BASED ON POLARITY Paper Chromatography https www youtube

BASED ON POLARITY

Paper Chromatography https: //www. youtube. com/watch? v=ej 2 z. XOw. ASVI
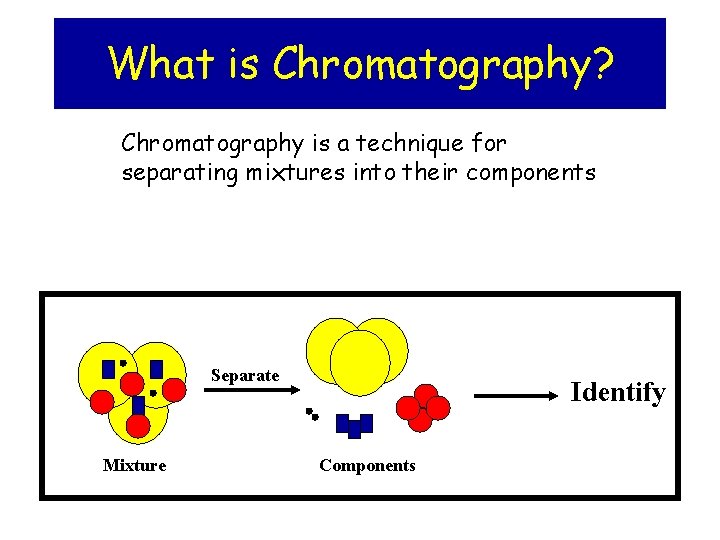
What is Chromatography? Chromatography is a technique for separating mixtures into their components Separate Mixture Identify Components

Uses for Chromatography Real-life examples of uses for chromatography: 1. Hospital – detect alcohol/drug levels in blood 2. Police – to compare a sample at a crime scene to a suspect sample 3. Environmental Agency – find pollutants in the water supply

Definition of Chromatography Detailed Definition: Chromatography separates components (parts) within a mixture by using the differential affinities of the components for the mobile medium and for the stationary medium (absorbing) Terminology: Differential – difference Affinity – attraction (polar or nonpolar) Mobile Medium – the moving part (solvent, like water or alcohol). The liquid (or gas) that carries the mixture. Stationary Medium – the part that does not move (the paper or gel) What is important is how much they adsorb on the stationary phase versus how much they dissolve in the mobile phase
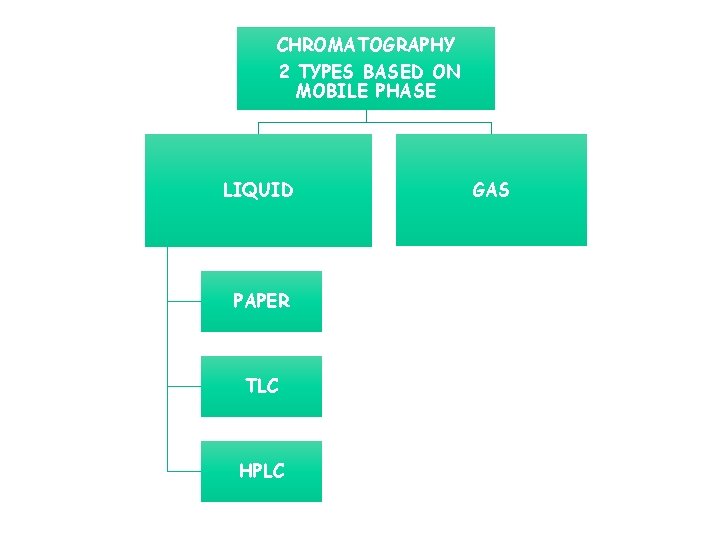
CHROMATOGRAPHY 2 TYPES BASED ON MOBILE PHASE LIQUID PAPER TLC HPLC GAS C HAA F R D

TLC AND PAPER https: //www. youtube. com/watch? v=J 8 r 8 h. N 05 x. Xk
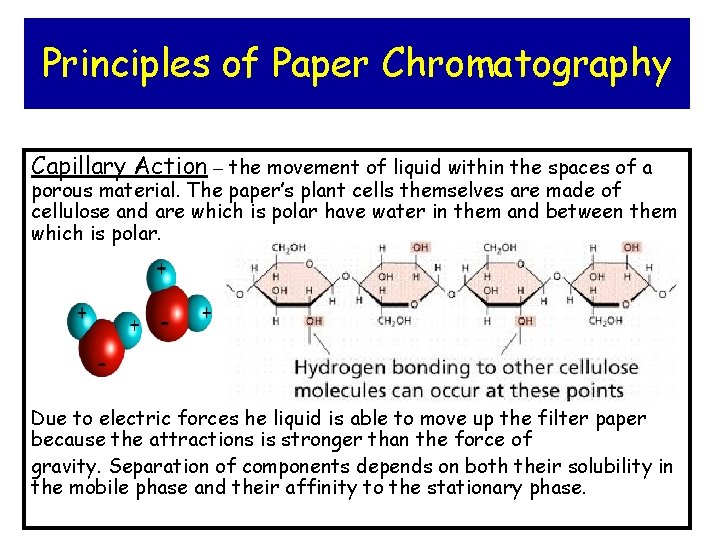
Principles of Paper Chromatography Capillary Action – the movement of liquid within the spaces of a porous material. The paper’s plant cells themselves are made of cellulose and are which is polar have water in them and between them which is polar. Due to electric forces he liquid is able to move up the filter paper because the attractions is stronger than the force of gravity. Separation of components depends on both their solubility in the mobile phase and their affinity to the stationary phase.
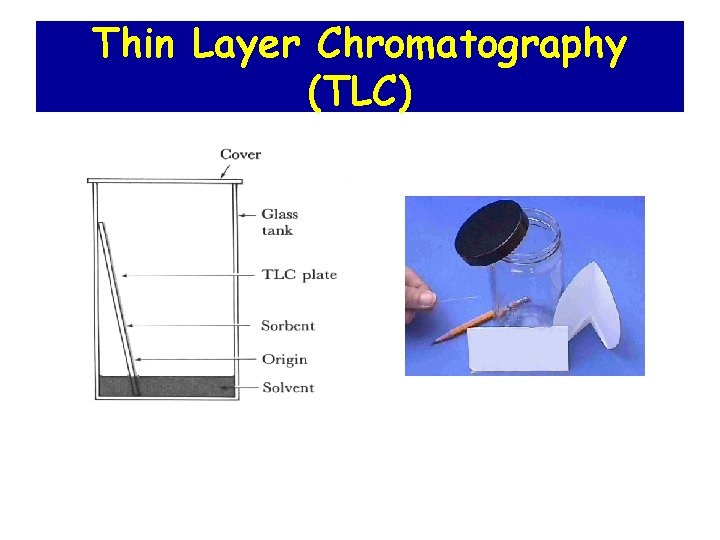
Thin Layer Chromatography (TLC)
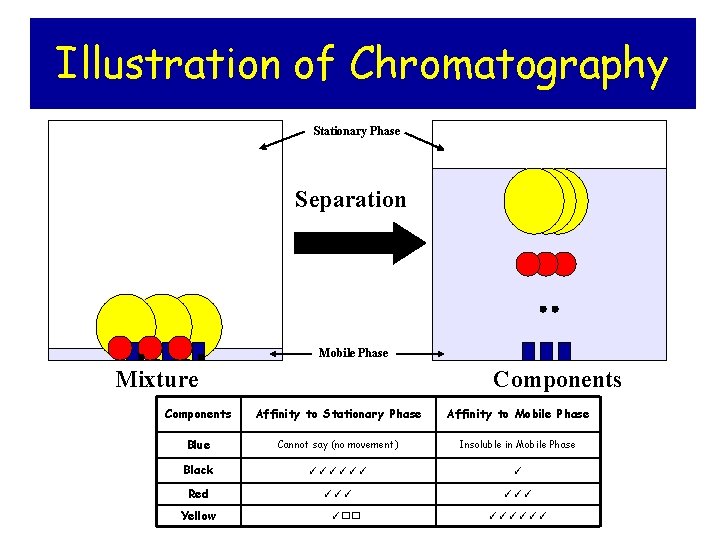
Illustration of Chromatography Stationary Phase Separation Mobile Phase Mixture Components Affinity to Stationary Phase Affinity to Mobile Phase Blue Cannot say (no movement) Insoluble in Mobile Phase Black ✓✓✓✓✓✓ ✓ Red ✓✓✓ Yellow ✓�� ✓✓✓✓✓✓
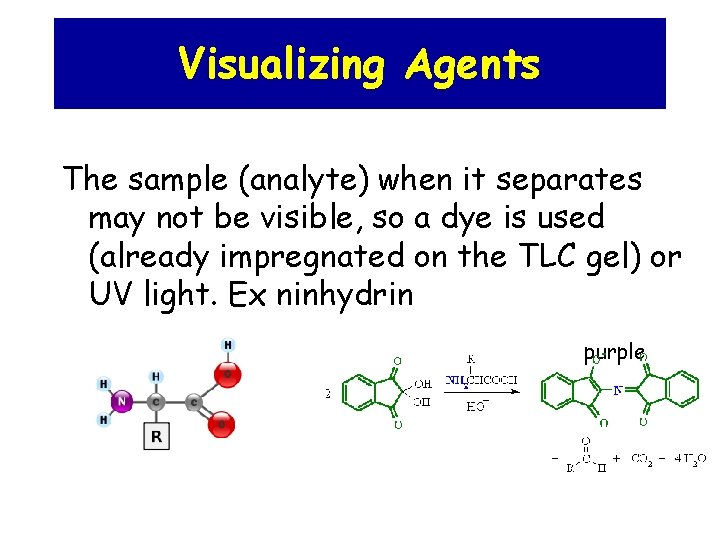
Visualizing Agents The sample (analyte) when it separates may not be visible, so a dye is used (already impregnated on the TLC gel) or UV light. Ex ninhydrin purple

Rf = distance moved by substance distance solvent front Substances that are soluble in the liquid the Rf will be close to. . 1, as it heads to the top For substances that are rather insoluble in the liquid R f will be close to. . 0, they don’t move Thus all numbers must never be more than 1 for Rf Notice Retardation factor (above) is based on distance for Paper and TLC. Next we visit HPLC and GLC which will not measure Distance, rater the measure retention time spent inside a column.

Column Chromatography Adsorbents: Alumina & Silica gel (is porous sand) in which more polar molecules are adsorbed more strongly & thus, will elute more slowly

COLUMN CHROMATOGRAPHY Stationary phase silica Mobile phase suitable organic solvent Separation components interact with the stationary phase to different extents Method • a chromatography column is filled with solvent and silica • drops of the mixture (analyte) are placed on top of the silica - A • the tap is opened to allow the solvent to flow out • components travel through at different rates and separate - B • batches of solvent are collected at intervals - C • the solvent in each batch is evaporated to obtain components A B B C

HIGH PRESSURE LIQUID CHROMATOGRAPHY (HPLC) A better form of column chromatography. Instead of draining down through the stationary phase, the solvent is forced through under high pressure. Stationary phase silica (tiny solid micro beads) Mobile phase suitable solvent Separation similar to column chromatography Alumina is also used (Al 2 O 3) with Iron impurities you get a sapphire And chromium impurities = ruby
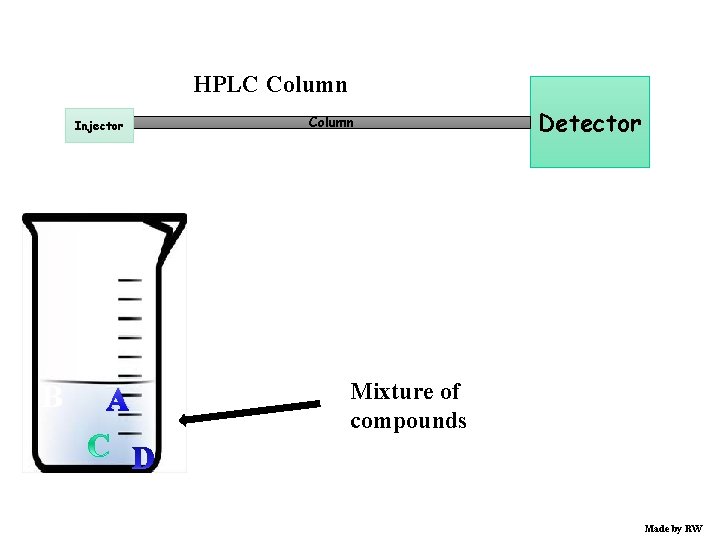
HPLC Column Injector B A Column Detector Mixture of compounds Made by RW

Injector B A Column Detector
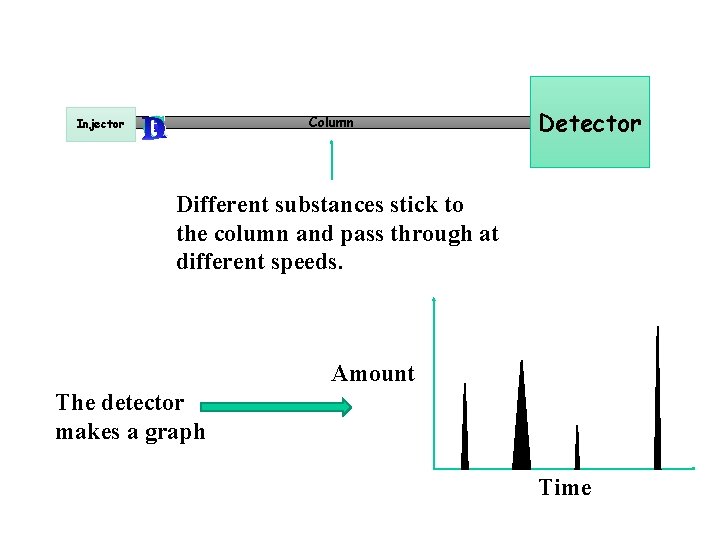
Injector B A Column Detector Different substances stick to the column and pass through at different speeds. Amount The detector makes a graph Time

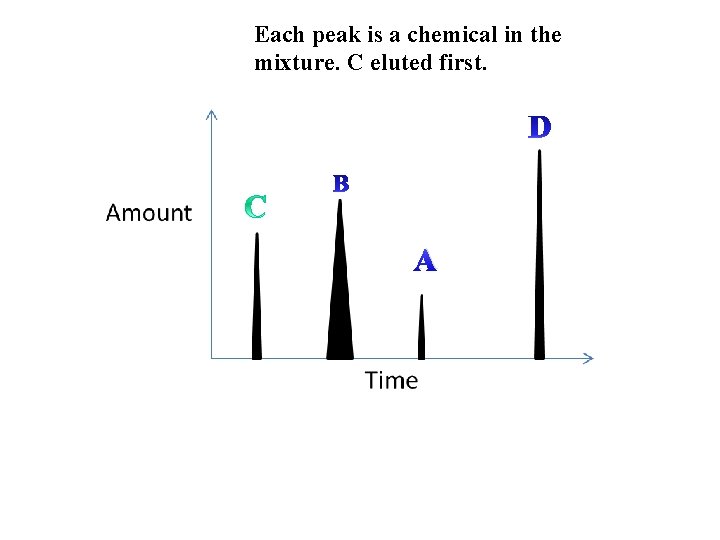
Each peak is a chemical in the mixture. C eluted first. B A

HLPC https: //www. youtube. com/watch? v=a 2 w wg. LV 80 U 8

GAS LIQUID CHROMATOGRAPHY (GLC) Stationary phase liquid adsorbed on an inert solid support Mobile phase gas Method a sample is injected and vaporized then carried along by an inert gas column contains a liquid stationary phase, adsorbed onto an inert solid the time taken to travel through the tube will depend on how much time is spent moving with the gas rather than being attached to the liquid.

GAS LIQUID CHROMATOGRAPHY (GLC) Detection one method is an FID - flame ionisation detector The FID • as a component exits, it is burned in a hydrogen flame • ions are produced • detected as the ions produce an electric current • the greater the amount = larger current • the current can be represented by a chromatogram

GLC https: //www. youtube. com/watch? v=08 Y Wh. LTjlfo

What happens in practice. • Compounds that have high affinity for mobile phase emerge first, (most volatile). • Chromatogram charts recorder response against time. • Retention time – characteristic of the compound under given conditions.
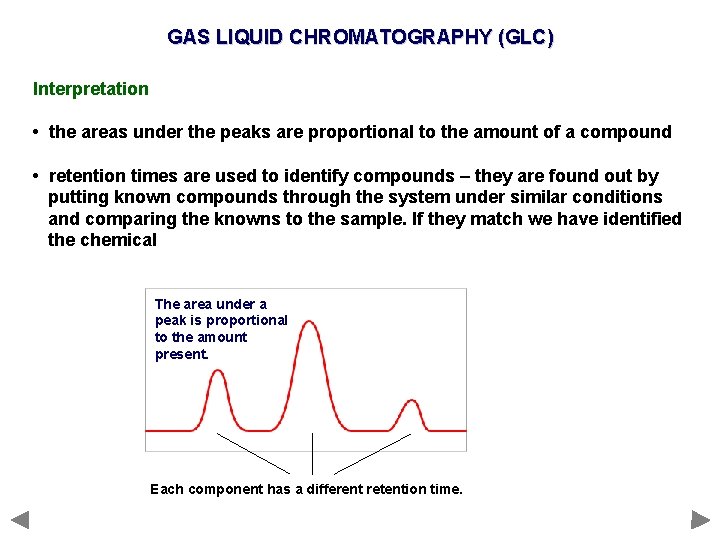
GAS LIQUID CHROMATOGRAPHY (GLC) Interpretation • the areas under the peaks are proportional to the amount of a compound • retention times are used to identify compounds – they are found out by putting known compounds through the system under similar conditions and comparing the knowns to the sample. If they match we have identified the chemical The area under a peak is proportional to the amount present. Each component has a different retention time.

Summary 4 Types of Chromatography Types Stationary Phase Moblie Phase Sample Paper (Liquid chromo) Paper (solid) Polar and nonpolar mixture of liquid solvents Dried onto stationary paper TLC (Liquid chromo) Silica (solid, Si. O 2) Polar and nonpolar mixture of liquid solvents Dried onto stationary gel HPLC (Liquid chromo) Silica (solid) and solvent slurry Polar and nonpolar mixture of liquid solvents Dissolved into the mobile phase of the gas Gas (Gas chromo) Beads (solid and Liquid waxy Inert gas (N 2, Hel polymer( Cellulose, coupled with water absorbed within the fibers) Heated to a Gas to go with N 2 or He
Interesting $1 Centrifuge https: //www. youtube. com/watch? v=L 5 p p. D 07 DMKQ
- Slides: 28