Balancing Equations Chemical Reaction A chemical reaction is

Balancing Equations
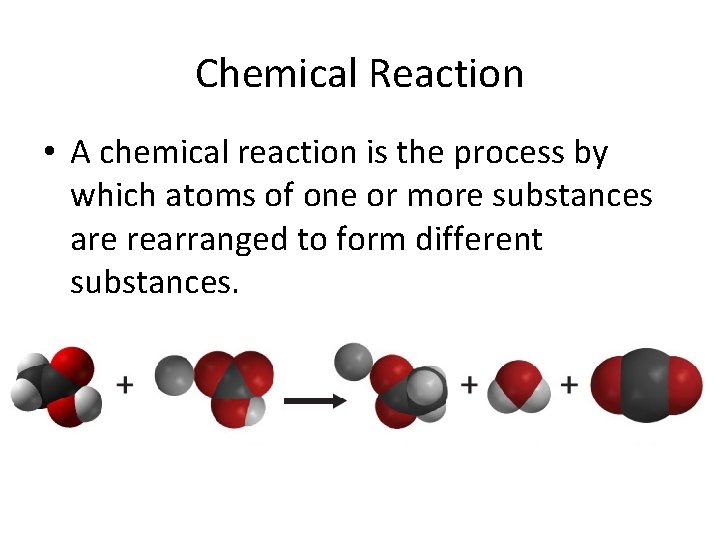
Chemical Reaction • A chemical reaction is the process by which atoms of one or more substances are rearranged to form different substances.

Reactant(s) • Reactant(s) are the starting substance(s) of a reaction • Reactant(s) are on the left
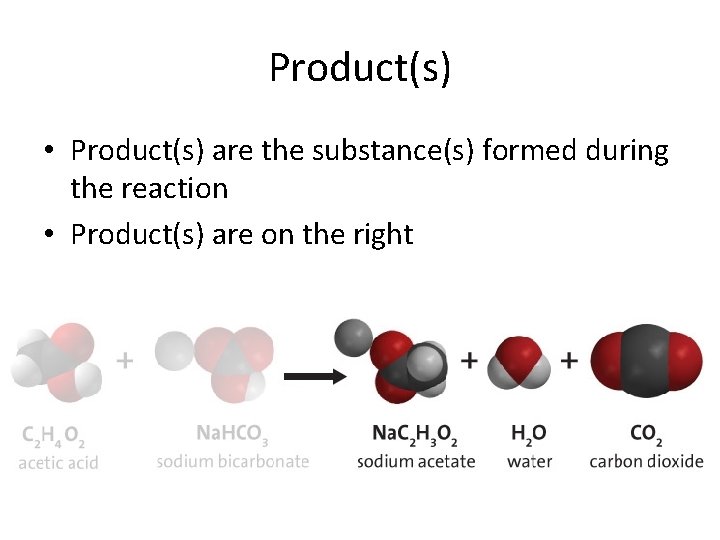
Product(s) • Product(s) are the substance(s) formed during the reaction • Product(s) are on the right
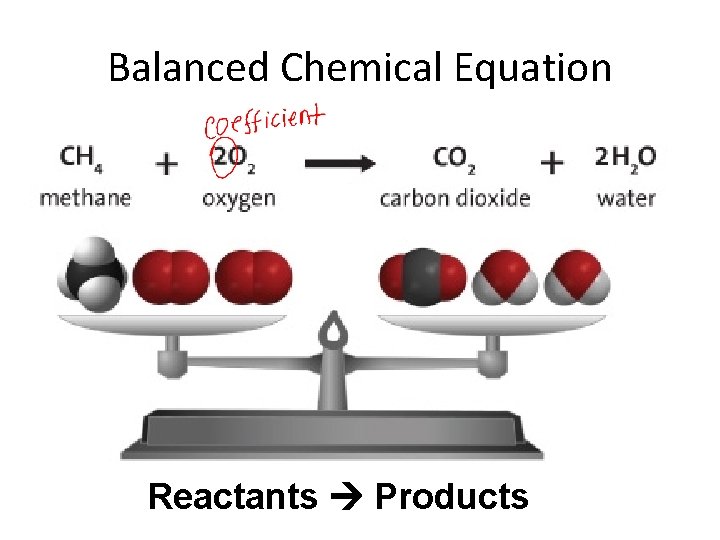
Balanced Chemical Equation • The equation that shows the number of atoms of each reactant and each product is equal on both sides of the arrow is a balanced chemical equation • Example 2 Fe(s) + 3 Cl 2(g) 2 Fe. Cl 3(s) Reactants Products

Step 1 • List the atoms present for the reactants • List the atoms present for the products • List in this order: – metals – non-metals met a non hairy oxen? – hydrogen – oxygen last **Make sure both lists are in the same order!

Step 2 • Total atoms on the reactant side • Total atoms on the product side **Combine like atoms on the same side of the reaction

Step 3 • Balance an element by placing a coefficient in front of the chemical formula **Select coefficients that will give the same number of atoms on each side of the equation.

Step 4 • Adjust atom totals for all atoms affected by the addition of your coefficient **Other elements may have become unbalanced.

Step 5 • Continue to add coefficients and make adjustments until all elements are balanced

Helpful Hints • If you have an odd number of oxygen atoms, multiply the coefficient by 2 first to make it an even number. Then try to balance the equation. • If you end up with an odd total of a diatomic element, try doubling all coefficients in the reaction to balance out the diatomic.

Example __

Example __

Example __
- Slides: 14