Basal Bolus The Strategy for Managing All Diabetes

Basal Bolus: The Strategy for Managing All Diabetes Fall, 2003 Paul Davidson, MD, FACE Atlanta Diabetes Associates Atlanta, Georgia

ACE / AACE Targets for Glycemic Control Hb. A 1 c < 6. 5 % Fasting/preprandial glucose < 110 mg/d. L Postprandial glucose < 140 mg/d. L ACE / AACE Consensus Conference, Washington DC August 2001

Step Therapy l Diet l Exercise l Sulfonylurea or Metformin l Add Alternate Agent l Add hs NPH l Switch to Mixed Insulin bid l Switch to Multiple Dose Insulin Utilitarian, Common Sense, Recommended Prone to Failure from Misscheduling and Mismanagement

Stumble Therapy l YAG Diet l Golf Cart Exercise l Sample of the Week Medication – Interupted, – Not Combined l Poor Understanding of Goals l Poor Monitoring Hb. A 1 c >8% (If Seen) Informed Patient Refers Self Elsewhere

PETS Therapy Step--Spelled Backwards All at once, nothing first, Just like bubbles, when they burst. l Start with Fast to Glucose <126 mg/d. L – Glargine, Wt x 0. 1 units qd l Feed PSMF Diet l Add SU, MF, TZD, Repaglanide + prn Lispro for BG <150 l “Normal” BG from Day 1 l Monitor BG qid l See Patient Monthly, HFP l Hb. A 1 c Bimonthly GI Problems: Cut MF Hypoglycemia: Cut SU Hypoglycemia Again: Cut Repaglinide Allow 2 Month to See TZD Effect

MIMICKING NATURE WITH INSULIN THERAPY All persons need both basal and mealtime insulin to control glucose (endogenous or exogenous) 6 -19

Limitations of NPH, Lente, and Ultralente l Do not mimic basal insulin profile – Variable absorption – Pronounced peaks – Less than 24 -hour duration of action l Cause unpredictable hypoglycemia – Major factor limiting insulin adjustments – More weight gain

Insulin Glargine A New Long-Acting Insulin Analog l Modifications to human insulin chain – Substitution of glycine at position A 21 – Addition of 2 arginines at position B 30 l Gradual release from injection site l Peakless, long-lasting insulin profile Gly 1 5 10 15 20 Asp 1 5 10 15 20 Substitution 25 30 Extension Arg
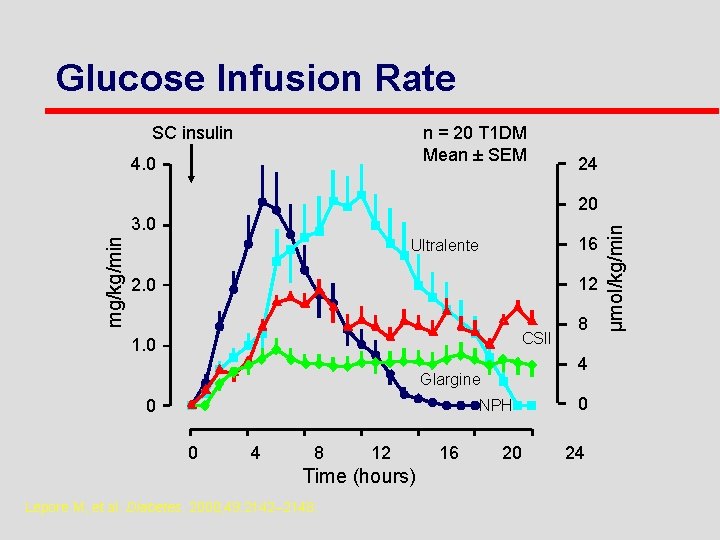
Glucose Infusion Rate SC insulin n = 20 T 1 DM Mean ± SEM 4. 0 24 mg/kg/min 3. 0 16 Ultralente 12 2. 0 CSII 1. 0 4 Glargine 0 NPH 0 4 8 12 Time (hours) Lepore M, et al. Diabetes. 2000; 49: 2142– 2148. 16 8 20 0 24 µmol/kg/min 20
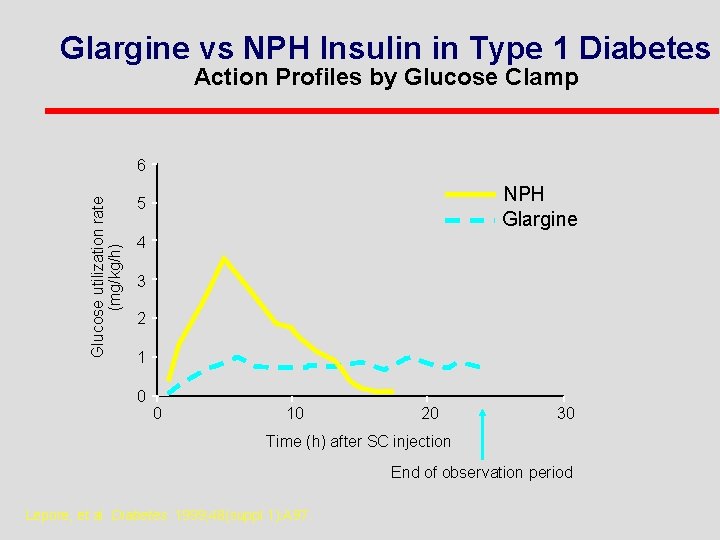
Glargine vs NPH Insulin in Type 1 Diabetes Action Profiles by Glucose Clamp Glucose utilization rate (mg/kg/h) 6 NPH Glargine 5 4 3 2 1 0 0 10 20 30 Time (h) after SC injection End of observation period Lepore, et al. Diabetes. 1999; 48(suppl 1): A 97.

Treat to Target Study: NPH vs Glargine in DM 2 patients on OHA l Add 10 units Basal insulin at bedtime or Glargine) (NPH l Continue current oral agents l Titrate insulin weekly to fasting BG < 100 mg/d. L • Based on average FBG of 6 th and 7 th day - if 100 -120 mg/d. L, increase 2 units - if 120 -140 mg/d. L, increase 4 units - if 140 -160 mg/d. L, increase 6 units - if 160 -180 mg/d. L, increase 8 units
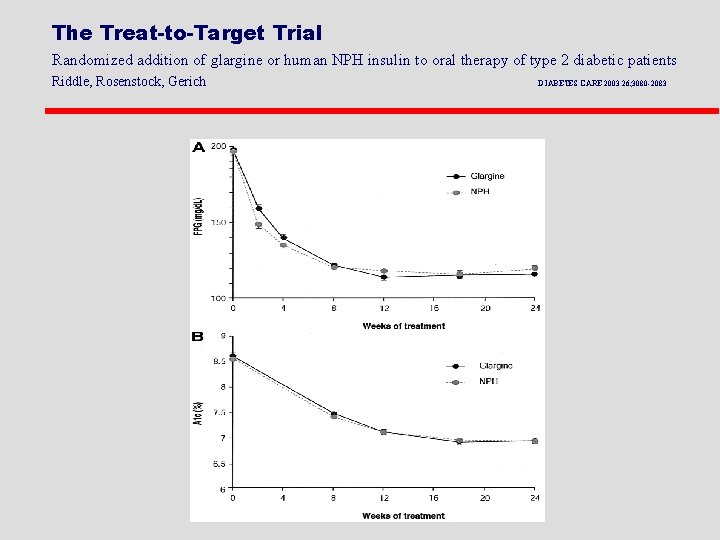
The Treat-to-Target Trial Randomized addition of glargine or human NPH insulin to oral therapy of type 2 diabetic patients Riddle, Rosenstock, Gerich DIABETES CARE 2003 26; 3080 -2083

The Treat-to-Target Trial Randomized addition of glargine or human NPH insulin to oral therapy of type 2 diabetic patients Riddle, Rosenstock, Gerich DIABETES CARE 2003 26; 3080 -2083 Percentage of Patients in Target (A 1 C < 7%)

GEMS--Glargine Evening Mealtime Secretagogue l Basal Dosing – (Weight in #`s x 0. 1) • Glargine hs l Prior to Meals – Short Acting Secretagogue • Rapaglinide 2 mg • Nateglinide 120 mg – Glimepiride 2 mg
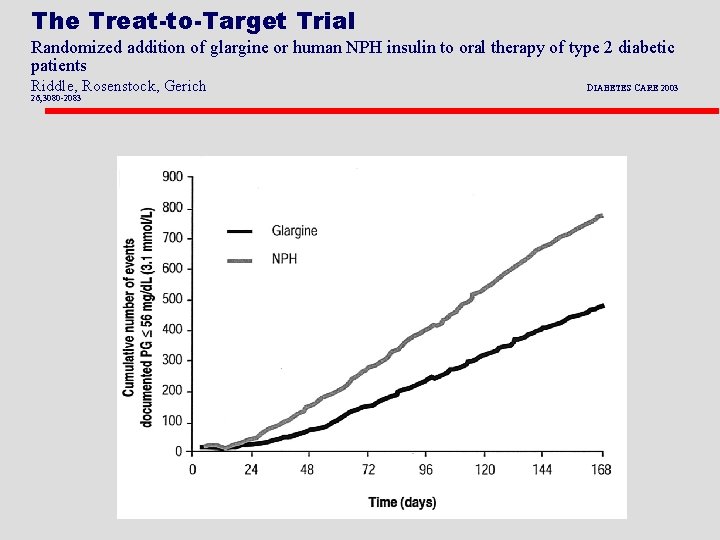
The Treat-to-Target Trial Randomized addition of glargine or human NPH insulin to oral therapy of type 2 diabetic patients Riddle, Rosenstock, Gerich 26; 3080 -2083 DIABETES CARE 2003
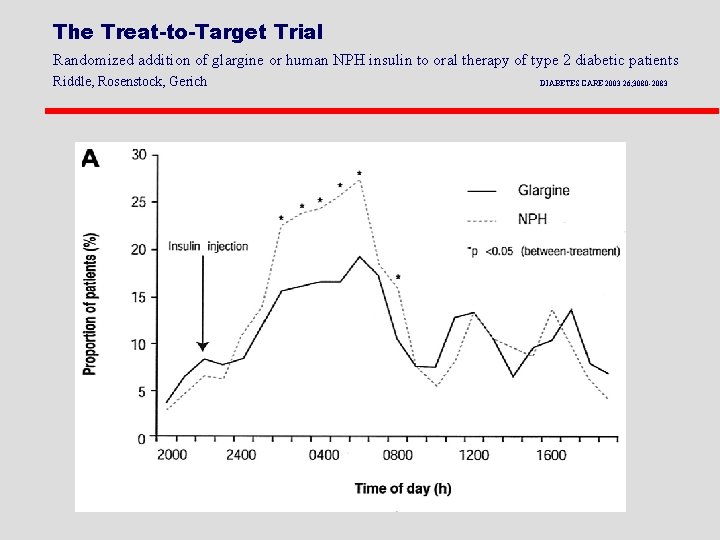
The Treat-to-Target Trial Randomized addition of glargine or human NPH insulin to oral therapy of type 2 diabetic patients Riddle, Rosenstock, Gerich DIABETES CARE 2003 26; 3080 -2083
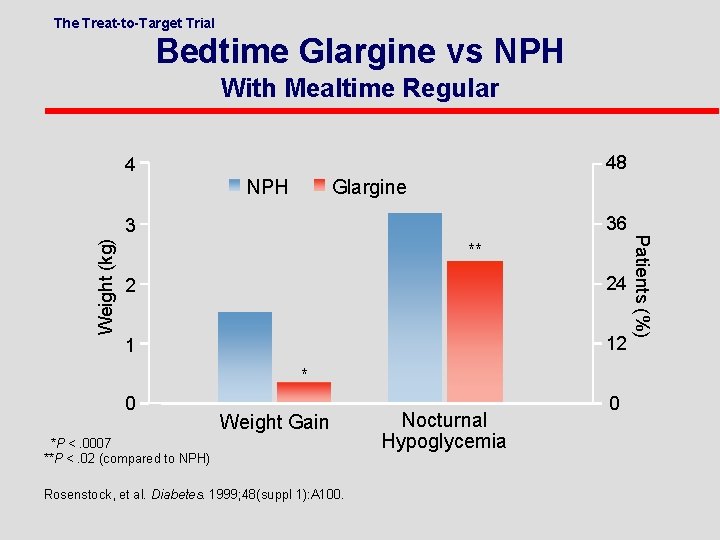
The Treat-to-Target Trial . Bedtime Glargine vs NPH With Mealtime Regular 48 4 Glargine NPH 36 Weight (kg) ** 2 24 1 12 Patients (%) 3 * 0 Weight Gain *P <. 0007 **P <. 02 (compared to NPH) Nocturnal Hypoglycemia 0 Rosenstock, et al. Diabetes. 1999; 48(suppl 1): A 100. 6 -52

Treatment to Target Study: NPH vs Glargine in DM 2 patients on OHA l 57% had Hb. A 1 c <7% l Nocturnal Hypoglycemia reduced by 42% in the Glargine group l 33% had Hb. A 1 c <7% without any nighttime hypoglycemia in glargine group l Results significantly better than with NPH

Establishing Basal Requirement for Glargine Body Weight in pounds x 0. 1 Average am BG x 2 after five days Add to Glargine (BG-100)/10 Repeat weekly Example: 200# 20 units glargine q hs AM BG averages 200 on 6 th and 7 th day Add (BG-100)10 to glargine, i. e. increase to 20 to 30 units q hs 2 nd week--average 130 increase glargine from 30 to 33

Overall Summary: Glargine l Insulin glargine has the following clinical benefits – Once-daily dosing because of its prolonged duration of action and smooth, peakless time-action profile – Comparable or better glycemic control (FBG) – Lower risk of nocturnal hypoglycemic events – Safety profile similar to that of human insulin
- Slides: 20