Barretts esophagus Joint hospital surgical grandround 1672016 Cheung

Barrett’s esophagus Joint hospital surgical grandround 16/7/2016 Cheung Hing Fong

Content n n n Case presentation Prevalence Updated management q n dysplastic, BE nodularity and adenocarcinoma Summary

54/M n Cerebral palsy reflux esophagitis dx in 2000 n on long term PPI n n developed increased symptom in 2014

OGD 2014 n n n SCJ at 30 cm; OGJ at 38 cm; diaphragmatic hiatus at 46 cm huge hiatus hernia 8 cm Prague C 8 M 8 Barrett esophagus a 1. 2 cm raised nodule at 34 cm from incisor biopsy: adenocarcinoma
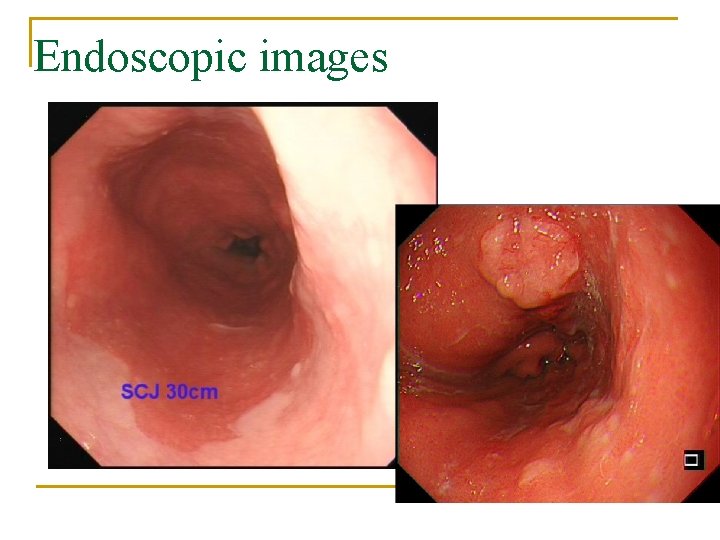
Endoscopic images


Progress q EUS: 1. 2 cm tumour involving mucosa and submucosa, MP preserved; no gross perioesophageal LN CT: no distant met q Ivor Lewis esophagectomy q n n n histology: moderately differentiated ADENOCARCINOMA, with background barrett's esophagus invades into submucosa, T 1 b. N 0 no lymphovascular permeation

Can this Barrett associated early adenocarcinoma be treated endoscopically without surgery?
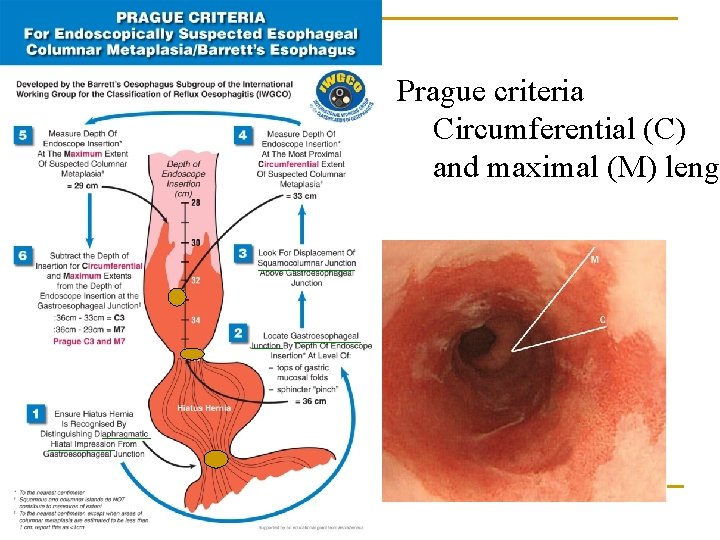
Prague criteria Circumferential (C) and maximal (M) lengt

Prevalence of Barrett’s esophagus in Asia n n Endoscopic BE 7. 8% Histological confirmed BE 1. 3% Pooled prevalence of dysplasia among histological confirmed BE q low grade dysplasia 6. 9% q high grade dysplasia 3% q adenocarcinoma 2% Trend of increased prevalence from 1991 to 2014, especially in Eastern Asian Countries Seiji Shiota, et al Clin Gastroenterol Hepatol. 2015 Nov ; 13(11): 1907 -18

Risk of progression to adenocarcinoma based on dysplasia n n n Non-dysplastic BE: 0. 33% per year Low grade dysplasia: 0. 5% per year High grade dysplasia: 7 -19% per year

Endoscopic surveillance for dysplastic BE Important for detection of dysplasia/ neoplasia Seattle protocol n 4 quadrant bx every 1 cm interval starting from the top of the gastric folds up to the most proximal extent of the BE n mucosal abnormality sampled separately Reid BJ et al. Optimizing endoscopic biopsy detection of early cancers in Barrett's high-grade dysplasia. Am J Gastroenterology 2000
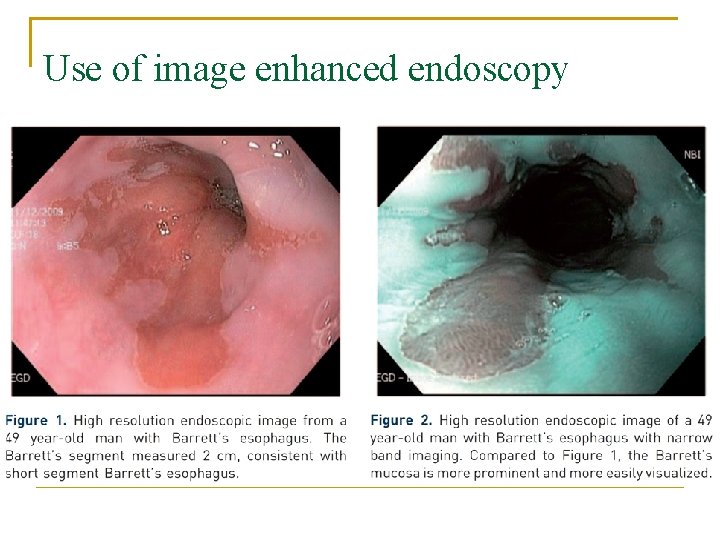
Use of image enhanced endoscopy

Use of image enhanced endoscopy n n n RCT NBI targeted biopsy compared with high definition white light endoscopy random biopsy no difference in detection of intestinal metaplasia NBI detected a higher proportion of areas with dysplasia (30% vs 21%, p=0. 01) Sharma P et al. Standard endoscopy with random biopsies vs. narrow band imaging targeted biopsies in Barrett's oesophagus: a prospective, international, randomised controlled trial. Gut 2013

Risk stratification of Barrett’s surveillance n n Loss of SOX 2 and aberrant p 53 expression increased the risk of neoplastic progression Improve risk stratification of Barrett’s surveillance van Olphen S et al. SOX 2 as a novel marker to predict neoplastic progression in Barrett’s esophagus. Am J Gastroenterol 2015
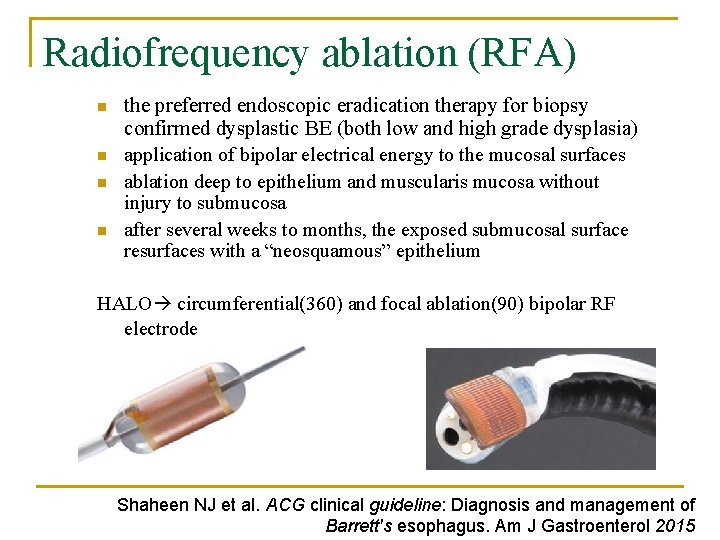
Radiofrequency ablation (RFA) n n the preferred endoscopic eradication therapy for biopsy confirmed dysplastic BE (both low and high grade dysplasia) application of bipolar electrical energy to the mucosal surfaces ablation deep to epithelium and muscularis mucosa without injury to submucosa after several weeks to months, the exposed submucosal surface resurfaces with a “neosquamous” epithelium HALO circumferential(360) and focal ablation(90) bipolar RF electrode Shaheen NJ et al. ACG clinical guideline: Diagnosis and management of Barrett's esophagus. Am J Gastroenterol 2015

RFA for dysplastic BE n High efficacy q n Eradication rate: 90% for LGD, 81% for HGD Durable q Dysplasia remained eradicated in 85% and intestinal metaplasia remained eradicated in 75% of patients after 3 year without maintenance RFA Shaheen NJ et al. Durability of radiofrequency ablation in Barrett's esophagus with dysplasia. Gastroenterology 2011

Radiofrequency ablation (RFA) q good safety profile n n q Stricture risk(1 -6%) Bleeding and perforation rare appear not to interfere with subsequent endoscopic resection for residual lesions n n less submucosal scarring possible to resect residual Barrett’s mucosa after multiple RFA without the need for submucosal lifting
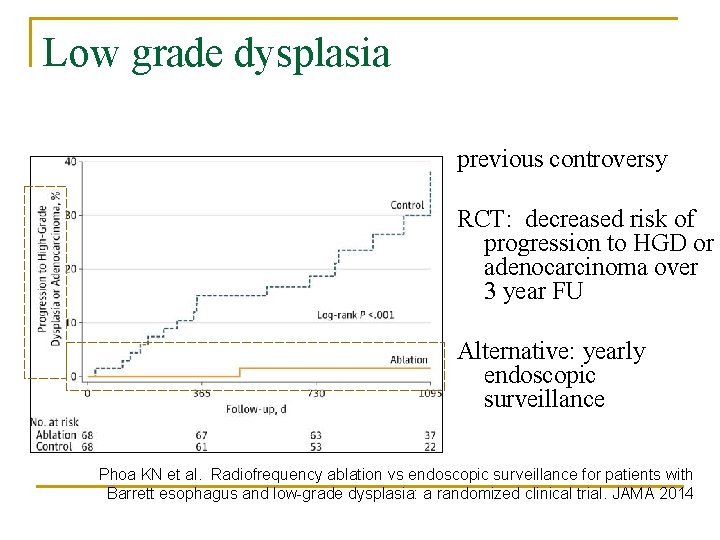
Low grade dysplasia previous controversy RCT: decreased risk of progression to HGD or adenocarcinoma over 3 year FU Alternative: yearly endoscopic surveillance Phoa KN et al. Radiofrequency ablation vs endoscopic surveillance for patients with Barrett esophagus and low-grade dysplasia: a randomized clinical trial. JAMA 2014

Recurrence and disease progression after RFA n n intestinal metaplasia recurrence-13% progression to cancer occurred in 0. 2% of patients during treatment and in 0. 7% of those after complete eradication of dysplasia and intestinal metaplasia (CEIM) intensive endoscopic surveillance needed Orman ES, Li N, Shaheen NJ. Efficacy and durability of radiofrequency ablation for Barrett's Esophagus: systematic review and meta-analysis. Clin Gastroenterol Hepatol 2013

Endoscopic resection (ER) n Endoscopic visible nodularity in BE q q EMR as the initial diagnostic and therapeutic maneuver Histological diagnosis n n Degree of dysplasia/ adenocarcinoma For adenocarcinoma q q q Depth of invasion Risk for lymph node metastasis Complications of EMR: bleeding, stricture, perforation Shaheen NJ et al. ACG clinical guideline: Diagnosis and management of Barrett's esophagus. Am J Gastroenterol 2015
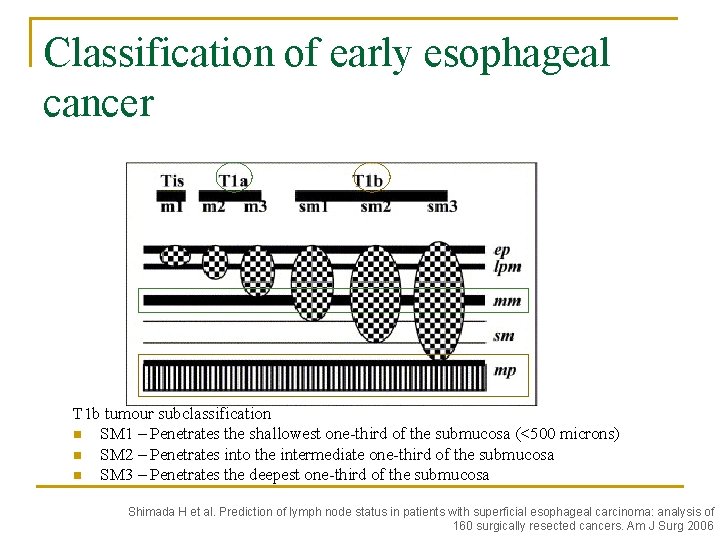
Classification of early esophageal cancer T 1 b tumour subclassification n SM 1 – Penetrates the shallowest one-third of the submucosa (<500 microns) n SM 2 – Penetrates into the intermediate one-third of the submucosa n SM 3 – Penetrates the deepest one-third of the submucosa Shimada H et al. Prediction of lymph node status in patients with superficial esophageal carcinoma: analysis of 160 surgically resected cancers. Am J Surg 2006

Prevalence of LN metastasis for early adenocarcinoma n Mucosal CA (T 1 a): 0% n Submucosal CA (T 1 b) n n n SM 1 6% SM 2 23% SM 3 58%

Risk stratification for early esophageal adenocarcinoma Lymphovascular invasion and poor tumour differentiation are significant poor prognostic factors Risk stratification for early esophageal adenocarcinoma: analysis of lymphatic spread and prognostic factors. Ann Surg Oncol. 2010

Management of BE associated adenocarcinoma n T 1 a with favorable histology EMR, followed by mucosal eradication therapy n T 1 b (SM 1, SM 2, and SM 3 tumors) esophagectomy with LN clearance is generally preferred Lesions with superficial submucosal invasion (T 1 b SM 1) n n conflicting data regarding the likelihood of lymph node invasion for high risk patient, endoscopic therapy can be considered as an alternative especially for CA with favorable histology

Endoscopic therapy for early adenocarcinoma--Limitation of EMR q Contraindications for EMR n n q Submucosal invasion detected by EUS Failure of submucosal lifting Lesion larger than 2 cm require piecemeal resection n Increased risk of recurrence and perforation

Endoscopic submucosal dissection (ESD) q q q Allow enbloc resection of large lesions Higher R 0 resection rate No difference in complete remission from neoplasia at 3 months Terheggen G et al. A randomised trial of endoscopic submucosal dissection versus endoscopic mucosal resection for early Barrett's neoplasia. Gut 2016 q Lower risk of local recurrence Guo HM et al. Endoscopic submucosal dissection vs endoscopic mucosal resection for superficial esophageal cancer. World J Gastroenterol. 2014

Cons of ESD n n Higher risk of perforation and stricture Longer procedure time

European Society of Gastrointestinal Endoscopy guideline 2015 EMR should be preferred for resection of mucosal cancer q ESD may be considered in selected cases q n n n lesions larger than 1. 5 cm poorly lifting tumors lesions at risk for submucosal invasion

Can the patient treated endoscopically without surgery? n T 1 b. N 0 tumour Esophagectomy with LN clearance is preferred n For consideration of endoscopic therapy n q n ESD depth of SM invasion and histological feature For T 1 b SM 1 cancer, reported outcome of endoscopic treatment in highly selected patient are encouraging Manner H et al. Efficacy, safety, and long-term results of endoscopic treatment for early stage adenocarcinoma of the esophagus with low risk sm 1 invasion. Clin Gastroenterol Hepatol 2013

Summary n n n RFA is currently the preferred endoscopic eradication therapy for dysplastic BE EMR serves as the initial diagnostic and therapeutic maneuver of nodular BE Management of BE associated adenocarcinoma depends on the depth of invasion and histological features


Diagnostic criteria Extension of salmon coloured mucosa into tubular esophagus >=1 cm proximal to the OGJ with biopsy confirmed intestinal metaplasia n. Asia pacific consensus and US histology confirmation required

Surgery for dysplastic BE n Anti-reflux surgery q Limited role in the prevention of adenocarcinoma n n q n one relatively small randomized trial showed no difference in progression outcomes meta-analyses revealed conflicting results Indication control of reflux symptom despite optimised medical therapy Esophagectomy q HGD with no durable eradication

Endoscopic surveillance after endoscopic eradication therapy n 4 quadrant biopsy every cm thought the extent of previous BE, GEJ and cardia to check for complete eradicaton of intestinal metaplasia and dysplasia (CEIM) n surveillance interval after CEIM q q HGD/ intramucosal carcinoma: Q 3 month in first year Q 6 month in second yearly LGD: Q 6 month in first yearly

Photodynamic therapy n n n light-sensitizing drug injected stimulated by exposing the BE portion of the esophagus to light of a specific wavelength cytotoxicity resulted
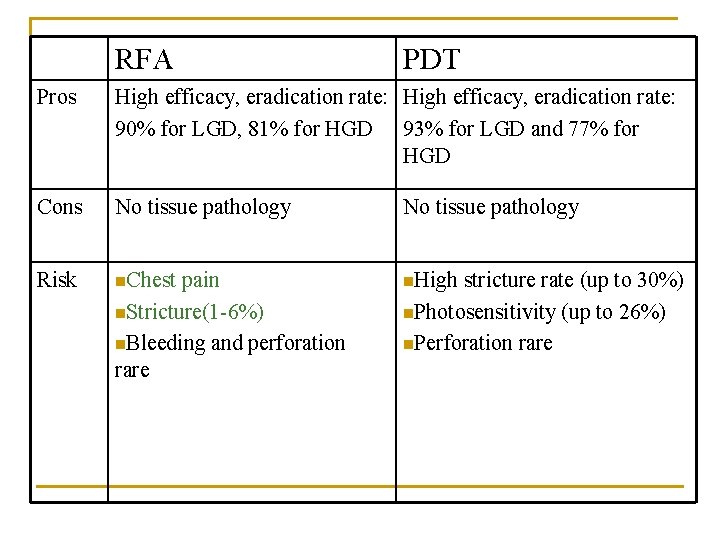
RFA PDT Pros High efficacy, eradication rate: 90% for LGD, 81% for HGD 93% for LGD and 77% for HGD Cons No tissue pathology Risk n. Chest n. High pain n. Stricture(1 -6%) n. Bleeding and perforation rare stricture rate (up to 30%) n. Photosensitivity (up to 26%) n. Perforation rare

Importance of expert pathologists n Interpreting EMR specimens of Barrett's neoplasia may be more difficult q q q tissue architecture differs from the layered architecture in squamous mucosa n more difficult to discern a clear transition between wall layers severe inflammation due to the reflux disease EMR involved use of electrocoagulation, the resection margins may have coagulation artifacts

Vagal sparing esophagectomy n significantly reduced incidence q q q overall infectious complications postoperative dumping and diarrhea less weight loss
- Slides: 39