Balancing Redox Reactions Mn O 4 Mn 2

Balancing Redox Reactions Mn. O 4 - Mn 2+ manganese is balanced balance oxygen using H 2 O Mn. O 4 - Mn 2+ + 4 H 2 O balance hydrogen using H+ Mn. O 4 - + 8 H+ Mn 2+ + 4 H 2 O balance charge using e- Mn. O 4 - + 8 H+ +5 e- Mn 2+ + 4 H 2 O

Oxidation States Mn. O 4 - + 8 H+ +5 e- Mn 2+ + 4 H 2 O Oxidation state of oxygen = 2 - in compounds 0 in O 2 Oxidation state of hydrogen = 1+ in compounds 0 in H 2 Mn. O 4 Mn 2+ 4 x (-2)+ 1(Mn) = -1 Mn = 7+ Mn = 2+ Mn 7+ + 5 e- Mn 2+

Mn. O 4 - + 8 H++5 e- Mn 2+ + 4 H 2 O o = 1. 51 V 2 H+ + 2 e- H 2 o = 0. 00 V anode cathode Pt(s) H 2(g)(1 atm) H+(aq)(1 M) Mn. O 4 -(aq)(1 M), Mn 2+(aq)(1 M) , H+(1 M) Pt(s) ( H 2 2 H++2 e- )5 ( Mn. O 4 -+8 H++5 e- Mn 2++4 H 2 O )2 oxidation reaction 6 reduction reaction 2 Mn. O 4 - + 16 H+ +10 e- + 5 H 2 2 Mn 2+ + 8 H 2 O + 10 H+ + 10 e- 2 Mn. O 4 - + 6 H+ + 5 H 2 2 Mn 2+ + 8 H 2 O

2 H+ + 2 e- H 2 o = 0. 00 V Mn. O 4 - + 8 H++5 e- Mn 2+ + 4 H 2 O o = 1. 51 V 2 Mn. O 4 - + 6 H+ + 5 H 2 2 Mn 2+ + 8 H 2 O ocell = ored - oox = 1. 51 - 0 = 1. 51 V Go = -n. F o = - (10)(96, 500)(1. 51) = -1457 k. J Go = -RT ln K= -1457 k. J -RT K = e 588

Nernst Equation G = Go + RT ln Q G = -n. F cell Go = -n. F ocell standard non-standard cell = o cell - RT ln Q n. F

Nernst Equation anode cell = ocell - RT ln Q n. F cathode Cu(s) Cu 2+(aq) (1 M 4 M ) Ag+(aq) (1 M) Ag (s) a) oxidation - 2+Cu Cu 2+ (s) + 2 e. Cu + 2 e o= 0. 34 V b) reduction 2( Ag+ + e- Ag(s) ) o= 0. 80 V ocell = ored - oox = 0. 80 - 0. 34 = 0. 46 V

Nernst Equation anode cell = ocell - RT ln Q n. F cathode Cu(s) Cu 2+(aq) (4 M) Ag+(aq) (1 M) Ag(s) Cu (s) Cu 2+ + 2 e- 2 Ag+ + 2 e- 2 Ag (s) Q = [products]minitial = [Cu 2+] = 4 [reactants]ninitial [Ag+]2 12

Nernst Equation anode cell = ocell - RT ln Q n. F cathode Cu(s) Cu 2+(aq) (4 M) Ag+(aq) (1 M) Ag(s) Cu(s) Cu 2+ + 2 e- 2 Ag+ + 2 e- 2 Ag cell = 0. 46 V - (8. 314) (298) ln 4 = 0. 44 V 2 (96, 500)

Non-standard conditions Cu(s) Cu 2+(aq) (4 M) Ag+(aq) (1 M) Ag(s) Cu(s) Cu 2+ + 2 e- 2 Ag+ + 2 e- 2 Ag G = -n. F Go = -(2 mol e-) (96, 500 C/mol e-) (0. 46 V) = -89 k. J G = -(2 mol e-) (96, 500 C/mol e-) (0. 44 V) = -85 k. J
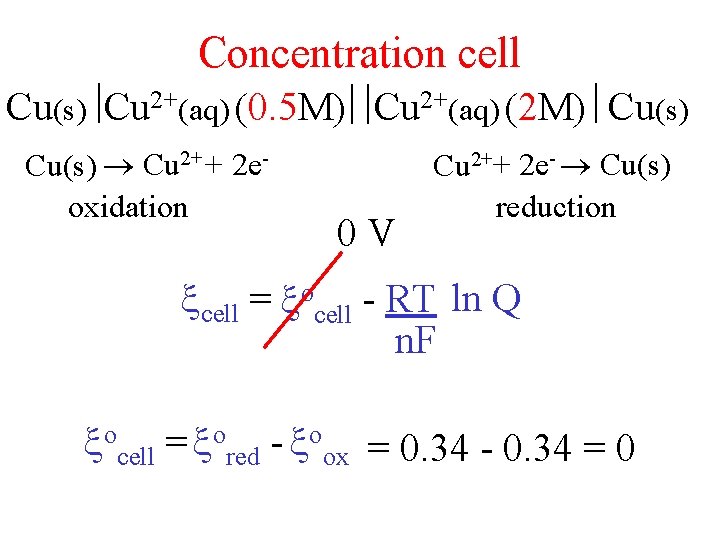
Concentration cell Cu(s) Cu 2+(aq) (0. 5 M) Cu 2+(aq) (2 M) Cu(s) Cu 2+ + 2 eoxidation 0 V Cu 2++ 2 e- Cu(s) reduction cell = ocell - RT ln Q n. F ocell = ored - oox = 0. 34 - 0. 34 = 0

Concentration Cells Cu(s) Cu 2+(aq) (0. 5 M) Cu 2+(aq) (2 M) Cu(s) Cu 2+ + 2 eoxidation Cu 2+ + 2 e- Cu(s) reduction Q = [products]minitial = [Cu 2+] = 0. 5 = 0. 25 anode [reactants]ninitial [Cu 2+]cathode 2 cathode cell = ocell -RT ln Q = 0 -RT ln. 25 =. 02 V 2 F n. F

Concentration Cells h QA- P+ 680 Mn + p. H = 3. 0 2 H 2 O p. H = 7. 0 Q = 10 -7 / 10 -3 ADP + Pi 4 H+ + O 2 + 4 e- cell = ocell - RT ln Q n. F cell = ocell - 0. 059 log Q n cell = 0. 24 V G < 0 ATP G > 0
- Slides: 12