Balancing Redox Equations Many redox equations can be

Balancing Redox Equations: • Many redox equations can be balanced through trial and error! You have been doing this for a least two years without even knowing it! : ) • Do the following: • ___Al + ____HCl ____ Al. Cl 3 + ___ H 2 • 2 6 2 3 • Done!

Balancing Redox Reactions: • Another Example: (spectator ions have not been included) • Sn + Ag+ Sn 2+ + Ag • 1. Assign oxidation numbers. • 0 +1 +2 0 • Sn + Ag+ Sn 2+ + Ag

• 2. Oxidation occurs when the oxidation number increases. Reduction occurs when the oxidation number decreases. Write the two half reactions. • Oxidation: Sn 2+ • Reduction: Ag+ Ag • 3. Use electrons to balance the charges in the half reactions. In oxidation the electrons appear on the right. In reduction the electrons appear on the left.
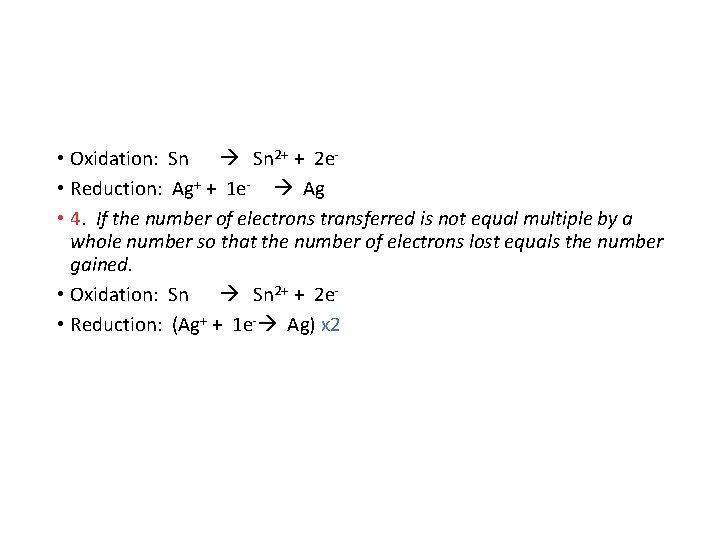
• Oxidation: Sn 2+ + 2 e • Reduction: Ag+ + 1 e- Ag • 4. If the number of electrons transferred is not equal multiple by a whole number so that the number of electrons lost equals the number gained. • Oxidation: Sn 2+ + 2 e • Reduction: (Ag+ + 1 e- Ag) x 2

• 5. Add the half reactions: • Oxidation: Sn 2+ + 2 e • Reduction: 2 Ag+ + 2 e- 2 Ag • Net Balanced Redox Reaction (notice that the electrons cancel!) • Sn + 2 Ag+ Sn 2+ + 2 Ag

Steps for Balancing More: • Step 1: Identify and write the two half-reactions. • Step 2: Balance the elements and charges for each half-reaction: • • Step 3: that the Step 4: product Step 5: charge on 2 a) 2 b) 2 c) 2 d) Balance all the elements except hydrogen and oxygen. Balance the oxygen atoms by adding H 2 O to the appropriate side. Balance the hydrogen by adding H+ to the appropriate side. Balance the charges by adding electrons to the appropriate side. Multiply one or both of the half-reactions by a whole number so number of electrons gained and lost is equal. Combine the half-reactions. Eliminate anything common to the and reactant sides. Check that all of the elements are balanced and that the total each side is the same.

Example: • Balance: Mn. O 4 - + Fe 2+ Mn 2+ + Fe 3+ • Step 1: • Reduction: Mn. O 4 - Mn 2+ +7, -2 +2 • • Oxidation: Step 2: b) Mn. O 4 Mn 2+ + 4 H 2 O c) Mn. O 4 - + 8 H+ Mn 2+ + 4 H 2 O d) Mn. O 4 - + 8 H+ + 5 e- Mn 2+ + 4 H 2 O Fe 2+ Fe 3+ +2 +3

• Oxidation: • Fe 2+ Fe 3+ +1 e- • Step 3: • Mn. O 4 - + 8 H+ + 5 e- Mn 2+ + 4 H 2 O • (Fe 2+ Fe 3+ +1 e-) x 5

• • Step 4: Reduction: Mn. O 4 - + 8 H+ + 5 e- Mn 2+ + 4 H 2 O Oxidation: 5 Fe 2+ 5 Fe 3+ +5 e. Net Reaction: Mn. O 4 - + 8 H+ +5 Fe 2+ Mn 2+ + 4 H 2 O + 5 Fe 3+ • (electrons are cancelled!) • Step 5: • Notice that the final redox equation is balanced by atom and by charge.

One to try • Redox reactions are usually too complex to use trial and error method. • Balance the following example that is in an acidic solution (assume the presence of H 2 O and H+): • HNO 3 + Fe 2+ Fe 3+ + NO 2
- Slides: 10