Balzs Molnr Key Account Manager Salmovac 440 Salmonella

Balázs Molnár – Key Account Manager Salmovac 440 Salmonella Control in Poultry

Schedule of presentation 1. 2. 3. Future challanges of Salmonella on poultry flocks The Salmonella vaccine Differentiation of the active vaccine strain from wild strains

Salmonella in EU Future challanges ● ● Where we see the development of Salmonella prevalances in poultry flocks and countries The trend in regulations

Salmonella awareness Starting point for Salmonella awareness in EU: 1988: UK Health minister Edwina Currie has provoked outrage by saying most of Britain's egg production is infected with Salmonella BBC: December 3 rd, 1988 1990: National Egg Awareness Campaign (UK) Rising awareness in media across Europe
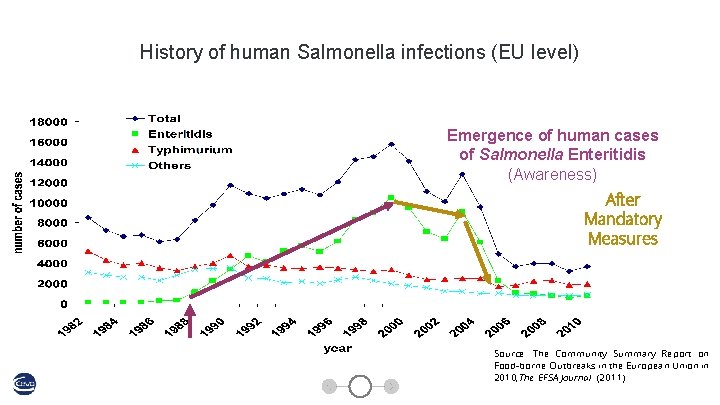
History of human Salmonella infections (EU level) Emergence of human cases of Salmonella Enteritidis (Awareness) After Mandatory Measures Source: The Community Summary Report on Food-borne Outbreaks in the European Union in 2010, The EFSA Journal (2011)

EFSA opinion on vaccination ● EU summary report on zoonoses, zoonotic agents and food-borne outbreaks 2010 EFSA Journal 2012; 10(3): 2597 ● “The results above indicate that the reduction of S. Enteritidis in laying hen flocks and of Salmonella spp. in table eggs is likely to have contributed to the decline of S. Enteritidis cases in humans, since eggs are regarded to be the most important source of these infections. Increased voluntary and compulsory vaccination of laying hens, as well as other hygiene-based control measures, are likely to have contributed to this, driven by the economic consequences of egg restrictions and the requirement to heat treat eggs from positive flocks”.

Salmonella incidences in EU MSs, 2017 • In 2017 the number of human salmonellosis cases was reported as 91. 662 • This means 19. 7 cases /100. 000 population in 2017 • This was the highest since 2013 (due to report on case based data for the first time • Outbreak related data were higher than in 2015 but lower than in 2016 7
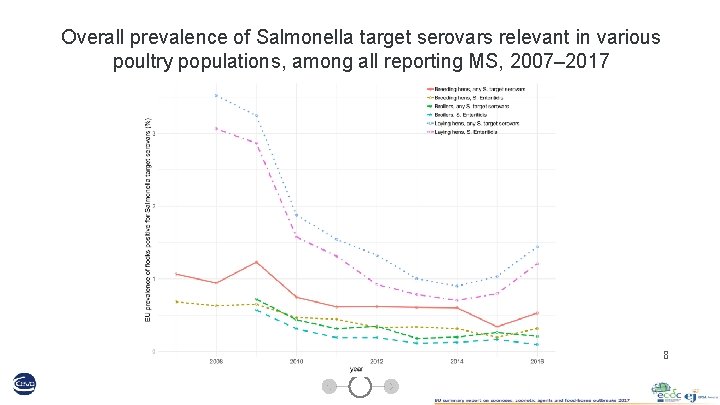
Overall prevalence of Salmonella target serovars relevant in various poultry populations, among all reporting MS, 2007– 2017 8
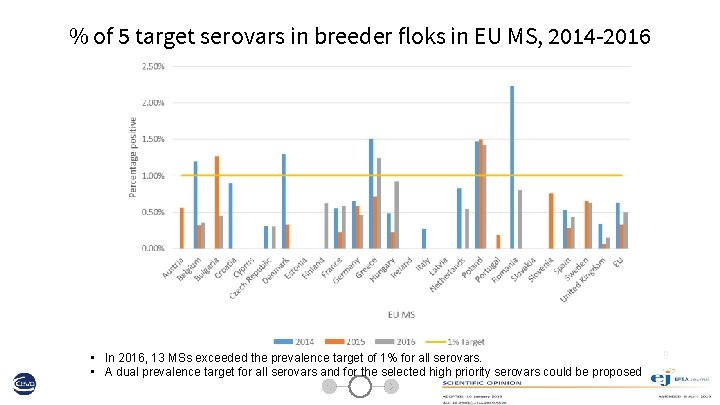
% of 5 target serovars in breeder floks in EU MS, 2014 -2016 • In 2016, 13 MSs exceeded the prevalence target of 1% for all serovars. • A dual prevalence target for all serovars and for the selected high priority serovars could be proposed 9
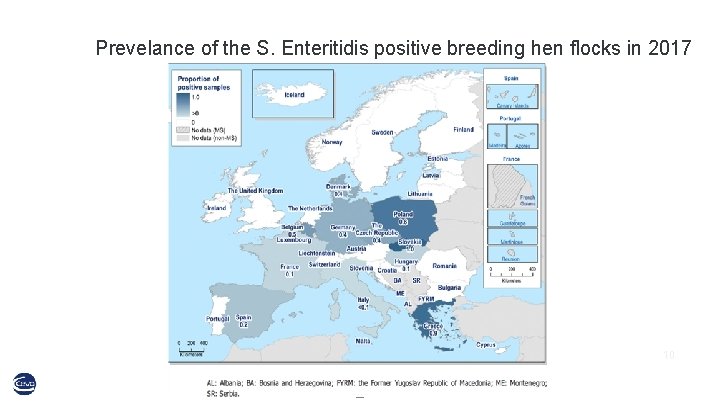
Prevelance of the S. Enteritidis positive breeding hen flocks in 2017 10
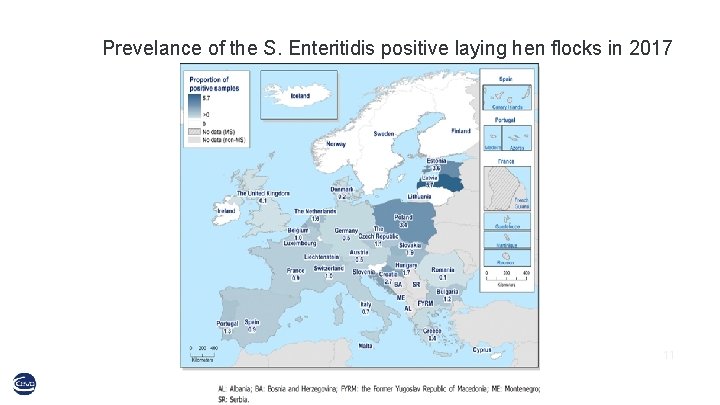
Prevelance of the S. Enteritidis positive laying hen flocks in 2017 11
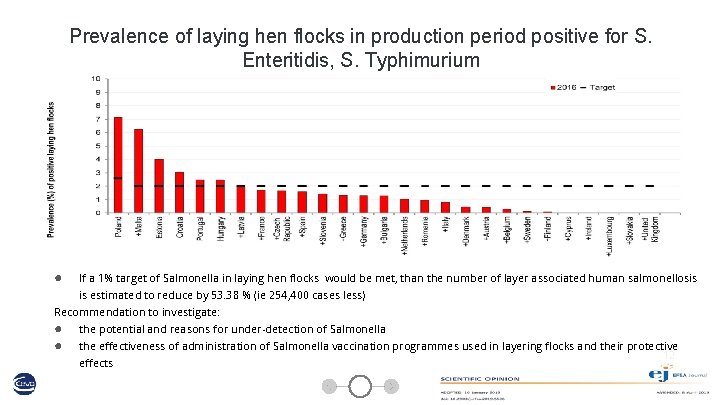
Prevalence of laying hen flocks in production period positive for S. Enteritidis, S. Typhimurium ● If a 1% target of Salmonella in laying hen flocks would be met, than the number of layer associated human salmonellosis is estimated to reduce by 53. 38 % (ie 254, 400 cases less) Recommendation to investigate: ● ● the potential and reasons for under-detection of Salmonella the effectiveness of administration of Salmonella vaccination programmes used in layering flocks and their protective 12 effects
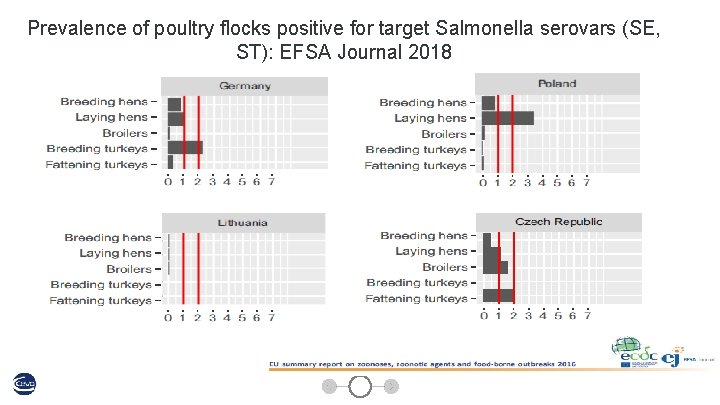
Prevalence of poultry flocks positive for target Salmonella serovars (SE, ST): EFSA Journal 2018 13
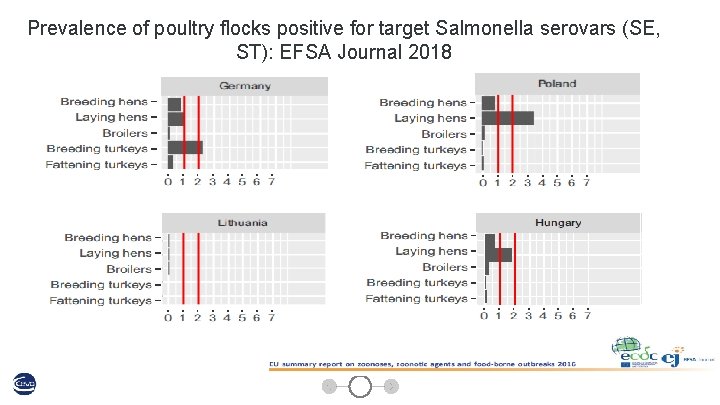
Prevalence of poultry flocks positive for target Salmonella serovars (SE, ST): EFSA Journal 2018 14
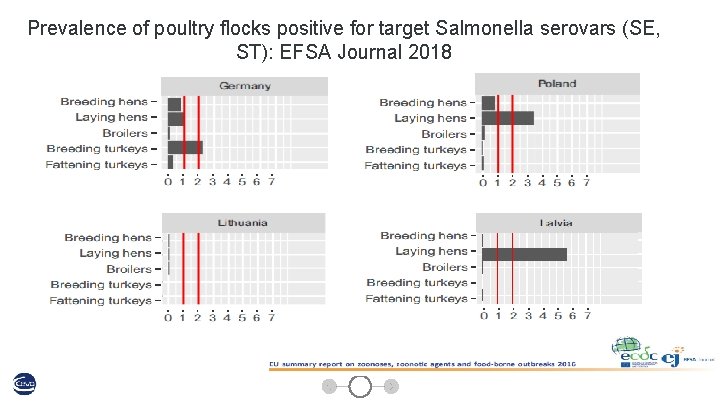
Prevalence of poultry flocks positive for target Salmonella serovars (SE, ST): EFSA Journal 2018 15
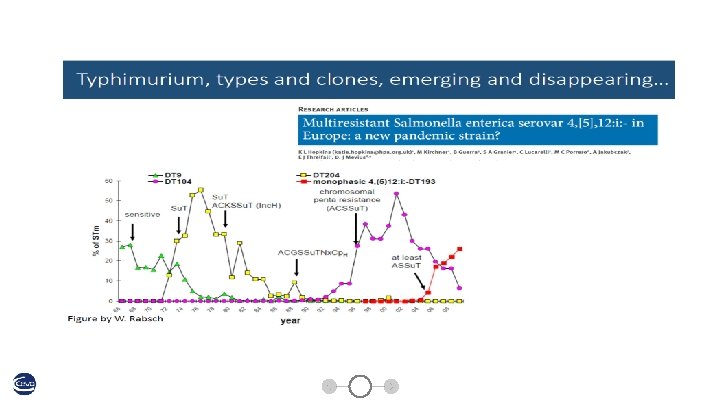
SE phagetypes across Europe Image source:

New challenges: Ø different Serotypes, different Phagetypes
New challenges: • Longer laying period: Ø 91 weeks • Progress in layer breeds • Low egg prices make longer production cycles more attractive ○ Longer Salmonella protection is needed! • Alternative housing systems 19
What should a „better“ Salmonella vaccine offer? Ø Broad protection • Ø to cover emerging Salmonella (like monophasic strain of S. Typhimurium) Early and long protection • to cover the full laying period

The Salmonella vaccine Preparation for the challanges Early, Long and broad protection against Salmonella in poultry

Salmovac 440 Early, long and broad protection against Salmonella in poultry IDT Biologika
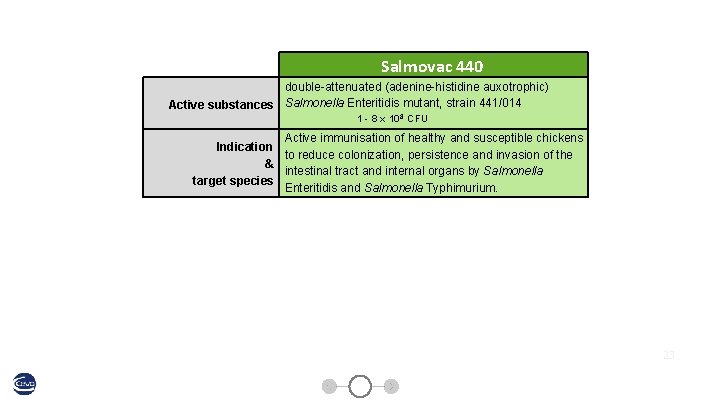
Salmovac 440 double-attenuated (adenine-histidine auxotrophic) Active substances Salmonella Enteritidis mutant, strain 441/014 1 - 8 x 108 CFU Active immunisation of healthy and susceptible chickens Indication to reduce colonization, persistence and invasion of the & intestinal tract and internal organs by Salmonella target species Enteritidis and Salmonella Typhimurium. 23

Salmovac 440: Broad protection Salmovac 440: proven protection against: - SE Phagetypes: PT 1, PT 4, PT 8, PT 21 - ST and monophasic strain of ST Sven Springer, Thomas Lindner, Hans-Joachim Selbitz: Testing of the immunogenicity of a Salmonella Enteritidis live vaccine against non. PT 4 Salmonella Enteritidis strains and a multi-drug resistant monophasic Salmonella Typhimurium strain International Symposium on Salmonella and Salmonellosis, St. Malo, France, 2013 24
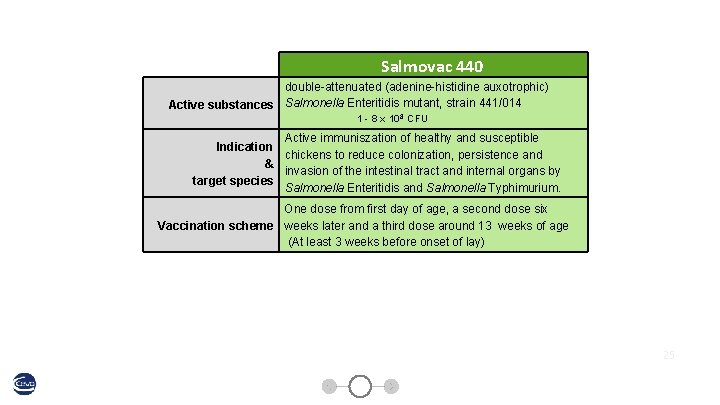
Salmovac 440 double-attenuated (adenine-histidine auxotrophic) Active substances Salmonella Enteritidis mutant, strain 441/014 1 - 8 x 108 CFU Active immuniszation of healthy and susceptible Indication chickens to reduce colonization, persistence and & invasion of the intestinal tract and internal organs by target species Salmonella Enteritidis and Salmonella Typhimurium. One dose from first day of age, a second dose six Vaccination scheme weeks later and a third dose around 13 weeks of age (At least 3 weeks before onset of lay) 25

Salmovac 440 26
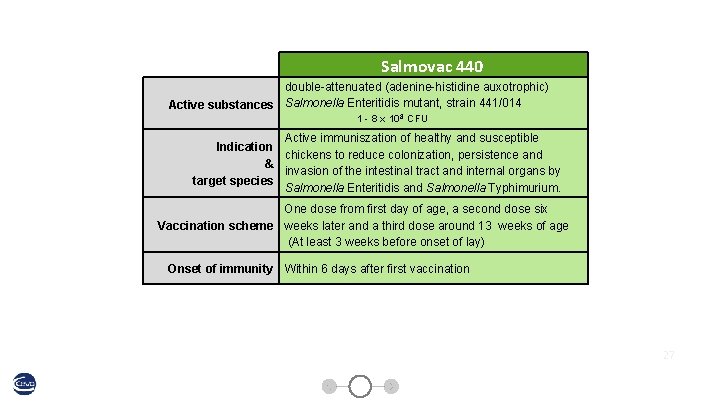
Salmovac 440 double-attenuated (adenine-histidine auxotrophic) Active substances Salmonella Enteritidis mutant, strain 441/014 1 - 8 x 108 CFU Active immuniszation of healthy and susceptible Indication chickens to reduce colonization, persistence and & invasion of the intestinal tract and internal organs by target species Salmonella Enteritidis and Salmonella Typhimurium. One dose from first day of age, a second dose six Vaccination scheme weeks later and a third dose around 13 weeks of age (At least 3 weeks before onset of lay) Onset of immunity Within 6 days after first vaccination 27
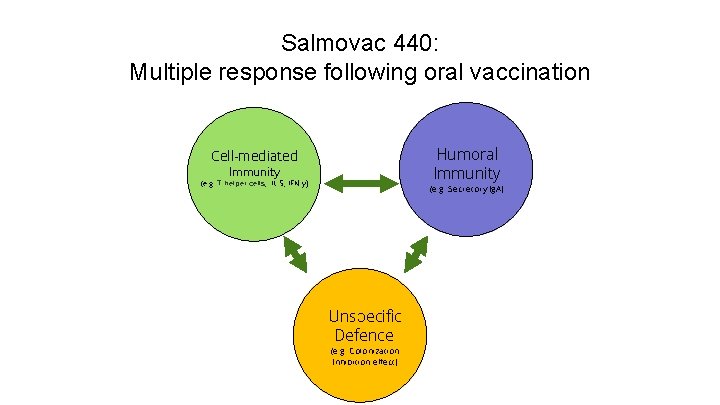
Salmovac 440: Multiple response following oral vaccination 21, 9% Humoral Immunity Cell-mediated Immunity 42% (e. g. T-helper cells, IL-5, IFN-y) 4, 7% 5, 9% (e. g. Secretory Ig. A) 13, 7% Unspecific Defence (e. g. Colonization Inhibition effect)

Salmovac 440: Multiple response following oral vaccination Unspecific Defence Early colonization - Inhibitory effect in the gut preventing 42% colonization by other Salmonellae (e. g. Colonization 21, 9% Inhibition effect) 4, 7% 5, 9% - - No effect if already infected with Salmonella First oral vaccination on 1 st day of live! Onset of immunity (OOI): 6 d 13, 7% (Avi. Pro: 14 d) 29
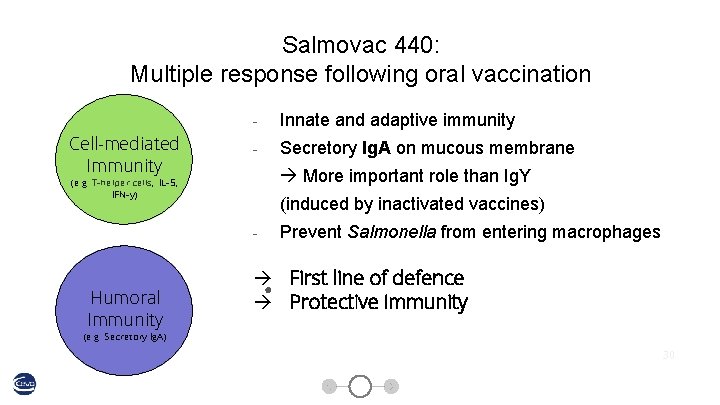
Salmovac 440: Multiple response following oral vaccination Cell-mediated Unspecific Immunity Defence - Innate and adaptive immunity - Secretory Ig. A on mucous membrane More important role than Ig. Y (e. g. T-helper cells, IL-5, Colonization IFN-y)effect) Inhibition 21, 9% 4, 7% 5, 9% Humoral Immunity (induced by inactivated vaccines) - Prevent Salmonella from entering macrophages ● First line of defence Protective immunity (e. g. Secretory Ig. A) 30
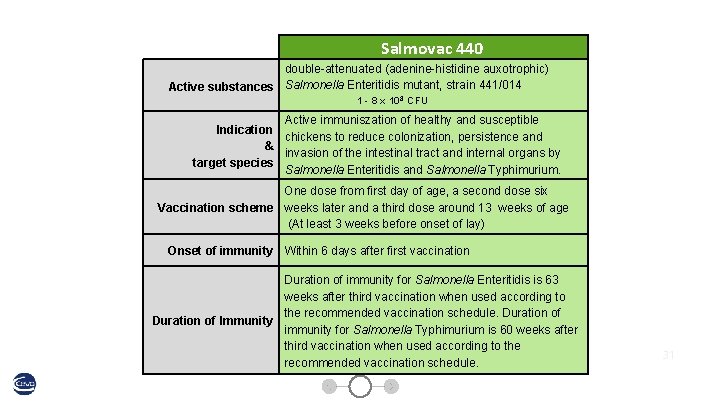
Salmovac 440 double-attenuated (adenine-histidine auxotrophic) Active substances Salmonella Enteritidis mutant, strain 441/014 1 - 8 x 108 CFU Active immuniszation of healthy and susceptible Indication chickens to reduce colonization, persistence and & invasion of the intestinal tract and internal organs by target species Salmonella Enteritidis and Salmonella Typhimurium. One dose from first day of age, a second dose six Vaccination scheme weeks later and a third dose around 13 weeks of age (At least 3 weeks before onset of lay) Onset of immunity Within 6 days after first vaccination Duration of immunity for Salmonella Enteritidis is 63 weeks after third vaccination when used according to the recommended vaccination schedule. Duration of Immunity immunity for Salmonella Typhimurium is 60 weeks after third vaccination when used according to the recommended vaccination schedule. 31

Salmovac 440: Efficacy testing – SE and STm challenge, DOI Up to 11 weeks longer protection against Salmonella Enteritidis and 14 weeks longer protection against Salmonella Typhimurium. Salmovac 440 32
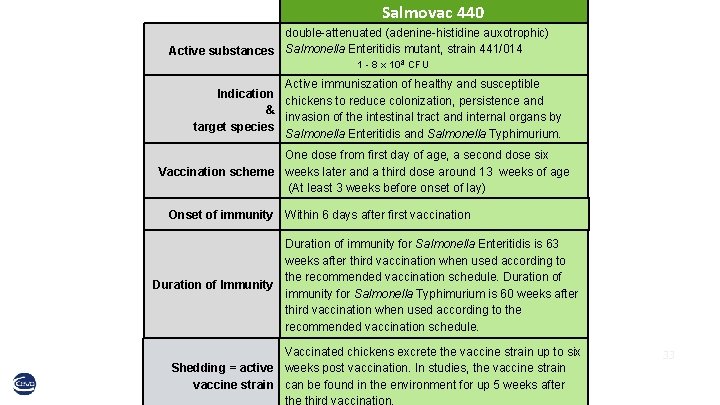
Salmovac 440 double-attenuated (adenine-histidine auxotrophic) Active substances Salmonella Enteritidis mutant, strain 441/014 1 - 8 x 108 CFU Active immuniszation of healthy and susceptible Indication chickens to reduce colonization, persistence and & invasion of the intestinal tract and internal organs by target species Salmonella Enteritidis and Salmonella Typhimurium. One dose from first day of age, a second dose six Vaccination scheme weeks later and a third dose around 13 weeks of age (At least 3 weeks before onset of lay) Onset of immunity Within 6 days after first vaccination Duration of immunity for Salmonella Enteritidis is 63 weeks after third vaccination when used according to the recommended vaccination schedule. Duration of Immunity immunity for Salmonella Typhimurium is 60 weeks after third vaccination when used according to the recommended vaccination schedule. Vaccinated chickens excrete the vaccine strain up to six Shedding = active weeks post vaccination. In studies, the vaccine strain can be found in the environment for up 5 weeks after 33
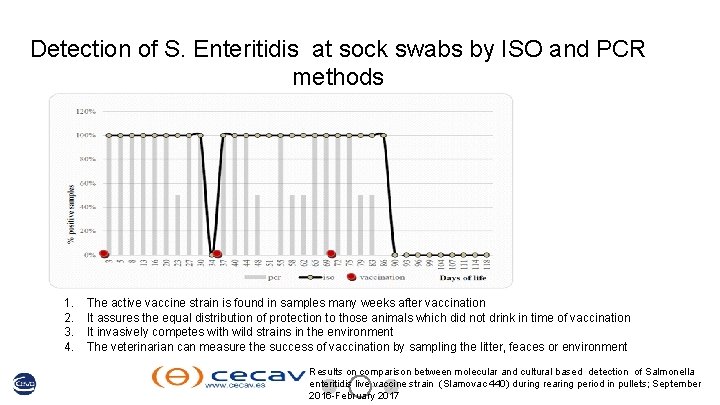
Detection of S. Enteritidis at sock swabs by ISO and PCR methods 1. 2. 3. 4. The active vaccine strain is found in samples many weeks after vaccination It assures the equal distribution of protection to those animals which did not drink in time of vaccination It invasively competes with wild strains in the environment The veterinarian can measure the success of vaccination by sampling the litter, feaces or environment Results on comparison between molecular and cultural based detection of Salmonella enteritidis live vaccine strain (Slamovac 440) during rearing period in pullets; September 2016 -February 2017
Laboratory Differentiation methods Differentiation of Salmovac 440 vaccine strain from wild strains

Salmovac 440 - Diagnostics • Bacteriological verification for Salmonella has to follow the ISO 6579: 2002 • Our Vaccine strain (Salmovac 440) grows in the used selective media „false positive“ 36

Salmovac 440 - Diagnostics Live Salmonella vaccines for which the manufacturer does not provide an appropriate method to distinguish bacteriologically wild-type strains of salmonella from vaccine strains shall not be used in the framework of national control programmes adopted pursuant to Article 6 of Regulation (EC) No 2160/2003.
Salmovac 440 - Diagnostics Field strain or Vaccine strain? Differentiation based on Adenin and Histidin Auxotrophy

IDT Salmonella Diagnostic kit The IDT Salmonella Diagnostic Kit RTU two ready-to-use media – Medium A and Medium B – for differentiation of Salmonella Dublin, Salmonella Typhimurium and Salmonella Enteritidis IDT vaccine strains from wild type strains of the same serotype. Medium A : Various salts, Glucose, and Nicotinamide Medium B : Various salts, Glucose, Nicotinamide and nutrients dependent on vaccine strain. • • • Wild type strains of S. Dublin, S. Typhimurium and S. Enteritidis from Salmonella enterica subsp. Enterica show a definitive growth (turbidity) in both, Medium A and B of the IDT Salmonella Diagnostic Kit RTU. The IDT vaccine strains, however, only grow in Medium B but not in Medium A, due to their special requirements for nutrients as auxotrophic strains. In very rare cases, the appearance of auxotrophic wild strains has been observed. These wild strains will not grow - neither in Medium A nor in Medium B of this kit.
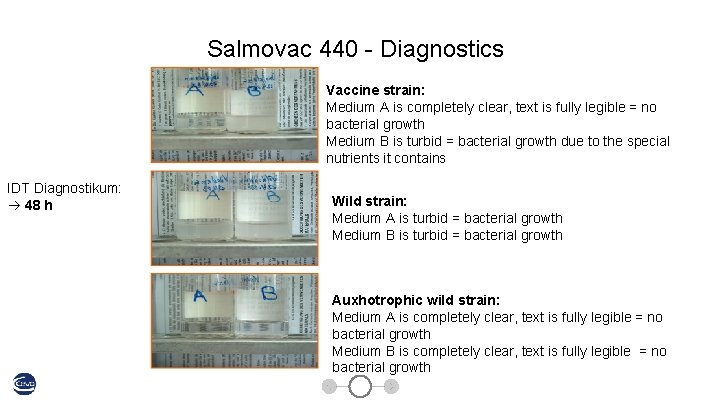
Salmovac 440 - Diagnostics Vaccine strain: Medium A is completely clear, text is fully legible = no bacterial growth Medium B is turbid = bacterial growth due to the special nutrients it contains IDT Diagnostikum: 48 h Wild strain: Medium A is turbid = bacterial growth Medium B is turbid = bacterial growth Auxhotrophic wild strain: Medium A is completely clear, text is fully legible = no bacterial growth Medium B is completely clear, text is fully legible = no bacterial growth
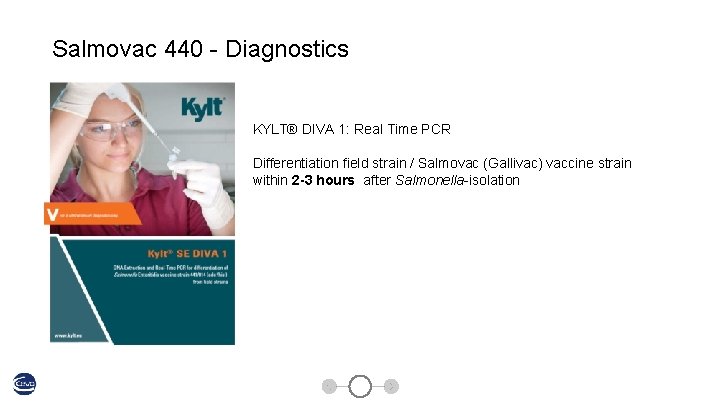
Salmovac 440 - Diagnostics KYLT® DIVA 1: Real Time PCR Differentiation field strain / Salmovac (Gallivac) vaccine strain within 2 -3 hours after Salmonella-isolation Salmovac 440 41
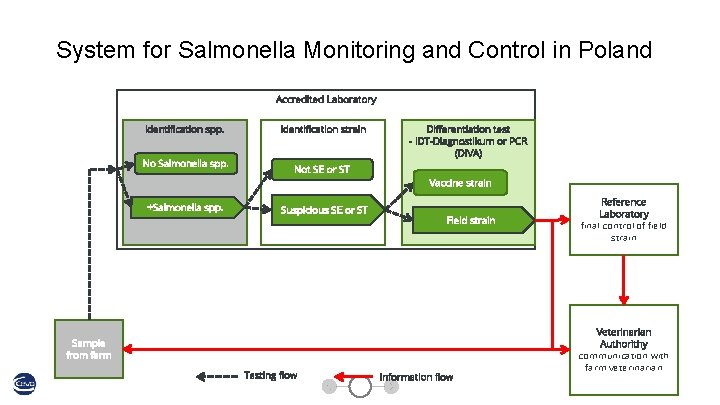
System for Salmonella Monitoring and Control in Poland Accredited Laboratory Identification spp. No Salmonella spp. +Salmonella spp. Identification strain Not SE or ST Suspicious SE or ST Differentiation test - IDT-Diagnostikum or PCR (DIVA) Vaccine strain Field strain Sample from farm Testing flow Information flow Reference Laboratory final control of field strain Veterinarian Authorithy communication with farm veterinarian
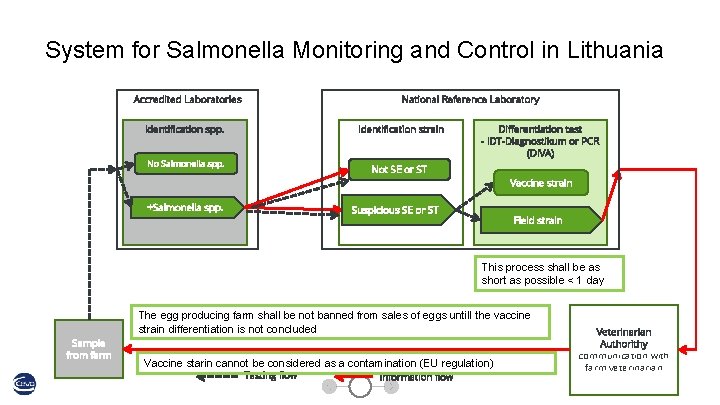
System for Salmonella Monitoring and Control in Lithuania National Reference Laboratory Accredited Laboratories Identification spp. Identification strain No Salmonella spp. Differentiation test - IDT-Diagnostikum or PCR (DIVA) Not SE or ST +Salmonella spp. Vaccine strain Suspicious SE or ST Field strain This process shall be as short as possible < 1 day The egg producing farm shall be not banned from sales of eggs untill the vaccine strain differentiation is not concluded Sample from farm Vaccine starin cannot be considered as a contamination (EU regulation) Testing flow Information flow Veterinarian Authorithy communication with farm veterinarian
Take home messages - It is not possible to eradicate Salmonella because the wide range of contamination sources: Natural reservoirs (e, g, poultry, pigs, cattle, pets ), Vectors (e. g. rodents, flies, darkling beetles) and other sources (e. g. feed, raw materials, fomites) - Salmonella is still and will be a problem in the future - Vaccination in general is the major intervention tool to prevent Salmonella infections significant decrease of Salmonella prevalence in EU-27

Salmovac 440 Early, long and broad protection against Salmonella in poultry OOI: 6 days after 1 st vaccination Avi. Pro Duo: 14 d IDT Biologika

Salmovac 440 Early, long and broad protection against Salmonella in poultry S. E. : 63 weeks S. T. : 60 weeks Avi. Pro Duo: 52 weeks Avi. Pro Duo: 46 weeks IDT Biologika

Salmovac 440 Early, long and broad protection against Salmonella in poultry different phage types of S. E. S. T. , also monophasic S. T. IDT Biologika

Salmovac 440 The active vaccine straincan be found by sampling It can be differentiated by ISO and PCRmethods in laboratories IDT Biologika


balazs. molnar@ceva. com

Thank you very much for your attention!

● FAQ: Are Antibodies important for day old chicks? 52

FAQ: Are Antibodies important for day old chicks? ● What do we know about Ig. Y: • Induced by inactivated vaccines • Easy to measure in the blood • Transmitted to their offspring via the egg • But: High level of Ig. Y does not correlate with good protection against salmonella Much more important: active protection with a live vaccine that has early onset of protection unspecific defense, cell-mediated immunity, Ig. A antibodies 53

FAQ: Do maternal antibodies interfere with an early live vaccination? Springer et al. (2006) Vaccine strain in the liver on day 14 after hatching in log c. f. u. / g Group Liver 1 (parents vaccinated with SALENVAC®) 1. 99 ± 0. 50 2 (parents unvaccinated) 1. 74 ± 0. 35 No significant differences Maternally-induced antibodies had no influence on the colonisation of the liver by the vaccine strain Protective effect against virulent field strain? 54

Salmovac 440 in (broiler) breeders Reduced colonization, persistence • and invasion of internal organs ● Reduced shedding Clean parent flock • No vertical transmission of Salmonella via the egg • Best protection against a pathogen is the absence of the pathogen 55
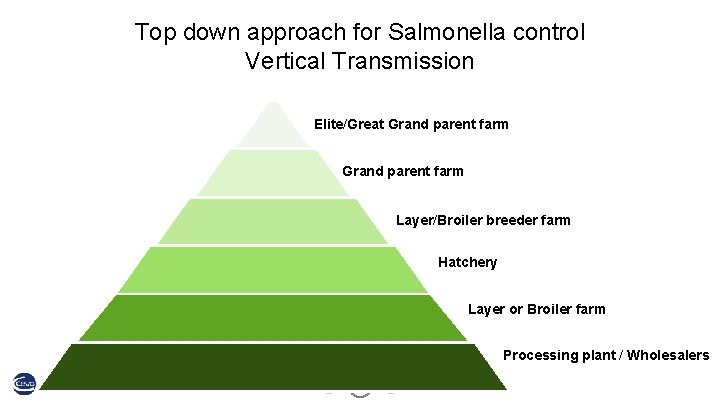
Top down approach for Salmonella control Vertical Transmission Elite/Great Grand parent farm Layer/Broiler breeder farm Hatchery Layer or Broiler farm Processing plant / Wholesalers

FAQ: Are there advantages in using 3 x Salmovac 440 instead of 2 x live vaccine + 1 x inactivated? ● 57

Comparative study: 3 x live vaccine – 2 x live, 1 x inactivated 58
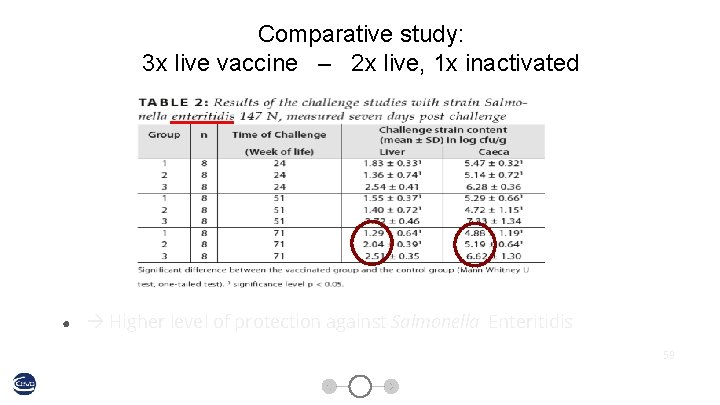
Comparative study: 3 x live vaccine – 2 x live, 1 x inactivated ● Higher level of protection against Salmonella Enteritidis 59
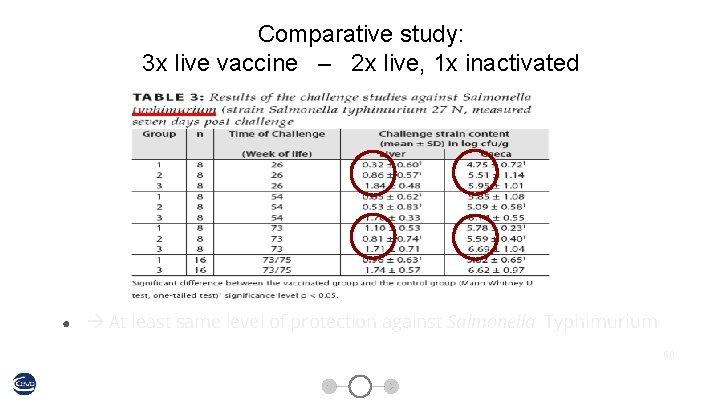
Comparative study: 3 x live vaccine – 2 x live, 1 x inactivated ● At least same level of protection against Salmonella Typhimurium 60

FAQ: Are there advantages in using 3 x Salmovac 440 instead of 2 x live vaccine + 1 x inactivated? • Cross protection against • • different phage types of Salmonella Enteritidis (PT 1, 4, 8, 21) Salmonella Typhimurium, also the monophasic strain Salmonella Infantis Salmonella Gallinarum: Reduced Mortality (week 20, week 32) • Long protection: S. E. 63 weeks, S. T. 60 weeks • Less stress: birds don’t have to be caught • Lower costs: employees, inactivated vaccine 61

Salmovac 440: Cross protection S. Gallinarum • S. Enteritidis and S. Gallinarum belong to same Salmonella group D • Better protection if 3 x Salmovac 440 • No official claim in the SPC 62

● FAQ: Can the vaccine be sprayed on day old chicks? 63

FAQ: Can the vaccine be sprayed on day old chicks? ● Yes! • Coarse spray application • Chicks are covered with vaccine, preen their feathers “drinking” the vaccine But: Disclaimer Off-label-use (no registration) Collected data is not yet published in a peer reviewed journal No official promotion 64

Field Spra-Vac ● 10 l tank ● 40. 000 chicks / hour ● Intermittent or continuous spray ● Rechargeable battery: 4 hours (continuous) 65

Field Spra-Vac ● 4 different nozzles ● Coarse spray: droplet size > 100 µm ● Do not use fine spray or aerosol ● Pressure: 1, 5 bar (20 psi) ● Distance: about 50 cm 66

Field Spra-Vac ● Dilution: 300 ml 1000 ID (+ 5% surplus) 1, 5 l ● 5000 ID (+ 5% surplus) 1 spray dose = 15 ml 2 spray doses = 30 ml 100 chicks or 1 box 67
Field Spra-Vac 1. Box with 100 day old chicks 2. 68
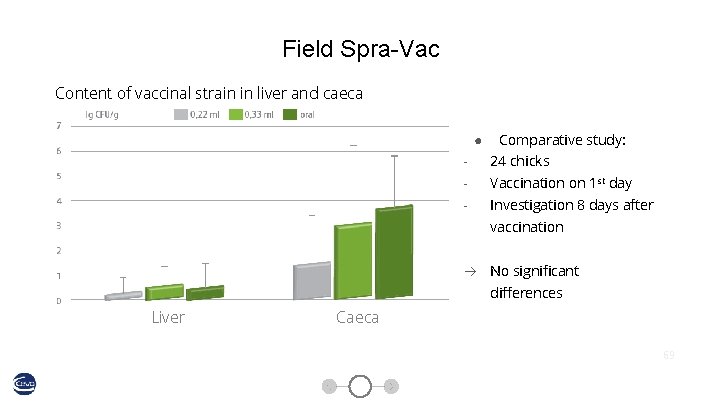
Field Spra-Vac Content of vaccinal strain in liver and caeca ● Comparative study: - 24 chicks - Vaccination on 1 st day - Investigation 8 days after vaccination No significant differences Liver Caeca 69

Field Spra-Vac Easy and efficientadministration of live vaccines to day old chicks (small hatcheries or on the farm) Early vaccination(1 st day) possible, although day old chicks do not drink much Early onset of protection Disclaimer remark 70
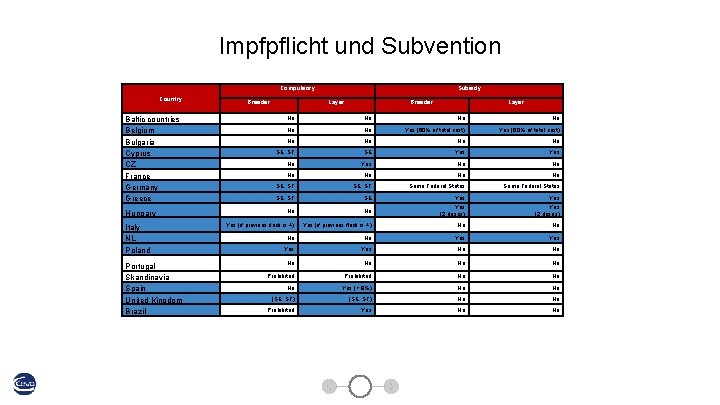
Impfpflicht und Subvention Compulsory Country Breeder Baltic countries Belgium Bulgaria Cyprus CZ France 4, 1% Germany Greece 12, 3% Italy NL Poland Portugal Skandinavia Spain United Kingdom Brazil 14, 4% Breeder Layer No No No Yes (60% of total cost) No No SE Yes No No No SE; ST Some Federal States SE; ST SE Yes No No Yes (2 doses) Yes (if previous flock is +) No No Yes Yes No No 43, 7% 5, 3% Layer SE; ST Hungary 10, 6% Subsidy No No Prohibited No No No Yes (> 9%) No No (SE; ST) No No Prohibited Yes No No

Thank you very much for your attention!
- Slides: 73