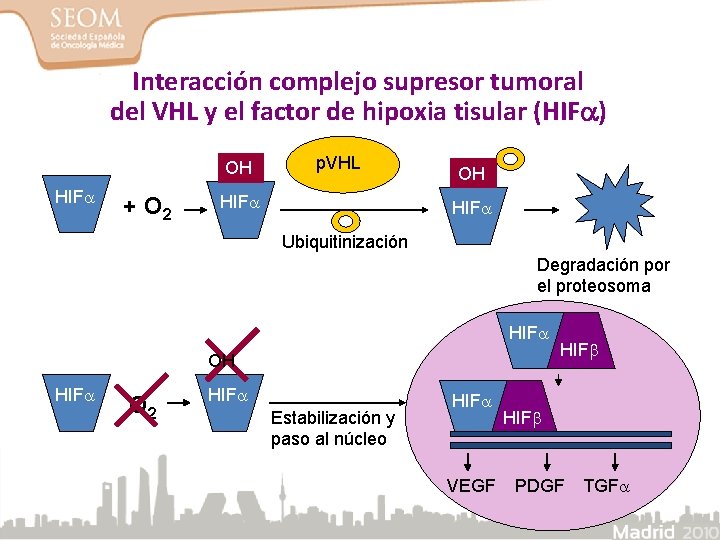
Interacción complejo supresor tumoral del VHL y el factor de hipoxia tisular (HIF ) OH HIF + O 2 p. VHL HIF OH HIF Ubiquitinización Degradación por el proteosoma HIF OH HIF O 2 HIF Estabilización y paso al núcleo HIF VEGF HIF PDGF TGF
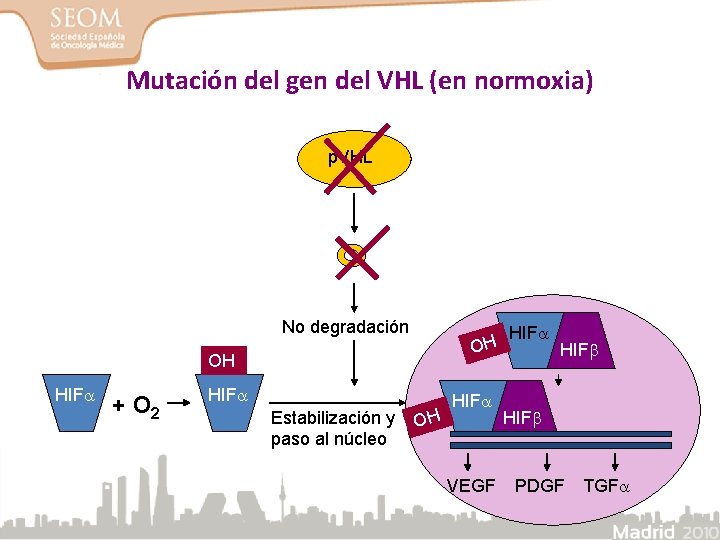
Mutación del gen del VHL (en normoxia) p. VHL No degradación OH OH HIF + O 2 HIF Estabilización y paso al núcleo OH HIF VEGF HIF PDGF TGF
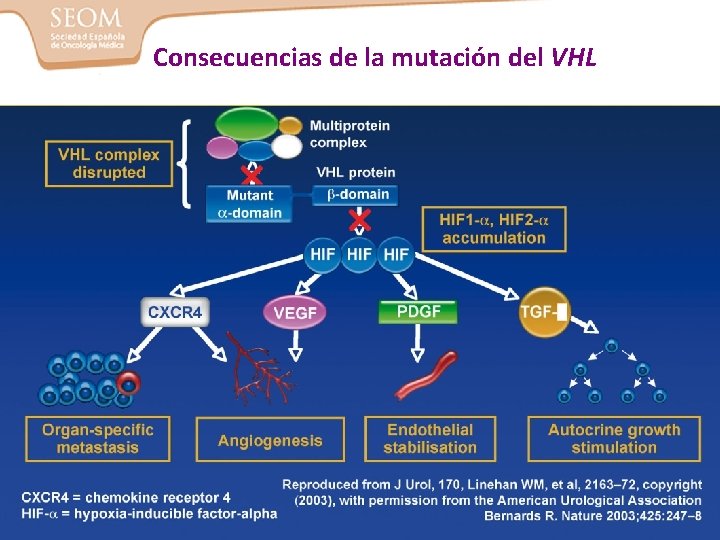
Consecuencias de la mutación del VHL
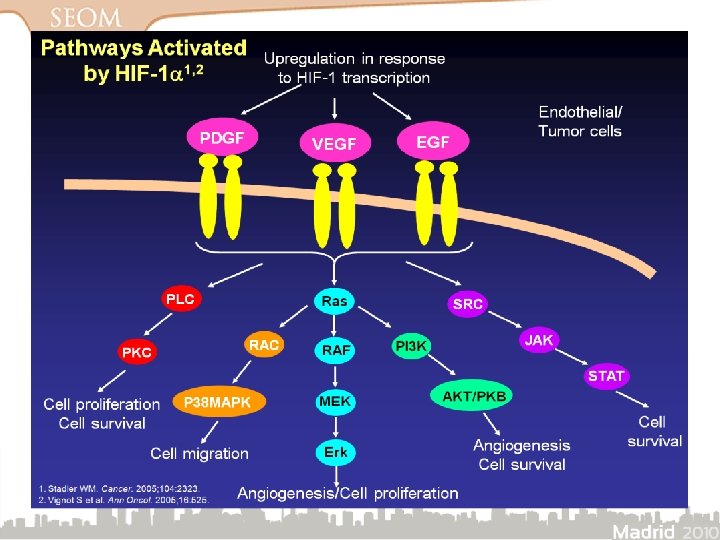
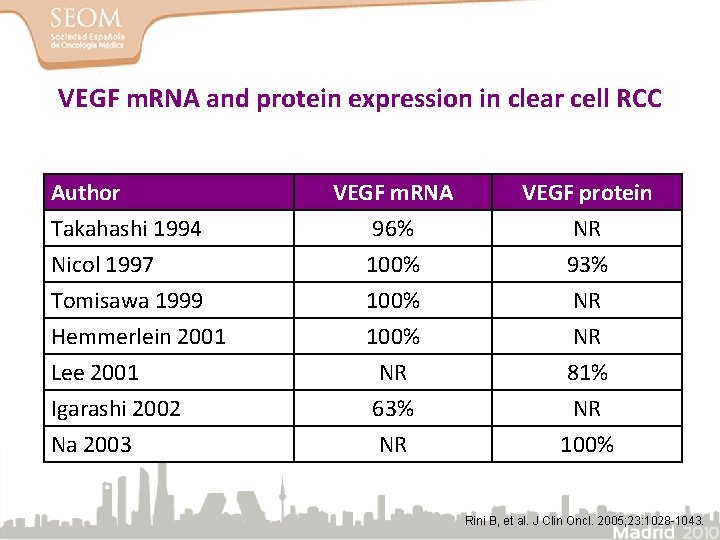
VEGF m. RNA and protein expression in clear cell RCC Author Takahashi 1994 Nicol 1997 Tomisawa 1999 Hemmerlein 2001 Lee 2001 Igarashi 2002 Na 2003 VEGF m. RNA 96% 100% VEGF protein NR 93% NR 100% NR 63% NR NR 81% NR 100% Rini B, et al. J Clin Oncl. 2005; 23: 1028 -1043.
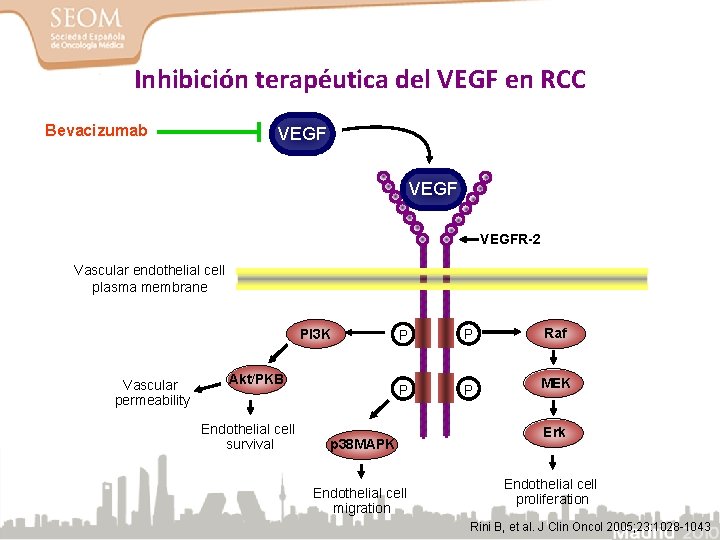
Inhibición terapéutica del VEGF en RCC Bevacizumab VEGFR-2 Vascular endothelial cell plasma membrane PI 3 K Vascular permeability Akt/PKB Endothelial cell survival P P Raf P P MEK p 38 MAPK Endothelial cell migration Erk Endothelial cell proliferation Rini B, et al. J Clin Oncol 2005; 23: 1028 -1043
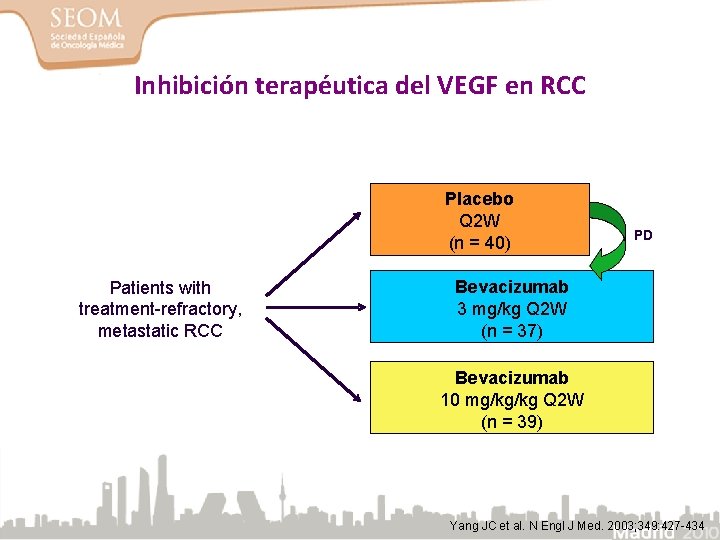
Inhibición terapéutica del VEGF en RCC Placebo Q 2 W (n = 40) Patients with treatment-refractory, metastatic RCC PD Bevacizumab 3 mg/kg Q 2 W (n = 37) Bevacizumab 10 mg/kg/kg Q 2 W (n = 39) Yang JC et al. N Engl J Med. 2003; 349: 427 -434
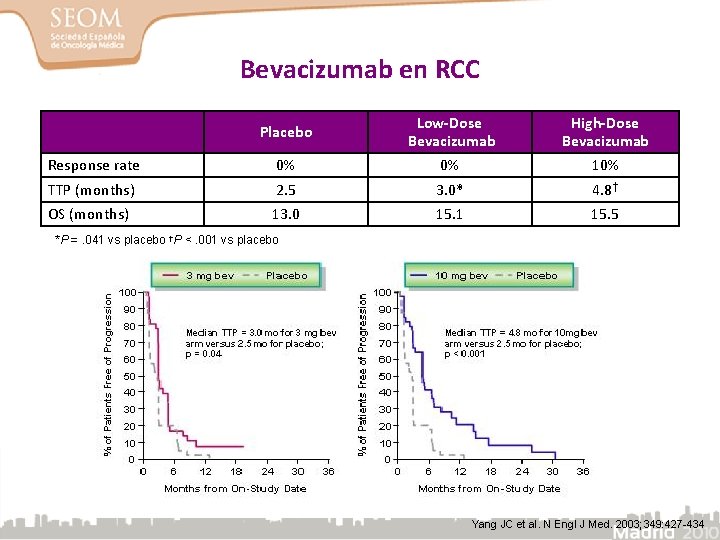
Bevacizumab en RCC Placebo Low-Dose Bevacizumab High-Dose Bevacizumab Response rate 0% 0% 10% TTP (months) 2. 5 3. 0* 4. 8† OS (months) 13. 0 15. 1 15. 5 *P =. 041 vs placebo †P <. 001 vs placebo Yang JC et al. N Engl J Med. 2003; 349: 427 -434
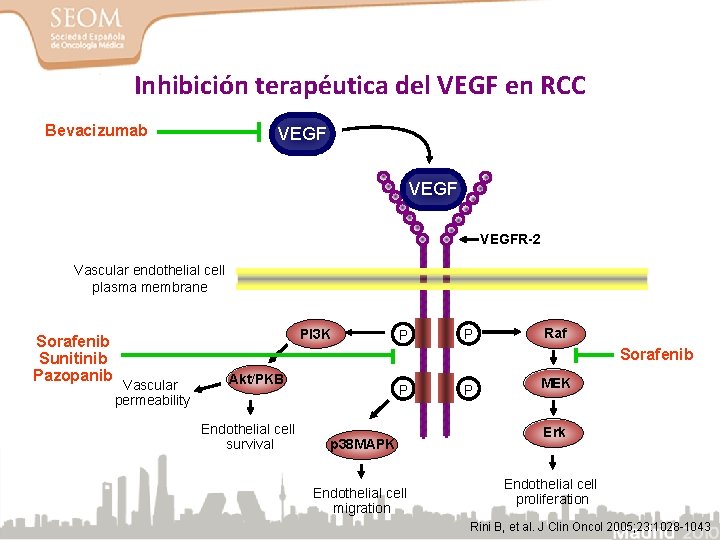
Inhibición terapéutica del VEGF en RCC Bevacizumab VEGFR-2 Vascular endothelial cell plasma membrane Sorafenib Sunitinib Pazopanib PI 3 K P P Raf Sorafenib Vascular permeability Akt/PKB Endothelial cell survival P p 38 MAPK Endothelial cell migration P MEK Erk Endothelial cell proliferation Rini B, et al. J Clin Oncol 2005; 23: 1028 -1043
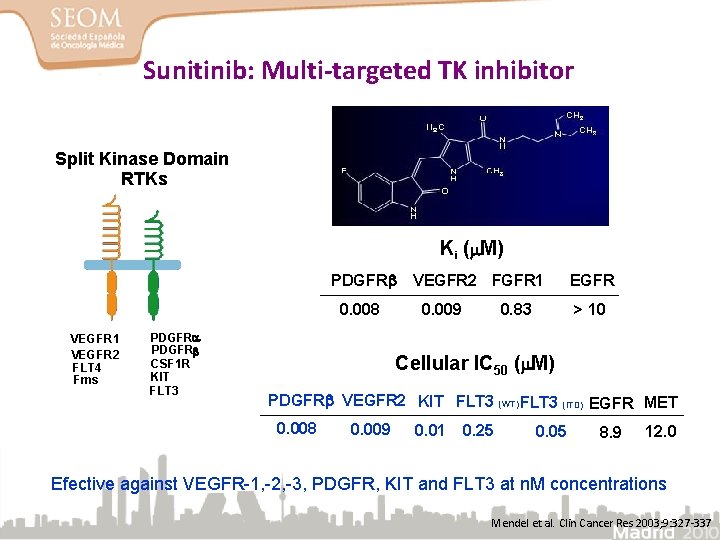
Sunitinib: Multi-targeted TK inhibitor Split Kinase Domain RTKs Ki (m. M) PDGFRb 0. 008 VEGFR 1 VEGFR 2 FLT 4 Fms PDGFRb CSF 1 R KIT FLT 3 VEGFR 2 FGFR 1 0. 009 EGFR > 10 0. 83 Cellular IC 50 (m. M) PDGFRb VEGFR 2 KIT FLT 3 (WT) FLT 3 (ITD) EGFR MET 0. 008 0. 009 0. 01 0. 25 0. 05 8. 9 12. 0 Efective against VEGFR-1, -2, -3, PDGFR, KIT and FLT 3 at n. M concentrations Mendel et al. Clin Cancer Res 2003; 9: 327 -337
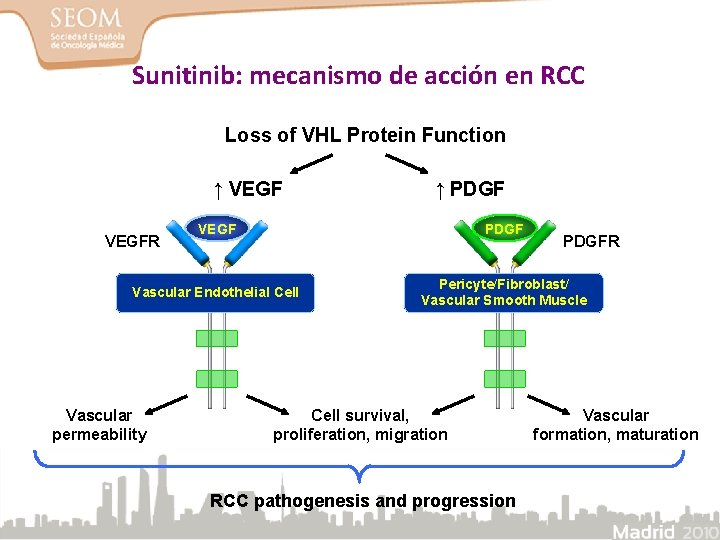
Sunitinib: mecanismo de acción en RCC Loss of VHL Protein Function ↑ VEGF ↑ PDGF VEGF PDGF Vascular Endothelial Cell Pericyte/Fibroblast/ Vascular Smooth Muscle VEGFR Vascular permeability Cell survival, proliferation, migration RCC pathogenesis and progression PDGFR Vascular formation, maturation
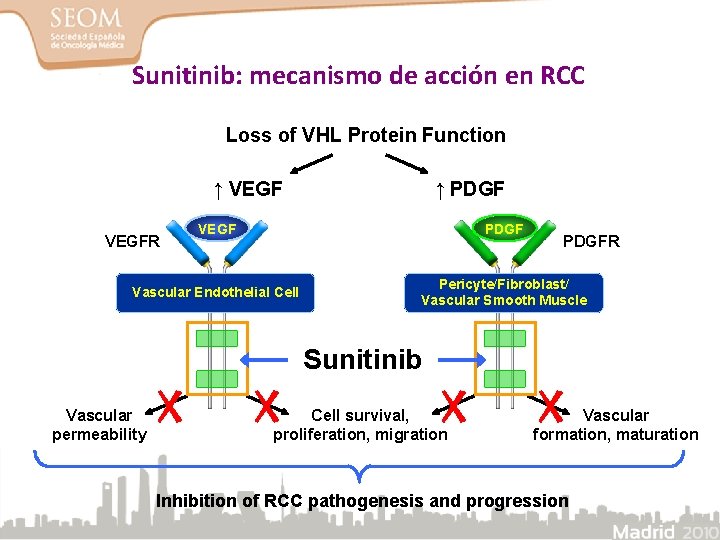
Sunitinib: mecanismo de acción en RCC Loss of VHL Protein Function ↑ VEGF ↑ PDGF VEGF PDGF Vascular Endothelial Cell Pericyte/Fibroblast/ Vascular Smooth Muscle VEGFR PDGFR Sunitinib Vascular permeability Cell survival, proliferation, migration Vascular formation, maturation Inhibition of RCC pathogenesis and progression
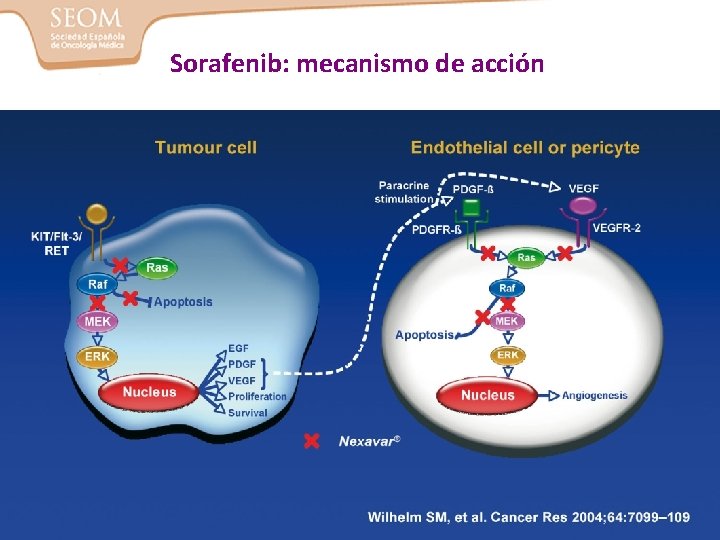
Sorafenib: mecanismo de acción
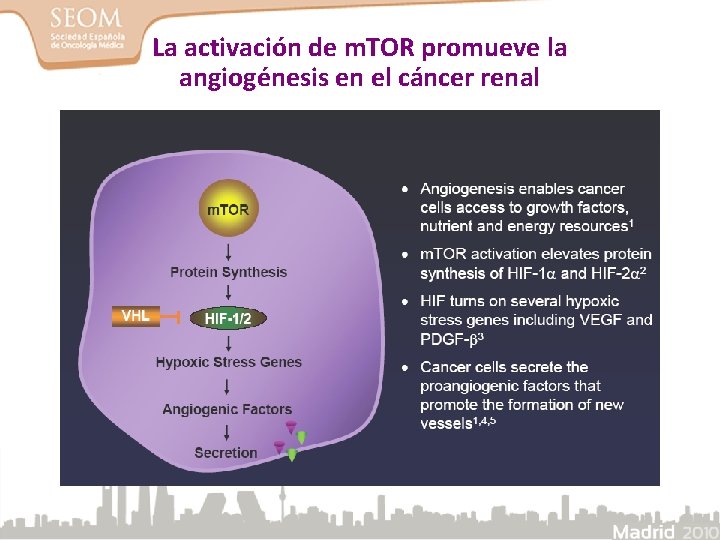
La activación de m. TOR promueve la angiogénesis en el cáncer renal
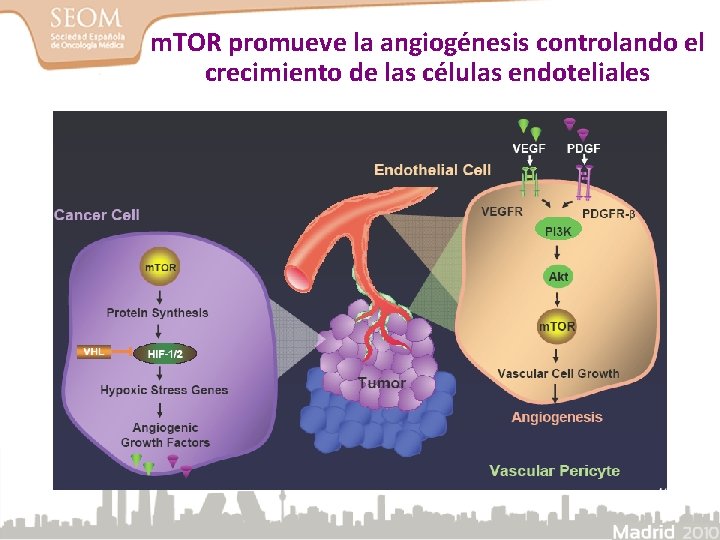
m. TOR promueve la angiogénesis controlando el crecimiento de las células endoteliales
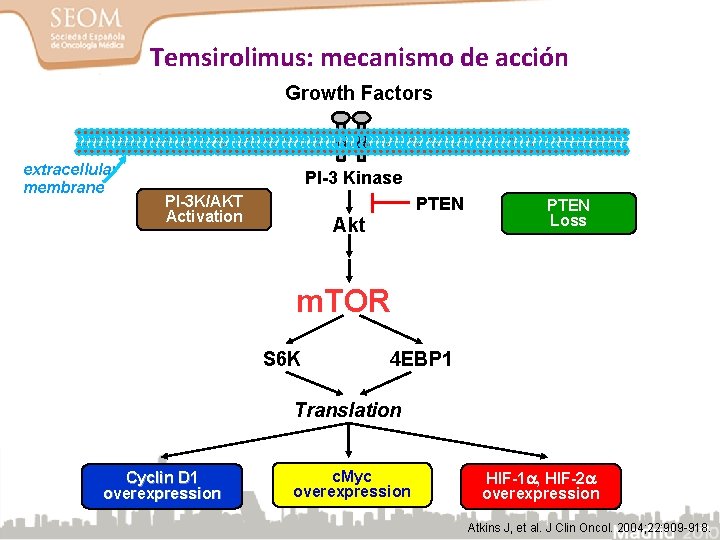
Temsirolimus: mecanismo de acción Growth Factors extracellular membrane PI-3 Kinase PI-3 K/AKT Activation PTEN Akt PTEN Loss m. TOR S 6 K 4 EBP 1 Translation Cyclin D 1 overexpression c. Myc overexpression HIF-1 , HIF-2 overexpression 29 Atkins J, et al. J Clin Oncol. 2004; 22: 909 -918.
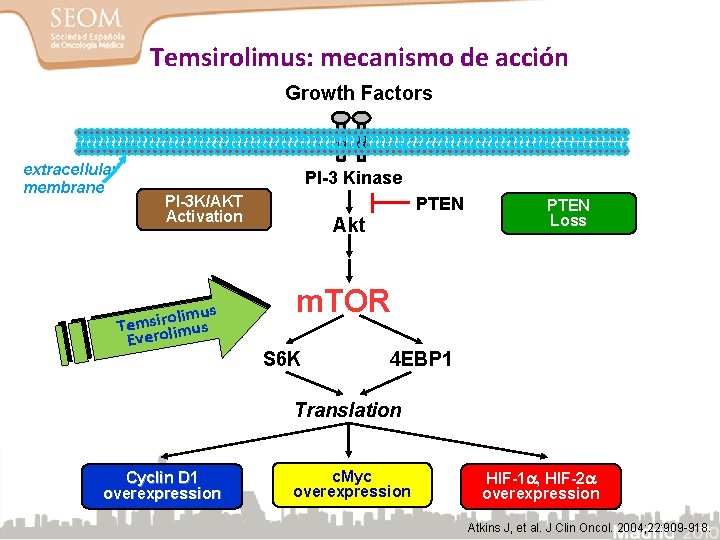
Temsirolimus: mecanismo de acción Growth Factors extracellular membrane PI-3 Kinase PI-3 K/AKT Activation limus o r i s m e T us Everolim PTEN Akt PTEN Loss m. TOR S 6 K 4 EBP 1 Translation Cyclin D 1 overexpression c. Myc overexpression HIF-1 , HIF-2 overexpression 30 Atkins J, et al. J Clin Oncol. 2004; 22: 909 -918.
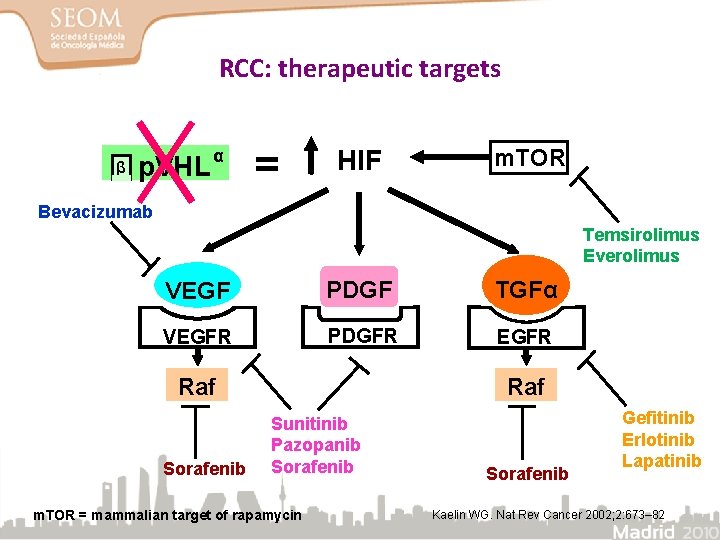
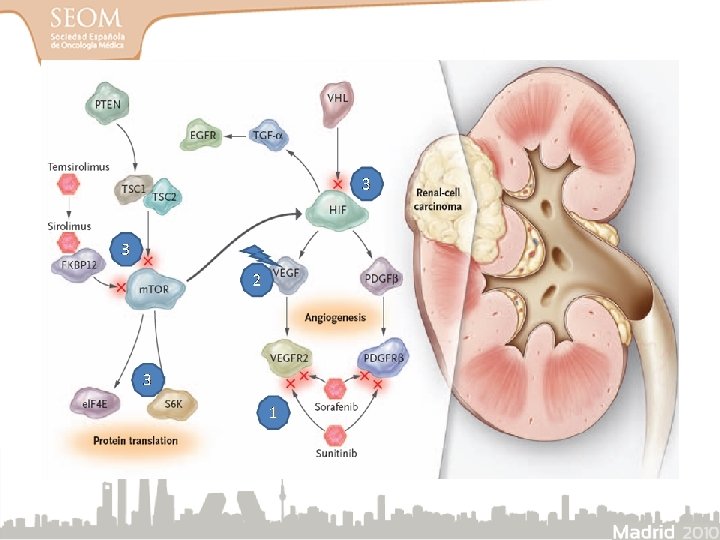
RCC: therapeutic targets β p. VHL α = HIF m. TOR Bevacizumab Temsirolimus Everolimus VEGF PDGF TGFα VEGFR PDGFR EGFR Raf Sorafenib Raf Sunitinib Pazopanib Sorafenib m. TOR = mammalian target of rapamycin Sorafenib Gefitinib Erlotinib Lapatinib Kaelin WG. Nat Rev Cancer 2002; 2: 673– 82
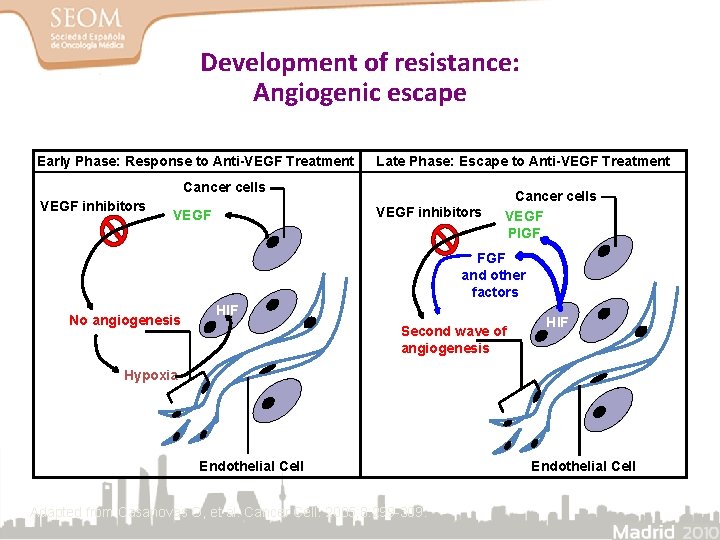
Development of resistance: Angiogenic escape Early Phase: Response to Anti-VEGF Treatment Late Phase: Escape to Anti-VEGF Treatment Cancer cells VEGF inhibitors VEGF Cancer cells VEGF PIGF FGF and other factors No angiogenesis HIF Second wave of angiogenesis HIF Hypoxia Endothelial Cell Adapted from Casanovas O, et al. Cancer Cell. 2005; 8: 299 -309. Endothelial Cell
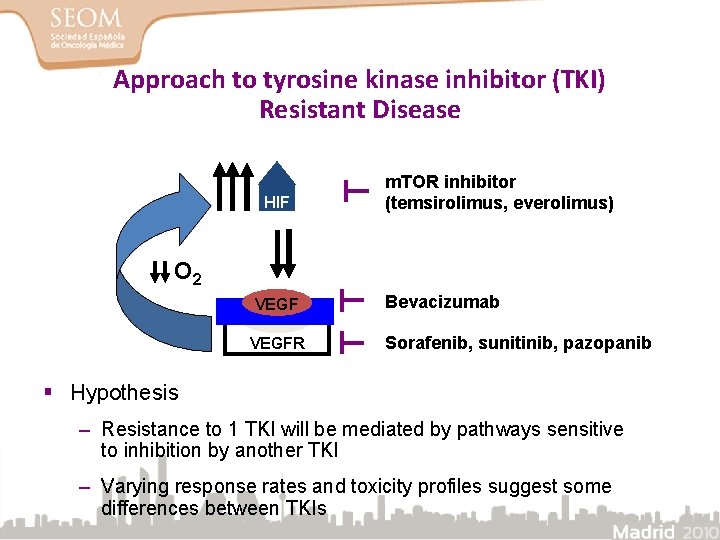
Approach to tyrosine kinase inhibitor (TKI) Resistant Disease HIF m. TOR inhibitor (temsirolimus, everolimus) O 2 VEGFR Bevacizumab Sorafenib, sunitinib, pazopanib § Hypothesis – Resistance to 1 TKI will be mediated by pathways sensitive to inhibition by another TKI – Varying response rates and toxicity profiles suggest some differences between TKIs
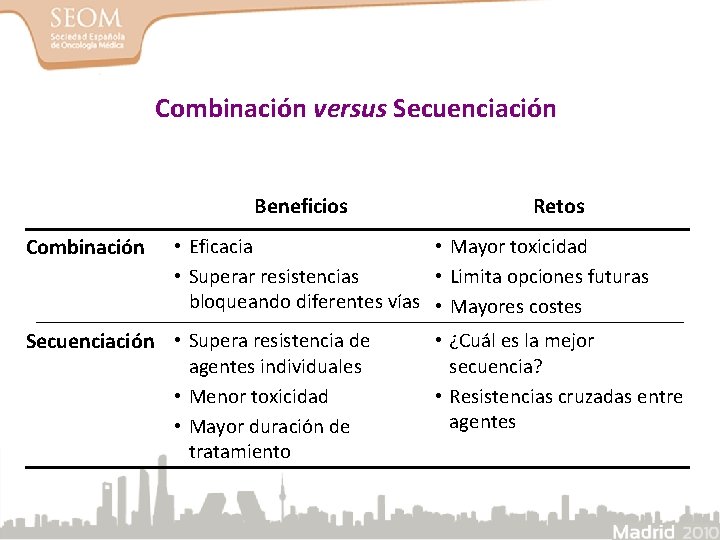
Combinación versus Secuenciación Beneficios Combinación Retos • Eficacia • Mayor toxicidad • Superar resistencias • Limita opciones futuras bloqueando diferentes vías • Mayores costes Secuenciación • Supera resistencia de agentes individuales • Menor toxicidad • Mayor duración de tratamiento • ¿Cuál es la mejor secuencia? • Resistencias cruzadas entre agentes
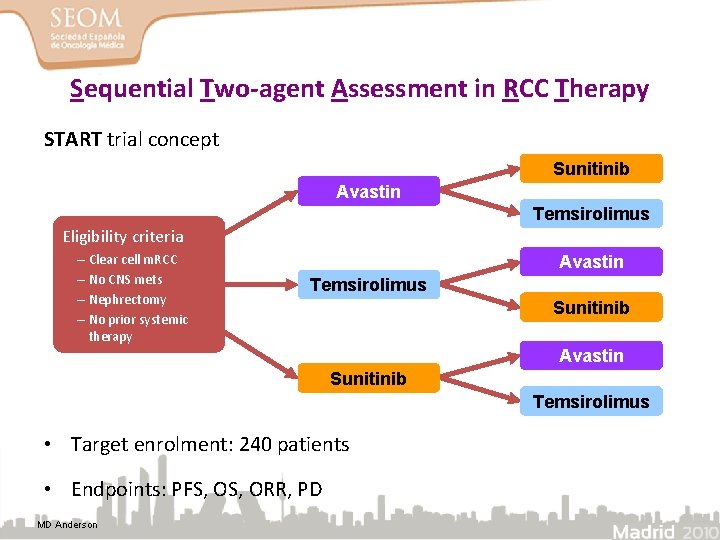
Sequential Two-agent Assessment in RCC Therapy START trial concept Sunitinib Avastin Temsirolimus Eligibility criteria – Clear cell m. RCC – No CNS mets – Nephrectomy – No prior systemic therapy Avastin Temsirolimus Sunitinib Avastin Sunitinib Temsirolimus • Target enrolment: 240 patients • Endpoints: PFS, ORR, PD MD Anderson
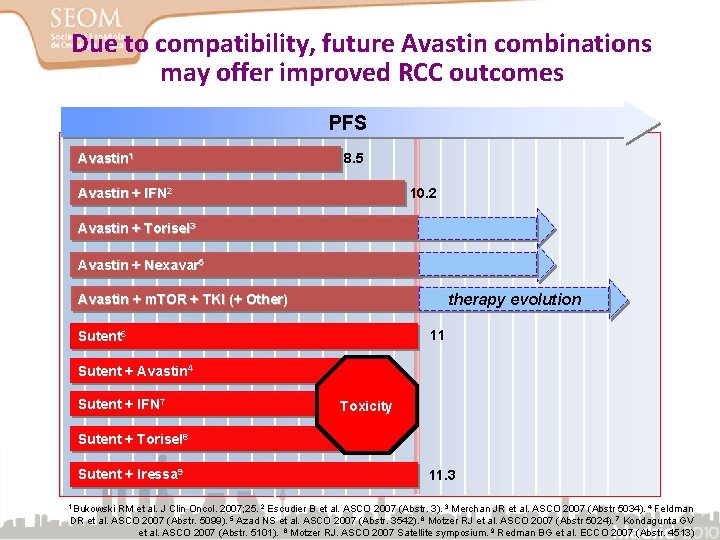
Due to compatibility, future Avastin combinations may offer improved RCC outcomes PFS Avastin 1 8. 5 Avastin + IFN 2 10. 2 Avastin + Torisel 3 Avastin + Nexavar 5 therapy evolution Avastin + m. TOR + TKI (+ Other) 11 Sutent 6 Sutent + Avastin 4 Sutent + IFN 7 Toxicity Sutent + Torisel 8 Sutent + Iressa 9 1 Bukowski 11. 3 RM et al. J Clin Oncol. 2007; 25. 2 Escudier B et al. ASCO 2007 (Abstr. 3). 3 Merchan JR et al. ASCO 2007 (Abstr 5034). 4 Feldman DR et al. ASCO 2007 (Abstr. 5099). 5 Azad NS et al. ASCO 2007 (Abstr. 3542). 6 Motzer RJ et al. ASCO 2007 (Abstr 5024). 7 Kondagunta GV et al. ASCO 2007 (Abstr. 5101). 8 Motzer RJ. ASCO 2007 Satellite symposium. 9 Redman BG et al. ECCO 2007 (Abstr. 4513)
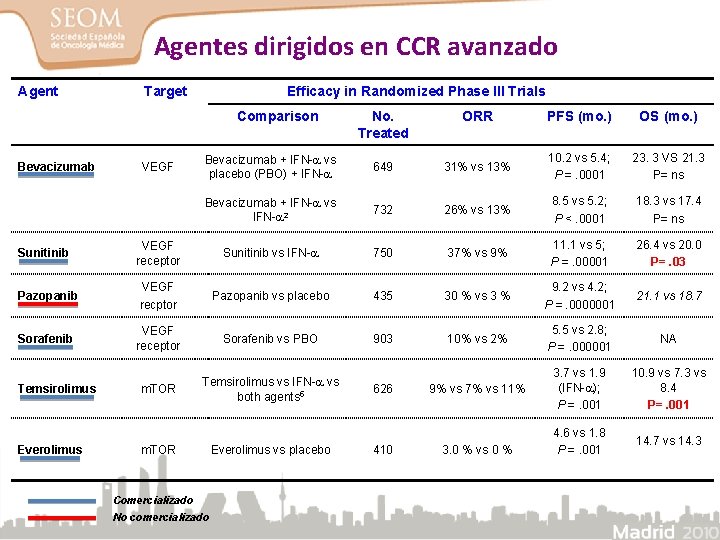
Agentes dirigidos en CCR avanzado Agent Target Efficacy in Randomized Phase III Trials Comparison Bevacizumab VEGF No. Treated ORR PFS (mo. ) OS (mo. ) Bevacizumab + IFN- vs placebo (PBO) + IFN- 649 31% vs 13% 10. 2 vs 5. 4; P =. 0001 23. 3 VS 21. 3 P= ns Bevacizumab + IFN- vs IFN- 2 732 26% vs 13% 8. 5 vs 5. 2; P <. 0001 18. 3 vs 17. 4 P= ns Sunitinib VEGF receptor Sunitinib vs IFN- 750 37% vs 9% 11. 1 vs 5; P =. 00001 26. 4 vs 20. 0 P=. 03 Pazopanib VEGF recptor Pazopanib vs placebo 435 30 % vs 3 % 9. 2 vs 4. 2; P =. 0000001 21. 1 vs 18. 7 Sorafenib VEGF receptor Sorafenib vs PBO 903 10% vs 2% 5. 5 vs 2. 8; P =. 000001 NA m. TOR Temsirolimus vs IFN- vs both agents 5 9% vs 7% vs 11% 3. 7 vs 1. 9 (IFN- ); P =. 001 10. 9 vs 7. 3 vs 8. 4 P=. 001 4. 6 vs 1. 8 P =. 001 14. 7 vs 14. 3 Temsirolimus Everolimus m. TOR Comercializado No comercializado Everolimus vs placebo 626 410 3. 0 % vs 0 %

Avances de SG en CCR avanzado 223. 3
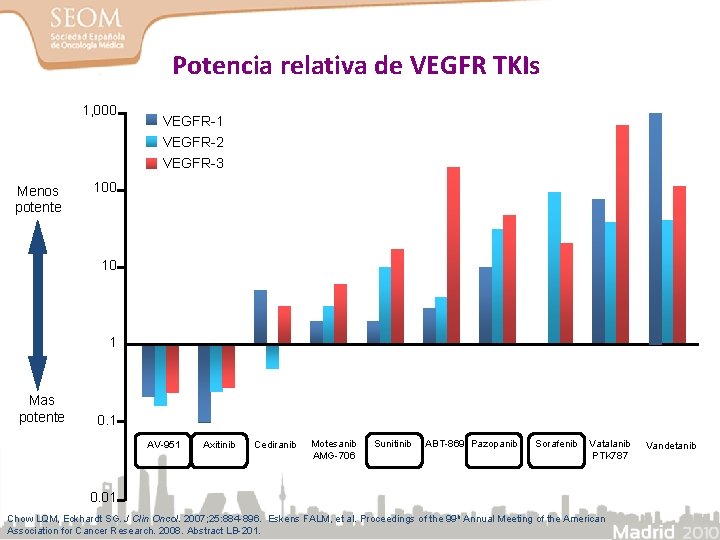
Potencia relativa de VEGFR TKIs 1, 000 VEGFR-1 VEGFR-2 VEGFR-3 100 Potency: IC 50 (n. M) Menos potente Mas potente 10 1 0. 1 AV-951 Axitinib Cediranib Motesanib AMG-706 Sunitinib ABT-869 Pazopanib Sorafenib Vatalanib PTK 787 0. 01 Chow LQM, Eckhardt SG. J Clin Oncol. 2007; 25: 884 -896. Eskens FALM, et al. Proceedings of the 99 th Annual Meeting of the American Association for Cancer Research. 2008. Abstract LB-201. Vandetanib
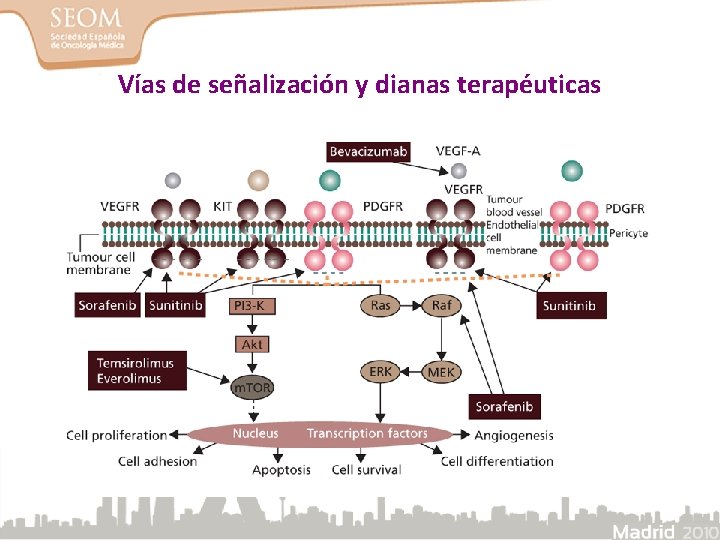
Vías de señalización y dianas terapéuticas
- Slides: 40